News & Views |
Featured
-
-
Article |
 Non-statistical assembly of multicomponent [Pd2ABCD] cages
Non-statistical assembly of multicomponent [Pd2ABCD] cages
Metal-mediated self-assembly of organic building blocks is a powerful strategy to generate complex supramolecular objects. The non-statistical combination of multiple different components, however, has been a major challenge. Now integrative self-sorting of low-symmetry multicomponent cages has been achieved by combining shape complementarity and selective backbone interactions under thermodynamic control.
- Kai Wu
- , Elie Benchimol
- & Guido H. Clever
-
News & Views |
 Sulfur stereochemistry takes centre stage
Sulfur stereochemistry takes centre stage
Directional interactions and three-dimensional functional groups are crucial to medicinal compounds. Consequently, new functional groups require stereocontrolled synthetic methods. Now, an enantiopure building block provides controlled and divergent access to valuable sulfonimidoyl functional groups.
- James A. Bull
-
Article |
 Asymmetric synthesis of sulfoximines, sulfonimidoyl fluorides and sulfonimidamides enabled by an enantiopure bifunctional S(VI) reagent
Asymmetric synthesis of sulfoximines, sulfonimidoyl fluorides and sulfonimidamides enabled by an enantiopure bifunctional S(VI) reagent
The stereochemical control and bifunctional manipulation of chiral sulfur functional groups is a long-standing challenge. Now, an enantiopure bench-stable S(VI) fluoride exchange reagent enables the asymmetric synthesis of sulfoximines, sulfonimidamides and sulfonimidoyl fluorides. The bifunctional nature of this reagent provides a practical method for the introduction of S(VI) functionality.
- Shun Teng
- , Zachary P. Shultz
- & Justin M. Lopchuk
-
Article
| Open Access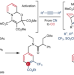 Skeletal editing of pyridines through atom-pair swap from CN to CC
Skeletal editing of pyridines through atom-pair swap from CN to CC
Skeletal editing enables diversification of compounds not possible by applying peripheral editing strategies. Now, a catalyst-free atom-pair swap strategy for pyridine editing has been developed via one-pot sequential dearomatization, cycloaddition and rearomative retrocyclization. Benzenes and naphthalenes with precisely installed functional groups are produced, and the mild conditions enable late-stage skeletal diversification of pyridine cores.
- Qiang Cheng
- , Debkanta Bhattacharya
- & Armido Studer
-
Article |
 Mirror-image trypsin digestion and sequencing of D-proteins
Mirror-image trypsin digestion and sequencing of D-proteins
The lack of effective methods for mirror-image (d-) protein sequencing hampers the development of mirror-image biology systems and related applications. Now, total chemical synthesis of mirror-image trypsin enables the sequencing of long d-peptides and d-proteins, which may facilitate applications of d-peptides and d-proteins as potential therapeutic and informational tools.
- Guanwei Zhang
- & Ting F. Zhu
-
Article |
 A molecular-level mechanistic framework for interfacial proton-coupled electron transfer kinetics
A molecular-level mechanistic framework for interfacial proton-coupled electron transfer kinetics
Although interfacial proton-coupled electron transfers are critical reaction steps in chemical and biological processes, studies investigating these reactions are complicated by surface heterogeneity. Now, interfacial proton-coupled electron transfer kinetics are studied and modelled at isolated, well-defined active sites to provide a foundation for understanding complex reactions involved in energy conversion and catalysis.
- Noah B. Lewis
- , Ryan P. Bisbey
- & Yogesh Surendranath
-
Article
| Open Access Chiral arylsulfinylamides as reagents for visible light-mediated asymmetric alkene aminoarylations
Chiral arylsulfinylamides as reagents for visible light-mediated asymmetric alkene aminoarylations
Single-electron-mediated difunctionalizations of internal olefins allow the simultaneous formation of two contiguous Csp3-stereocentres. Here, we describe enantioenriched arylsulfinylamides as all-in-one reagents for the efficient asymmetric, intermolecular aminoarylation of alkenes. Mechanistic studies revealed an interesting dichotomy in the initiation of the reaction depending on the olefin redox potential.
- Cédric Hervieu
- , Mariia S. Kirillova
- & Cristina Nevado
-
Article |
 A general alkene aminoarylation enabled by N-centred radical reactivity of sulfinamides
A general alkene aminoarylation enabled by N-centred radical reactivity of sulfinamides
Single-step addition of an aryl ring and a protected amine across an alkene is a succinct route to valuable phenethylamine products, but existing methods suffer from limited scope. Now a family of compounds containing a sulfinamide functional group have been developed to react via electrophilic radicals to yield phenethylamines through an aryl migration with precise stereochemical control.
- Efrey A. Noten
- , Cody H. Ng
- & Corey R. J. Stephenson
-
Article |
 Light-driven ammonia synthesis under mild conditions using lithium hydride
Light-driven ammonia synthesis under mild conditions using lithium hydride
Photon-driven dinitrogen reduction to ammonia is a promising but challenging reaction. Now it has been shown that lithium hydride undergoes photolysis upon ultraviolet illumination to yield long-lived photon-generated electrons residing in hydrogen vacancies, favouring the activation of the N≡N bond and achieving photocatalytic ammonia synthesis.
- Yeqin Guan
- , Hong Wen
- & Ping Chen
-
Article |
 Structural characterization and reactivity of a room-temperature-stable, antiaromatic cyclopentadienyl cation salt
Structural characterization and reactivity of a room-temperature-stable, antiaromatic cyclopentadienyl cation salt
The synthesis, structure and reactivity of room-temperature-stable [Cp(C6F5)5]+[Sb3F16]− is presented. Coordination of the cyclopentadienyl cation by [Sb3F16]− or C6F6 stabilizes the antiaromatic singlet state in the solid state. Calculated hydride and fluoride ion affinities of the [Cp(C6F5)5]+ cation are higher than those of the tritylium cation [C(C6F5)3]+.
- Yannick Schulte
- , Christoph Wölper
- & Stephan Schulz
-
Article
| Open Access Surface stratification determines the interfacial water structure of simple electrolyte solutions
Surface stratification determines the interfacial water structure of simple electrolyte solutions
The organization of electrolytes at the air/water interface affects the structure of interfacial water and therefore numerous natural processes. It has now been demonstrated that the surface of an electrolyte solution is stratified and consists of an ion-depleted outer surface and an ion-enriched subsurface layer, jointly determining the water interfacial structure.
- Yair Litman
- , Kuo-Yang Chiang
- & Mischa Bonn
-
Article
| Open Access Directed ultrafast conformational changes accompany electron transfer in a photolyase as resolved by serial crystallography
Directed ultrafast conformational changes accompany electron transfer in a photolyase as resolved by serial crystallography
Photoinduced electron transfer forms the basis for photosynthesis and DNA repair. Ultrafast structural changes recorded for a DNA-repairing photolyase now reveal specific and directed protein motions accompanying the electron transfer.
- Andrea Cellini
- , Madan Kumar Shankar
- & Sebastian Westenhoff
-
Article |
 Trapping of a phenoxyl radical at a non-haem high-spin iron(II) centre
Trapping of a phenoxyl radical at a non-haem high-spin iron(II) centre
The activation of dioxygen at metal centres, and subsequent functionalization of unactivated C‒H bonds, requires the generation of high-energy radical intermediates that often result in undesirable side reactions. Now an elusive oxygen-derived reactive iron(II)–radical intermediate is spectroscopically characterized as part of a strategy to stabilize phenoxyl radical cofactors during substrate oxidation reactions.
- Dustin Kass
- , Virginia A. Larson
- & Kallol Ray
-
Article |
 Analogies between photochemical reactions and ground-state post-transition-state bifurcations shed light on dynamical origins of selectivity
Analogies between photochemical reactions and ground-state post-transition-state bifurcations shed light on dynamical origins of selectivity
Selectivity of photochemical reactions is notoriously difficult to model. Now it has been shown that by employing an analogy to ground-state reactions with post-transition-state bifurcations, selectivity for a complex photochemical denitrogenation reaction can be captured and rationalized, and its dynamical origins understood.
- Zhitao Feng
- , Wentao Guo
- & Dean J. Tantillo
-
Article
| Open Access Accelerating the discovery of alkyl halide-derived natural products using halide depletion
Accelerating the discovery of alkyl halide-derived natural products using halide depletion
Natural products discovery remains an ongoing challenge. Now, halide depletion offers a complementary approach to discover natural products whose biosynthesis requires halides, including products of cryptic halogenation. Halide depletion reveals that nostochlorosides, the products of an orphan biosynthetic gene cluster in Nostoc punctiforme, are polymerized by a halide-displacing etherifying enzyme.
- Nathaniel R. Glasser
- , Dongtao Cui
- & Emily P. Balskus
-
Research Briefing |
 Perpendicular state of stilbene unveiled with ultrafast Raman spectroscopy
Perpendicular state of stilbene unveiled with ultrafast Raman spectroscopy
Trans–cis photoisomerization is a fundamental photochemical reaction that is thought to proceed through an intermediate with a perpendicular conformation. However, unambiguous identification of this state has proved challenging. The combination of state-of-the-art ultrafast spectroscopy and quantum chemical calculations now provides evidence for its structural observation in stilbene photoisomerization.
-
News & Views |
 Boron-enabled 1,3-metallate shift towards axially chiral alkenes
Boron-enabled 1,3-metallate shift towards axially chiral alkenes
Tetracoordinate boron molecules are the key intermediates in many organoboron-related chemical transformations. Now, using alkynyl tetracoordinate boron species, a nickel-catalysed asymmetric 1,3-metallate shift towards axial chirality has been developed, giving access to various axially chiral alkenes in high efficiency.
- Li-Qing Ren
- & Chuan He
-
Article |
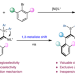 Ni-catalysed assembly of axially chiral alkenes from alkynyl tetracoordinate borons via 1,3-metallate shift
Ni-catalysed assembly of axially chiral alkenes from alkynyl tetracoordinate borons via 1,3-metallate shift
Enantioselective transformations based on tetracoordinate borons are elusive. Now, an enantioselective nickel-catalysed strategy for the construction of axially chiral alkenes has been reported, via a 1,3-metallate shift of alkynyl tetracoordinate boron species. The reaction uses readily accessible starting materials and a cheap transition-metal catalyst, and the chemoselectivity, regioselectivity and atroposelectivity could be well controlled.
- Xingxing Ma
- , Mengwei Tan
- & Qiuling Song
-
Article |
 Ultrafast Raman observation of the perpendicular intermediate phantom state of stilbene photoisomerization
Ultrafast Raman observation of the perpendicular intermediate phantom state of stilbene photoisomerization
The prototypical trans–cis photoisomerization of stilbenes is thought to occur via a transient intermediate with a perpendicular conformation—often called ‘the phantom state’—but its unambiguous identification has thus far proved difficult. Now, using ultrafast ultraviolet Raman spectroscopy and ab initio molecular dynamics simulation, evidence for its existence and its perpendicular conformation has been obtained.
- Hikaru Kuramochi
- , Takuro Tsutsumi
- & Tahei Tahara
-
Perspective |
 Photoswitchable coordination cages
Photoswitchable coordination cages
The self-assembly of stimuli-responsive building blocks yields functional nano-systems and smart materials. This Perspective discusses how the integration of photoswitches into discrete coordination cages enables control over their assembly, guest binding and systems behaviour. Four scenarios are drawn to highlight the relationship between the photoswitching and dynamic assembly equilibria.
- Elie Benchimol
- , Jacopo Tessarolo
- & Guido H. Clever
-
Article
| Open Access Probing the chemical ‘reactome’ with high-throughput experimentation data
Probing the chemical ‘reactome’ with high-throughput experimentation data
High-throughput experimentation (HTE) has great utility for chemical synthesis. However, robust interpretation of high-throughput data remains a challenge. Now, a flexible analyser has been developed on the basis of a machine learning-statistical analysis framework, which can reveal hidden chemical insights from historical HTE data of varying scopes, sizes and biases.
- Emma King-Smith
- , Simon Berritt
- & Alpha A. Lee
-
Article |
 Metastable gallium hydride mediates propane dehydrogenation on H2 co-feeding
Metastable gallium hydride mediates propane dehydrogenation on H2 co-feeding
The heterogeneous catalytic dehydrogenation of hydrocarbons usually suffers from a negative pressure dependence on H2. Now it has been shown that for propane dehydrogenation on gallium oxide-based catalysts, a positive activity dependence on H2 partial pressure arises from a metastable hydride-mediated catalysis in which gallium hydrides promote C–H activation.
- Guodong Sun
- , Zhi-Jian Zhao
- & Jinlong Gong
-
News & Views |
 How membranes influence intracellular phase separation
How membranes influence intracellular phase separation
Liquid–liquid phase separation (LLPS) within cells is a captivating phenomenon known to aid the organization of cellular components; however, its complex kinetics have remained a puzzle. Now, a new study elucidates the crosstalk between the phase state of an encapsulating membrane and LLPS dynamics.
- Rumiana Dimova
-
Thesis |
 WT19F
WT19F
Given the fraught history of fluorine, Michelle Francl wonders what made medicinal chemists consider fluorine derivatives?
- Michelle Francl
-
Article
| Open Access Chemoselective umpolung of thiols to episulfoniums for cysteine bioconjugation
Chemoselective umpolung of thiols to episulfoniums for cysteine bioconjugation
Cysteine bioconjugation is an important method to modify biomolecules, but synthetic efforts to diversify reactive warheads and the low reactivity of introducible linchpins often impede application in biological laboratories. Now, a thianthrenium-based reagent permits site-selective installation of episulfonium on biomacromolecules, enabling one-step addition of bioorthogonal nucleophiles and further applications in quantitative proteomics and cross-linking.
- Philipp Hartmann
- , Kostiantyn Bohdan
- & Tobias Ritter
-
Article |
 Molecular jackhammers eradicate cancer cells by vibronic-driven action
Molecular jackhammers eradicate cancer cells by vibronic-driven action
In contrast to photothermal therapy requiring high powers over extended times and photodynamic therapy being abrogated by inhibitors of reactive oxygen species, actuation of vibronic modes in single molecules—molecular jackhammers—can now induce efficient cancer cell death. Here, the mechanical disassembly of cell membranes is characterized as the underlying mechanism by which this vibronic-driven action promotes necrotic cell death.
- Ciceron Ayala-Orozco
- , Diego Galvez-Aranda
- & James M. Tour
-
News & Views |
 Engineering cyanine cyclizations for new fluorogenic probes
Engineering cyanine cyclizations for new fluorogenic probes
As the need for specific fluorescent probes that enable high sensitivity and super-resolution imaging experiments continues to grow, it is imperative to develop new, well-characterized methods to modulate the emission of fluorophores. Now, a general platform affords visible-to-NIR fluorogenic fluorophores by engineering a simple cyclization event into cyanine dyes.
- Quintashia D. Wilson
- & Ellen M. Sletten
-
Article |
 Proteolysis-targeting chimeras with reduced off-targets
Proteolysis-targeting chimeras with reduced off-targets
Current proteolysis-targeting chimeras can promote the ubiquitination and subsequent degradation of both target and off-target proteins by inducing their respective proximity with the cereblon ubiquitin ligase. Now, by developing and deploying an off-target profiling platform, ‘bumped proteolysis-targeting chimeras’ can maintain on-target degradation efficacy with reduced off-targets.
- Tuan M. Nguyen
- , Vedagopuram Sreekanth
- & Amit Choudhary
-
News & Views |
 Isotropic models for anisotropic inorganics
Isotropic models for anisotropic inorganics
The study of disordered materials poses numerous challenges, and computational approaches have proved useful to supplement and support structural experiments. Now, an abstract computational model has been used to study the structure of amorphous calcium carbonate, providing mechanistic insights into the emergence of the disordered phase as well as its atomic-level configurations.
- Julia Dshemuchadse
-
Article |
 Click processes orthogonal to CuAAC and SuFEx forge selectively modifiable fluorescent linkers
Click processes orthogonal to CuAAC and SuFEx forge selectively modifiable fluorescent linkers
Orthogonal click reactions enable the rapid assembly of molecules of all sizes. Now, it has been shown that a nitrile, an allene, a diborane and a hydrazine or aniline can be catalytically merged in a click process that is orthogonal to SuFEx and CuAAC, delivering fluorescent linkages.
- Paulo H. S. Paioti
- , Katherine E. Lounsbury
- & Amir H. Hoveyda
-
Article |
 Modular α-tertiary amino ester synthesis through cobalt-catalysed asymmetric aza-Barbier reaction
Modular α-tertiary amino ester synthesis through cobalt-catalysed asymmetric aza-Barbier reaction
Robust protocols for the synthesis of chiral α-tertiary amino acids remain scarce due to the challenge of constructing congested tetrasubstituted stereocentres. Now a cobalt-catalysed enantioselective aza-Barbier reaction of ketimines with various unactivated alkyl halides has been developed, forming diverse chiral α-tertiary amino esters with high enantioselectivity and excellent functional group tolerance.
- Xianqing Wu
- , Hanyu Xia
- & Yifeng Chen
-
Article |
 Organocatalytic aromatization-promoted umpolung reaction of imines
Organocatalytic aromatization-promoted umpolung reaction of imines
The umpolung functionalization of imines bears vast synthetic potential, but polarity inversion is less efficient compared with the carbonyl counterparts. Now, an alternative strategy exploiting chiral phosphoric acid catalytic aromatization has been developed, affording structures possessing a central chirality or a stereogenic C–N axis with high efficiency and enantiocontrol.
- Ye-Hui Chen
- , Meng Duan
- & Bin Tan
-
Article |
 A general copper-catalysed enantioconvergent C(sp3)–S cross-coupling via biomimetic radical homolytic substitution
A general copper-catalysed enantioconvergent C(sp3)–S cross-coupling via biomimetic radical homolytic substitution
Methods for transition-metal-catalysed enantioselective C(sp3)–S bond construction are underdeveloped. Now, by taking advantage of the biomimetic radical homolytic substitution manifold, the copper-catalysed enantioconvergent C(sp3)–S cross-coupling of racemic secondary and tertiary alkyl halides with highly transformable sulfur nucleophiles has been realized. This reaction provides access to an array of α-chiral alkyl organosulfur compounds.
- Yu Tian
- , Xi-Tao Li
- & Xin-Yuan Liu
-
Article |
 Cooperative H2 activation at a nickel(0)–olefin centre
Cooperative H2 activation at a nickel(0)–olefin centre
Activation of H2 by a metal–olefin complex is characterized experimentally and computationally using a nickel pincer complex, showing that the reaction proceeds via a direct ligand-to-ligand hydrogen transfer mechanism. An application of this cooperative H2-activation mechanism is demonstrated in the nickel-catalysed semihydrogenation of diphenylacetylene.
- María L. G. Sansores-Paredes
- , Martin Lutz
- & Marc-Etienne Moret
-
Article |
 Air- and photo-stable luminescent carbodicarbene-azaboraacenium ions
Air- and photo-stable luminescent carbodicarbene-azaboraacenium ions
Borenium ions have traditionally served as main group (pre-)catalysts, and their use in materials-related applications have been limited by their instability. Now, a series of fully π-conjugated azaboraacenium ions derived from carbodicarbene have been developed that exhibit high air and moisture stability with full colour-tunable luminescence. Furthermore, these azaboraacene cations mimic the electronic structures of higher-order carbonaceous acenes while featuring enhanced resistance to photo-oxidation.
- Chun-Lin Deng
- , Akachukwu D. Obi
- & Robert J. Gilliard Jr.
-
Article |
 Azobenzene as a photoswitchable mechanophore
Azobenzene as a photoswitchable mechanophore
Light-induced azobenzene cis/trans isomerization has been extensively investigated, but the mechanical strength of its cis/trans structure is not well understood. Now it has been shown that cis azobenzene is mechanically less stable than the trans isomer due to its regiochemical structure, as revealed by single-molecule force spectroscopy.
- Yiran Li
- , Bin Xue
- & Yi Cao
-
Article
| Open Access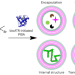 Artificial cell synthesis using biocatalytic polymerization-induced self-assembly
Artificial cell synthesis using biocatalytic polymerization-induced self-assembly
Enzyme-initiated polymerization-induced self-assembly has been used to generate various biomimetic structures. Now, myoglobin’s activity is used for biocatalytic polymerization-induced self-assembly to generate vesicular artificial cells. As various cargoes can be encapsulated during polymerization, these artificial cells are capable of protein expression and can act as microreactors for distinct enzymatic reactions.
- Andrea Belluati
- , Sètuhn Jimaja
- & Nico Bruns
-
Article |
 Subdomain dynamics enable chemical chain reactions in non-ribosomal peptide synthetases
Subdomain dynamics enable chemical chain reactions in non-ribosomal peptide synthetases
Many natural products are produced by non-ribosomal peptide synthetases in an assembly-line fashion. How these molecular machines orchestrate the biochemical sequences has remained elusive. It is now understood that an extended-conformation ensemble is needed to coordinate chemical-transformation steps whereas the biosynthesis directionality is driven by the enzyme’s innate conformational free energies.
- Xun Sun
- , Jonas Alfermann
- & Haw Yang
-
News & Views |
 From feedstock to pharmaceuticals
From feedstock to pharmaceuticals
Fluoroalkyl fragments are ubiquitous motifs in pharmaceuticals and agrochemicals, but their introduction to a given molecule typically involves expensive or difficult-to-handle reagents. Now, the photocatalysed hydrofluoroalkylation of alkenes has been achieved using simple and readily available fluoroalkyl carboxylic acids.
- Fabio Juliá
-
News & Views |
 An acid for an acid
An acid for an acid
Stereoselective decarboxylative protonation can produce diverse chiral molecules from widely available carboxylic acids. However, general and practical strategies are lacking. Now, a chiral spirocyclic phosphoric acid-catalysed decarboxylation of aminomalonic acids has enabled the modular synthesis of α-amino acids.
- Xufeng Lin
- & Alemayehu Gashaw Woldegiorgis
-
Article |
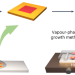 Lateral epitaxial growth of two-dimensional organic heterostructures
Lateral epitaxial growth of two-dimensional organic heterostructures
The synthesis of two-dimensional (2D) organic lateral heterostructures with desirable properties from organic single crystals remains challenging. Now, 2D organic lateral heterostructures have been produced by using a liquid-phase growth approach and vapour-phase growth method, enabling the structural inversion of organic lateral heterostructures via a two-step strategy.
- Qiang Lv
- , Xue-Dong Wang
- & Liang-Sheng Liao
-
Article
| Open Access A general strategy to develop fluorogenic polymethine dyes for bioimaging
A general strategy to develop fluorogenic polymethine dyes for bioimaging
Polymethine dyes are bright and red-shifted fluorophores that lack an intrinsic turn-on mechanism, which leads to non-specific staining when applied to biological samples. Now the fluorescence of polymethine dyes was masked through an intracellular cyclization strategy that gets reversed upon binding an intended macromolecular target, providing specificity for live-cell imaging.
- Annabell Martin
- & Pablo Rivera-Fuentes
-
Article
| Open Access Enabling late-stage drug diversification by high-throughput experimentation with geometric deep learning
Enabling late-stage drug diversification by high-throughput experimentation with geometric deep learning
Late-stage functionalization of complex drug molecules is challenging. To address this problem, a discovery platform based on geometric deep learning and high-throughput experimentation was developed. The computational model predicts binary reaction outcome, reaction yield and regioselectivity with low error margins, enabling the functionalization of complex molecules without de novo synthesis.
- David F. Nippa
- , Kenneth Atz
- & Gisbert Schneider
-
Research Briefing |
 Phase-separation behaviour of RNAs
Phase-separation behaviour of RNAs
Ribonucleoprotein granules are ubiquitous in living organisms with the protein and RNA components having distinct roles. In the absence of proteins, RNAs are shown to undergo phase separation upon heating. This transition is driven by desolvation entropy and ion-mediated crosslinking and is tuned by the chemical specificity of the RNA nucleobases.
-
Article |
 Remote stereocontrol with azaarenes via enzymatic hydrogen atom transfer
Remote stereocontrol with azaarenes via enzymatic hydrogen atom transfer
The inherent rigidity of the azaarene ring structure has made it challenging to achieve remote stereocontrol through asymmetric catalysis on these substrates. Now, through a photoenzymatic process, an ene-reductase system facilitates the production of diverse azaarenes with distant γ-stereocentres, highlighting the potential of biocatalysts for stereoselectivity at remote sites.
- Maolin Li
- , Wesley Harrison
- & Huimin Zhao
-
Article |
 Modular and diverse synthesis of amino acids via asymmetric decarboxylative protonation of aminomalonic acids
Modular and diverse synthesis of amino acids via asymmetric decarboxylative protonation of aminomalonic acids
Asymmetric decarboxylation can transform abundant carboxylic acids into valuable chiral molecules but faces major limitations due to the challenging enantiocontrol of proton transfer. Now the use of Brønsted acid catalysis in conjunction with an anchoring group strategy has enabled the decarboxylative protonation of aminomalonic acids to access diverse amino acids.
- Wei-Feng Zheng
- , Jingdan Chen
- & Zhongxing Huang
-
Article
| Open Access Masked alkynes for synthesis of threaded carbon chains
Masked alkynes for synthesis of threaded carbon chains
Long polyynes have fascinating properties but they are difficult to synthesize as a consequence of their high reactivity. Now, it has been shown that cobalt carbonyl complexes can be used as masked alkyne equivalents, enabling the preparation of stable polyyne polyrotaxanes with up to 34 contiguous triple bonds.
- Connor W. Patrick
- , Yueze Gao
- & Harry L. Anderson
-
Article |
 Photocatalytic hydrofluoroalkylation of alkenes with carboxylic acids
Photocatalytic hydrofluoroalkylation of alkenes with carboxylic acids
Alkene hydrofluoroalkylation offers a promising route to diverse fluoroalkylated compounds but current methods have limitations, such as needing expensive fluoroalkylating reagents. Now, leveraging iron photocatalysis and hydrogen-atom-transfer catalysis, a hydrofluoroalkylation method has been developed that utilizes feedstock chemicals such as trifluoroacetic acid as direct fluoroalkyl radical precursors, providing a redox-neutral, general protocol to introduce fluoroalkyl moieties.
- Kang-Jie Bian
- , Yen-Chu Lu
- & Julian G. West
-
Research Briefing |
 In vitro selection of covalent aptamers with sulfur(VI) fluoride exchange chemistry
In vitro selection of covalent aptamers with sulfur(VI) fluoride exchange chemistry
To develop covalent inhibitors with high potency and low off-target effects, combinatorial approaches that search for candidates from large libraries are desired. Here, sulfur(VI) fluoride exchange (SuFEx) in vitro selection is established for the evolution of covalent aptamers from trillions of SuFEx-modified oligonucleotides. Through this technique, covalent aptamers with optimally balanced selectivity and reactivity are identified.
Browse broader subjects
Browse narrower subjects
- Analytical chemistry
- Biochemistry
- Biosynthesis
- Catalysis
- Chemical biology
- Chemical education
- Chemical engineering
- Chemical safety
- Cheminformatics
- Chemistry publishing
- Communicating chemistry
- Coordination chemistry
- Electrochemistry
- Energy
- Environmental chemistry
- Green chemistry
- History of chemistry
- Inorganic chemistry
- Materials chemistry
- Medicinal chemistry
- Nuclear chemistry
- Organic chemistry
- Chemical origin of life
- Photochemistry
- Physical chemistry
- Polymer chemistry
- Process chemistry
- Supramolecular chemistry
- Surface chemistry
- Chemical synthesis
- Theoretical chemistry

 Doing away with radium’s proxies
Doing away with radium’s proxies