Thesis |
Featured
-
-
News & Views |
 Photophysical oxidation of atmospheric carbonyls
Photophysical oxidation of atmospheric carbonyls
Light is a major driver of the chemistry of the atmosphere and usually involves the photolytic fragmentation of molecules into radicals before their reaction. New results show that formaldehyde, excited by low-energy light, can react with oxygen, opening up alternative atmospheric oxidation pathways.
- Paul W. Seakins
-
Article |
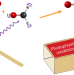 Photophysical oxidation of HCHO produces HO2 radicals
Photophysical oxidation of HCHO produces HO2 radicals
In the atmosphere, photolysis of formaldehyde generates H and HCO radicals, which then react with O2 to form HO2 (important in converting atmospheric carbon to CO2). Now it has been shown that internally excited formaldehyde can also react with atmospheric O2 to make HO2 in a direct, one-step ‘photophysical oxidation’, a mechanism likely to be general in the troposphere.
- Blair A. Welsh
- , Maggie E. Corrigan
- & Scott H. Kable
-
Article
| Open Access The gas-phase formation mechanism of iodic acid as an atmospheric aerosol source
The gas-phase formation mechanism of iodic acid as an atmospheric aerosol source
Iodic acid (HIO3) forms aerosols very efficiently, but its gas-phase formation mechanism is not well understood. Atmospheric simulation chamber experiments, quantum chemical calculations and kinetic modelling have now revealed that HIO3 forms as an early iodine oxidation product from hypoiodite. The mechanism explains field measurements and suggests a catalytic role for iodine in particle formation.
- Henning Finkenzeller
- , Siddharth Iyer
- & Rainer Volkamer
-
Thesis |
 Beyond Hubble
Beyond Hubble
Sit back and settle in for a tour of the chemical analysis instruments aboard the James Webb Space Telescope and the techniques being used to explore the planets beyond our Solar System.
- Bruce C. Gibb
-
News & Views |
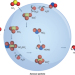 An interfacial role for NO2
An interfacial role for NO2
Atmospheric chemists have been unable to explain the rapid sulfate formation observed during wintertime aerosol pollution events. Now, kinetic measurements in atmospherically relevant aerosol particles have highlighted a significant role for nitrogen dioxide in sulfate formation via its interfacial reaction with dissolved SO2.
- Jian Zhen Yu
-
Article |
 Oxidation of sulfur dioxide by nitrogen dioxide accelerated at the interface of deliquesced aerosol particles
Oxidation of sulfur dioxide by nitrogen dioxide accelerated at the interface of deliquesced aerosol particles
In polluted atmospheres, NO2 oxidation of SO2 in particles is a potentially important reaction leading to sulfate aerosol formation. Now it has been shown that the reaction occurs much faster than predicted by known bulk solution chemistry, implicating an interfacial mechanism.
- Tengyu Liu
- & Jonathan P. D. Abbatt
-
Q&A |
 Change is in the air
Change is in the air
Krystal Vasquez, PhD student at Caltech, talks to Nature Chemistry about her experiences as an atmospheric researcher — both in the lab with her favourite instruments and in the scientific community as a disabled scientist — as well as her love for science writing.
- Anne Pichon
-
Thesis |
 The road to hell is irreversible
The road to hell is irreversible
The hole in the ozone layer is a huge warning sign that society is in danger of ignoring, argues Bruce C. Gibb, who reflects on the legacy of Paul Crutzen and the Anthropocene.
- Bruce C. Gibb
-
Thesis |
 Chemistry of the sky god
Chemistry of the sky god
If you feel like shifting your attention away from what’s happening here on planet Earth in 2020 for a moment, join Bruce C. Gibb on a tour through the chemistry of the largest planet in the Solar System.
- Bruce C. Gibb
-
Comment |
 The complex chemical effects of COVID-19 shutdowns on air quality
The complex chemical effects of COVID-19 shutdowns on air quality
Stay-at-home policies invoked in response to COVID-19 have led to well-publicized drops in some air pollutants. The extent to which such reductions translate to improved air quality is dictated by not only emissions and meteorology, but also chemical transformations in the atmosphere.
- Jesse H. Kroll
- , Colette L. Heald
- & Allison L. Steiner
-
Article |
 Coupling N2 and CO2 in H2O to synthesize urea under ambient conditions
Coupling N2 and CO2 in H2O to synthesize urea under ambient conditions
Conventionally, urea is synthesized via two consecutive processes, N2 + H2 → NH3 followed by NH3 + CO2. Now, an electrocatalyst consisting of PdCu alloy nanoparticles on TiO2 nanosheets has been shown to directly couple N2 and CO2 in H2O to produce urea under ambient conditions.
- Chen Chen
- , Xiaorong Zhu
- & Shuangyin Wang
-
Thesis |
 Lightning-fast chemistry
Lightning-fast chemistry
When lightning strikes, Bruce C. Gibb reminds us that not only do sparks fly, but there’s also some chemistry going on too.
- Bruce C. Gibb
-
Thesis |
 Abiogenesis and the reverse Hofmeister effect
Abiogenesis and the reverse Hofmeister effect
Bruce C. Gibb takes us on a journey through the physical and chemical evolution of planet Earth and suggests that the reverse Hofmeister effect, the phenomenon whereby poorly solvated ions associate in water, could be a powerful driving force towards the first hint of life on the rock we call home.
- Bruce C. Gibb
-
Article |
 O2−O2 and O2−N2 collision-induced absorption mechanisms unravelled
O2−O2 and O2−N2 collision-induced absorption mechanisms unravelled
Molecular collisions can lead to the absorption of incident light even for transitions that are spectroscopically forbidden for the isolated molecules. Now the electronic–vibrational transitions of O2 have been theoretically studied and, contrary to textbook knowledge, it is shown that the absorption mechanism and the spectral line shape depend on the collision partner, oxygen or nitrogen.
- Tijs Karman
- , Mark A. J. Koenis
- & Gerrit C. Groenenboom
-
Article |
 Chemical evolution of atmospheric organic carbon over multiple generations of oxidation
Chemical evolution of atmospheric organic carbon over multiple generations of oxidation
Nearly all organic carbon has now been quantified and characterized in a highly complex evolving atmospheric system, specifically, the multigenerational oxidation of α-pinene. It has been observed that initial addition of functional groups quickly gives way to fragmentation reactions, with organic carbon ultimately becoming sequestered in chemically resistant reservoirs: organic aerosols and long-lived gas-phase species.
- Gabriel Isaacman-VanWertz
- , Paola Massoli
- & Jesse H. Kroll
-
Editorial |
Life, but not as we know it
There are many unanswered questions regarding how the biomolecules and biomechanical processes that define life came to be. A collection of Articles in this issue show how intermediates in RNA synthesis might have formed and how the initiation and evolution of RNA replication might have occurred.
-
-
Article |
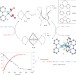 Hydrolytic cleavage of both CS2 carbon–sulfur bonds by multinuclear Pd(II) complexes at room temperature
Hydrolytic cleavage of both CS2 carbon–sulfur bonds by multinuclear Pd(II) complexes at room temperature
Converting CS2 and COS pollutants into benign products is critical in eliminating waste exhaust fumes. Now, a series of air-stable palladium complexes mediate hydrolysis of both CS2 carbon–sulfur bonds at 25 °C to produce CO2. Oxidation of the resulting complexes regenerates the starting complexes with SO2 and NO2 release.
- Xuan-Feng Jiang
- , Hui Huang
- & Tobin J. Marks
-
Thesis |
 A brief history of water
A brief history of water
Michelle Francl wonders just how old the water in her tea is.
- Michelle Francl
-
Thesis |
 Weird and wonderful water
Weird and wonderful water
When it comes to water and its unique properties, Bruce Gibb explains that although there is undoubtedly some mystery, there is certainly no magic.
- Bruce C. Gibb
-
-
Article |
 Unimolecular dissociation dynamics of vibrationally activated CH3CHOO Criegee intermediates to OH radical products
Unimolecular dissociation dynamics of vibrationally activated CH3CHOO Criegee intermediates to OH radical products
An important source of atmospheric hydroxyl radicals is from the dissociation of Criegee intermediates produced in alkene ozonolysis reactions. The dissociation dynamics of the prototypical CH3CHOO Criegee intermediate have now been determined. Complementary experimental and theoretical studies were carried out and the translational and internal energy distributions of the OH radical products were characterized.
- Nathanael M. Kidwell
- , Hongwei Li
- & Marsha I. Lester
-
Thesis |
 The organic Solar System
The organic Solar System
In the second of two essays looking at organic chemistry that can be found in the Solar System, Bruce C. Gibb focuses on the gas and ice giants as well as their satellites — concluding the tour on Saturn's fascinating moon Titan.
- Bruce C. Gibb
-
Thesis |
 Faith, chemistry and extraterrestrial life
Faith, chemistry and extraterrestrial life
Bruce Gibb wonders whether our faith in chemistry — and what it can teach us about the Universe beyond our Earthly bounds — will have a role to play in the search for alien life.
- Bruce C. Gibb
-
-
Article |
 Irreversible xenon insertion into a small-pore zeolite at moderate pressures and temperatures
Irreversible xenon insertion into a small-pore zeolite at moderate pressures and temperatures
Several solutions to the ‘missing xenon’ problem have been proposed that involve the selective sorption of Xe in minerals found in the Earth. It is now shown that a zeolite, Ag-natrolite, absorbs and retains 28 wt% Xe at 1.7 GPa and 250 °C, conditions found in subsurface Earth, through expulsion of metallic Ag(0).
- Donghoon Seoung
- , Yongmoon Lee
- & Yongjae Lee
-
News & Views |
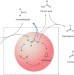 Intermediates just want to react
Intermediates just want to react
Many of the rate parameters used in models of tropospheric chemistry are obtained through laboratory ozonolysis experiments. Now, results on the self-reaction of an important, but long-elusive, intermediate could alter many of those inferences.
- Craig A. Taatjes
- , Dudley E. Shallcross
- & Carl J. Percival
-
Article |
 Reactions of xenon with iron and nickel are predicted in the Earth's inner core
Reactions of xenon with iron and nickel are predicted in the Earth's inner core
Studies of the Earth's atmosphere have shown that more than 90% of xenon is depleted — the so-called missing Xe paradox. Now a theoretical study shows that Xe and Fe/Ni can form inter-metallic compounds of XeFe3 and XeNi3 under conditions found in the Earth's inner core, and could provide a solution to the puzzle.
- Li Zhu
- , Hanyu Liu
- & Yanming Ma
-
Article |
 Extremely rapid self-reaction of the simplest Criegee intermediate CH2OO and its implications in atmospheric chemistry
Extremely rapid self-reaction of the simplest Criegee intermediate CH2OO and its implications in atmospheric chemistry
Criegee intermediates play an important role in atmospheric chemistry but their direct study has proved difficult. Transient infrared absorption spectroscopy has now been used to probe the decay kinetics of the Criegee intermediate CH2OO directly, revealing that its self-reaction is extremely rapid. This may have important consequences for the interpretation of previous laboratory experiments.
- Yu-Te Su
- , Hui-Yu Lin
- & Yuan-Pern Lee
-
In Your Element |
 Mesmerized by mercury
Mesmerized by mercury
Joel D. Blum considers the two faces of mercury. It has many unique and useful properties in chemistry — yet it comes with a dark and dangerous side.
- Joel D. Blum
-
-
-
Article |
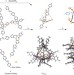 Anion-induced reconstitution of a self-assembling system to express a chloride-binding Co10L15 pentagonal prism
Anion-induced reconstitution of a self-assembling system to express a chloride-binding Co10L15 pentagonal prism
A coordination cage has been prepared that self-assembles through second-order templation. Peripheral perchlorate or hexafluorophosphate template anions direct the formation of a hollow prism whose central pocket was able to bind a small anionic guest such as halide or azide, in a manner reminiscent to signal transduction in biological systems.
- Imogen A. Riddell
- , Maarten M. J. Smulders
- & Jonathan R. Nitschke
-
-
In Your Element |
 Meteoric calcium
Meteoric calcium
Calcium is found throughout the solar system, the Earth's crust and oceans, and is an essential constituent of cells, shells and bones — yet it is curiously scarce in the upper atmosphere. John Plane ponders on this 25-year-old mystery.
- John M. C. Plane
-
In Your Element |
 Anisotropic dysprosium
Anisotropic dysprosium
Beginning with its origins as the archetypal and eponymously elusive rare-earth element, Dante Gatteschi explains why dysprosium and other lanthanides have cornered the market in molecular magnetism.
- Dante Gatteschi
-
News & Views |
 Roaming in the dark
Roaming in the dark
The mechanism of NO and O2 production through the photolysis of the nitrate radical has long proved elusive. Now, theoretical studies help to explain previous experiments, suggesting that two distinct pathways are responsible that both involve 'roaming dynamics' on the 'dark' excited electronic state.
- Simon W. North
-
Article |
 Near-threshold H/D exchange in CD3CHO photodissociation
Near-threshold H/D exchange in CD3CHO photodissociation
Unexpected and significant isotope exchange is observed in the near-threshold photodissociation of isopically labelled acetaldehyde. Theoretical modelling indicates that, at the lowest energies considered, an average of 20 H- or D-shifts occur before dissociation — evidence for extensive isomerization.
- Brianna R. Heazlewood
- , Alan T. Maccarone
- & Scott H. Kable
-
Article |
 The role of long-lived reactive oxygen intermediates in the reaction of ozone with aerosol particles
The role of long-lived reactive oxygen intermediates in the reaction of ozone with aerosol particles
It is shown that long-lived reactive oxygen intermediates are formed in heterogeneous reactions of ozone with aerosol particles, resolving apparent discrepancies between earlier quantum mechanical calculations and kinetic experiments. These intermediates play a key role in the chemical transformations and adverse health effects of toxic and allergenic air particulates.
- Manabu Shiraiwa
- , Yulia Sosedova
- & Ulrich Pöschl
-
News & Views |
 The reaction that wouldn't quit
The reaction that wouldn't quit
The radical–radical association reaction between hydroxyl and nitrogen dioxide plays a central role in atmospheric chemistry but has challenged experimentalists for decades. A study now measures all reactants and products and largely settles the issue.
- Neil M. Donahue
-
In Your Element |
 In the nickel of time
In the nickel of time
Although reports on the use of nickel can be traced back to 3,500 BC, Catherine Drennan points to a resurgence of interest in nickel-based chemistry in the energy and environmental areas.
- Catherine L. Drennan
-
Review Article |
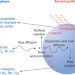 Heterogeneous oxidation of atmospheric aerosol particles by gas-phase radicals
Heterogeneous oxidation of atmospheric aerosol particles by gas-phase radicals
Aerosol particles are crucially important to the chemistry of the atmosphere, influencing both climate and air quality. This Review discusses progress in understanding the radical-initiated heterogeneous oxidation of atmospheric aerosols, focusing on the reaction mechanisms and kinetics, and also their effects on the physico-chemical properties of particles.
- I. J. George
- & J. P. D. Abbatt
-
Research Highlights |
 Buckyballs in space
Buckyballs in space
Infrared measurements from a space telescope have identified fullerene molecules in a planetary nebula.
- Neil Withers
-
Research Highlights |
A molecular affair
The intrinsic characteristics that determine the global warming potential of molecules can be assessed, so that compounds with minimum impact on the climate can be prepared for industrial applications.
- Anne Pichon
-
Research Highlights |
 Environmental issues
Environmental issues
A computational investigation into how environmental factors influence phosphoester hydrolysis reveals differences between the mechanisms observed in aqueous solution and within an enzyme active site.
- Gavin Armstrong
-
Article |
 Formation of mixed-phase particles during the freezing of polar stratospheric ice clouds
Formation of mixed-phase particles during the freezing of polar stratospheric ice clouds
Chlorine-activation reactions on polar stratospheric cloud (PSC) particles are crucial to ozone depletion in the winter/spring polar stratosphere and their rates depend on the phase state of the PSC particle surface. Now experiments show that, on particle formation, a phase separation into pure ice with a residual solution coating takes place.
- Anatoli Bogdan
- , Mario J. Molina
- & Thomas Loerting
-
Research Highlights |
Particle particulars
Key intermediates and their roles in secondary organic aerosol formation from isoprene have been elucidated.
- Gavin Armstrong
-
Research Highlights |
Growing old together
Atmospheric organic aerosols from very different sources evolve towards similar characteristics, simplifying the models needed to investigate their effects on climate and air quality.
- Anne Pichon

 The search for Europan life
The search for Europan life Nitrate NO more
Nitrate NO more Captivating COFs
Captivating COFs Catalysis in the clouds
Catalysis in the clouds