News & Views |
Featured
-
-
Article |
 Non-canonical MLL1 activity regulates centromeric phase separation and genome stability
Non-canonical MLL1 activity regulates centromeric phase separation and genome stability
Sha et al. show that Borealin is a non-canonical methylation substrate for MLL1 that regulates phase separation of the inner centromeric chromosome passenger complex, and dictates genome integrity and tumour development in hepatocellular carcinoma.
- Liang Sha
- , Zi Yang
- & Yali Dou
-
News & Views |
 Guiding DNA repair at the nuclear periphery
Guiding DNA repair at the nuclear periphery
The nuclear envelope participates in the spatial regulation of DNA repair, but the mechanisms behind this are unclear. A study now reports that a nuclear envelope-localized nuclease, NUMEN/ENDOD1, guides the choice of DNA-repair pathway by inhibiting the resection of DNA ends and aberrant recombination, ensuring genome stability.
- Sylvain Audibert
- & Evi Soutoglou
-
Article
| Open Access Actin-driven chromosome clustering facilitates fast and complete chromosome capture in mammalian oocytes
Actin-driven chromosome clustering facilitates fast and complete chromosome capture in mammalian oocytes
Harasimov et al. show that, in human and porcine oocytes, actin cables and microtubule loops move chromosomes into a cluster before spindle assembly to ensure fast and complete chromosome capture in meiosis.
- Katarina Harasimov
- , Julia Uraji
- & Melina Schuh
-
News & Views |
 Telomeres are a life-extending gift
Telomeres are a life-extending gift
Cellular senescence induced by DNA replication and telomere attrition contributes to organ dysfunction, inflammation and impaired immunity. A study reveals that antigen-presenting cells provide telomeric DNA to CD4+ T cells in synaptic contact, which enables the suspension of senescence, T cell expansion and long-lived immunity.
- Anna Carey
- , Laura Niedernhofer
- & Christina Camell
-
Article |
 An intercellular transfer of telomeres rescues T cells from senescence and promotes long-term immunological memory
An intercellular transfer of telomeres rescues T cells from senescence and promotes long-term immunological memory
Lanna and colleagues discover extracellular vesicle-mediated transfer of telomeres from antigen-presenting cells to T cells, which enables elongation of chromosomes, protection against replicative senescence and long-term immune defence.
- Alessio Lanna
- , Bruno Vaz
- & Michael Karin
-
Article |
 Epigenetic, genetic and maternal effects enable stable centromere inheritance
Epigenetic, genetic and maternal effects enable stable centromere inheritance
Das et al. show that chromatin incorporation of histone H3 variant CENP-A at centromeres in early mouse embryos depends on the maternally provided Cenpa mRNA pool and on repetitive centromere satellite DNA, rather than pre-existing CENP-A nucleosomes.
- Arunika Das
- , Aiko Iwata-Otsubo
- & Michael A. Lampson
-
Letter
| Open Access Cell cycle-specific phase separation regulated by protein charge blockiness
Cell cycle-specific phase separation regulated by protein charge blockiness
Yamazaki et al. show that cell cycle-regulated changes in hyperphosphorylation of Ki-67 and NPM1 modulate alternating charge blocks in these proteins, which defines their propensity for liquid–liquid phase separation at chromatin.
- Hiroya Yamazaki
- , Masatoshi Takagi
- & Shige H. Yoshimura
-
Review Article |
 Telomere dysfunction in ageing and age-related diseases
Telomere dysfunction in ageing and age-related diseases
Rossiello et al. review the contributions of telomere shortening and/or dysfunction to ageing and a broad spectrum of age-associated human diseases.
- Francesca Rossiello
- , Diana Jurk
- & Fabrizio d’Adda di Fagagna
-
News & Views |
 Cohesin puts a break on distal homology hunt
Cohesin puts a break on distal homology hunt
During the repair of DNA breaks by homologous recombination, damaged DNA seeks out intact, similar in sequence segments to use as a template. In this issue, Piazza, Bordelet and colleagues describe three-dimensional chromosome architecture remodelling during homologous recombination and show that cohesin restricts homology search in cis, independently of sister chromatid cohesion.
- Argyris Papantonis
- & Vassilis Roukos
-
Article |
 Diverse heterochromatin-associated proteins repress distinct classes of genes and repetitive elements
Diverse heterochromatin-associated proteins repress distinct classes of genes and repetitive elements
McCarthy, Kaeding et al. identify H3K9me3-heterochromatin proteins that repress heterochromatic genes with implications in hepatic reprogramming and show that ERH is key to global H3K9me3 maintenance in human cells.
- Ryan L. McCarthy
- , Kelsey E. Kaeding
- & Kenneth S. Zaret
-
Article |
 A chromosome folding intermediate at the condensin-to-cohesin transition during telophase
A chromosome folding intermediate at the condensin-to-cohesin transition during telophase
Abramo et al. show that during mitosis, condensin-mediated loops are lost by telophase and a transient chromosome folding intermediate is formed that lacks cohesin-generated loops.
- Kristin Abramo
- , Anne-Laure Valton
- & Job Dekker
-
News & Views |
 Centromere maintenance during DNA replication
Centromere maintenance during DNA replication
Centromere identity must be maintained through multiple generations. A new study reveals a Constitutive Centromere-Associated Network (CCAN)-dependent retention of CENP-A, a key epigenetic mark for centromeres, in centromeres during DNA replication and a replication-dependent error correction to eliminate ectopic CENP-A in chromosome arms.
- Masatoshi Hara
- & Tatsuo Fukagawa
-
Article |
 DNA replication acts as an error correction mechanism to maintain centromere identity by restricting CENP-A to centromeres
DNA replication acts as an error correction mechanism to maintain centromere identity by restricting CENP-A to centromeres
Cleveland and colleagues report a DNA-replication-dependent error correction mechanism that acts in S phase to remove ectopically loaded CENP-A outside the centromeres, while retaining centromere-bound CENP-A.
- Yael Nechemia-Arbely
- , Karen H. Miga
- & Don W. Cleveland
-
News & Views |
 Y chromothripsis?
Y chromothripsis?
Micronucleation of missegregated chromatin can lead to substantial chromosome rearrangements via chromothripsis. However, the molecular details of micronucleus-based chromothripsis are still unclear. Now, an elegant system that specifically induces missegregation of the Y chromosome provides insight into this process, including a role for non-homologous end joining.
- Emily M. Hatch
-
Letter |
 Selective Y centromere inactivation triggers chromosome shattering in micronuclei and repair by non-homologous end joining
Selective Y centromere inactivation triggers chromosome shattering in micronuclei and repair by non-homologous end joining
Ly et al. establish a method to selectively inactivate the centromere of the Y chromosome to follow chromosome shattering and micronuclei formation through several cell cycles, and suggest re-ligation of chromosome fragments is dependent on non-homologous end joining.
- Peter Ly
- , Levi S. Teitz
- & Don W. Cleveland
-
Article |
 DNA damage signalling targets the kinetochore to promote chromatin mobility
DNA damage signalling targets the kinetochore to promote chromatin mobility
Durocher and colleagues find that in budding yeast, the movement of chromosomes induced by DNA breaks is due to the loss of attachment of kinetochores to spindle pole bodies and of telomeres to the nuclear periphery, and may promote checkpoint arrest.
- Jonathan Strecker
- , Gagan D. Gupta
- & Daniel Durocher
-
Article |
 Microtubule-driven nuclear rotations promote meiotic chromosome dynamics
Microtubule-driven nuclear rotations promote meiotic chromosome dynamics
Huynh and colleagues discover nuclear rotations driven by centrosomes, microtubules and Dynein in Drosophila germ cells, and find that these movements facilitate the pairing of homologous chromosomes.
- Nicolas Christophorou
- , Thomas Rubin
- & Jean-René Huynh
-
Article |
 Non-canonical NF-κB signalling and ETS1/2 cooperatively drive C250T mutant TERT promoter activation
Non-canonical NF-κB signalling and ETS1/2 cooperatively drive C250T mutant TERT promoter activation
The TERT promoter is mutated in many cancers. Li et al. show that non-canonical NF-κB signalling and ETS1/2 transcription factors are jointly needed to activate the C250T mutant TERT promoter, leading to telomerase activity and glioblastoma growth.
- Yinghui Li
- , Qi-Ling Zhou
- & Vinay Tergaonkar
-
News & Views |
 Condensing chromosome condensation
Condensing chromosome condensation
Mitotic chromosome condensation has fascinated biologists since Flemming's early illustrations of mitosis in the late nineteenth century. Now — 130 years later — chromatid condensation is reconstituted in vitro with the minimum components. The results are remarkably and beautifully simple, requiring only core histones, three histone chaperones, topoisomerase II and condensin I.
- Jason C. Bell
- & Aaron F. Straight
-
Article |
 Reconstitution of mitotic chromatids with a minimum set of purified factors
Reconstitution of mitotic chromatids with a minimum set of purified factors
Hirano and colleagues have reconstituted mitotic chromatin condensation in cell-free extracts using six purified factors, including histone chaperones, topoisomerase II and condensin I.
- Keishi Shintomi
- , Tatsuro S. Takahashi
- & Tatsuya Hirano
-
Letter |
 Kinetochore components are required for central spindle assembly
Kinetochore components are required for central spindle assembly
Using C. elegans zygotes, Dumont and colleagues find that kinetochore proteins, including KNL-1, participate in assembly of central spindle microtubules for cytokinesis.
- Gilliane Maton
- , Frances Edwards
- & Julien Dumont
-
Article |
 The telomere bouquet regulates meiotic centromere assembly
The telomere bouquet regulates meiotic centromere assembly
Studying spindle formation in fission yeast, Cooper and colleagues discover a role for the meiotic telomere bouquet in supporting centromere assembly.
- Michael Klutstein
- , Alex Fennell
- & Julia Promisel Cooper
-
Article |
 Kinetochore–microtubule error correction is driven by differentially regulated interaction modes
Kinetochore–microtubule error correction is driven by differentially regulated interaction modes
Tanaka and colleagues find that aurora B regulates end-on versus lateral kinetochore–microtubule attachments differently to ensure that chromosomes orient correctly for segregation in mitosis.
- Maria Kalantzaki
- , Etsushi Kitamura
- & Tomoyuki U. Tanaka
-
Letter |
 Kinetochore motors drive congression of peripheral polar chromosomes by overcoming random arm-ejection forces
Kinetochore motors drive congression of peripheral polar chromosomes by overcoming random arm-ejection forces
By live imaging and laser microsurgery, Maiato and colleagues characterize how the chromokinesin, dynein and CENP-E motor proteins cooperate to congress chromosomes peripheral to the spindle poles of the metaphase plate in mitosis.
- Marin Barisic
- , Paulo Aguiar
- & Helder Maiato
-
Research Highlights |
Mitotic suppression of repair prevents telomere fusions
- Christina Karlsson Rosenthal
-
Review Article |
 The biogenesis of chromosome translocations
The biogenesis of chromosome translocations
Repair of a chromosome break can result in part of a chromosome attaching to a different chromosome, causing gene deregulation and disease. Roukos and Misteli discuss the spatial aspect of chromosome translocation and the role of DNA repair pathways in this process.
- Vassilis Roukos
- & Tom Misteli
-
Article |
 The TRF1-binding protein TERB1 promotes chromosome movement and telomere rigidity in meiosis
The TRF1-binding protein TERB1 promotes chromosome movement and telomere rigidity in meiosis
Meiotic chromosome movement is needed for homologue pairing and synapsis. Watanabe and colleagues identify TERB1 as a protein needed for telomere mobility and attachment to the nuclear envelope, and for telomere enrichment of meiotic cohesin.
- Hiroki Shibuya
- , Kei-ichiro Ishiguro
- & Yoshinori Watanabe
-
News & Views |
 Swapping CENP-A at the centromere
Swapping CENP-A at the centromere
Faithful genome segregation depends on the functions of the eukaryotic centromere, which is characterized by the histone variant CENP-A. Gene replacement in human cells and fission yeast has now been used to show how CENP-A biochemically encodes centromere identity, as well as reveal an unexpected role for CENP-B in centromere function.
- Bradley T. French
- & Aaron F. Straight
-
Article |
 A two-step mechanism for epigenetic specification of centromere identity and function
A two-step mechanism for epigenetic specification of centromere identity and function
The centromere-specific histone H3 variant CENP-A is sufficient for centromere specification in many species. Cleveland and colleagues have used an elegant gene targeting strategy to define a two-step mechanism for how CENP-A acts in centromere targeting and kinetochore assembly and function.
- Daniele Fachinetti
- , H. Diego Folco
- & Don W. Cleveland
-
Article |
 TRF2 inhibits a cell-extrinsic pathway through which natural killer cells eliminate cancer cells
TRF2 inhibits a cell-extrinsic pathway through which natural killer cells eliminate cancer cells
Gilson and colleagues report that increased expression of the telomeric protein TRF2 in tumour cells promotes tumorigenesis in a non-cell-autonomous manner, by inhibiting the recruitment and activation of natural killer cells at the tumour site.
- Annamaria Biroccio
- , Julien Cherfils-Vicini
- & Eric Gilson
-
Article |
 Microtubules and Alp7–Alp14 (TACC–TOG) reposition chromosomes before meiotic segregation
Microtubules and Alp7–Alp14 (TACC–TOG) reposition chromosomes before meiotic segregation
Chromosome segregation requires the capture of kinetochores by microtubules, a process that in yeast mitosis is facilitated by kinetochores being tethered to spindle poles. Sato and colleagues find that in meiosis I, when kinetochores are not tethered to poles, a microtubule array associated with TACC (Alp7) and TOG proteins (Alp14 and Dis1) retrieves kinetochores in a Polo-kinase-dependent manner.
- Yasutaka Kakui
- , Masamitsu Sato
- & Masayuki Yamamoto
-
Article |
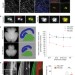 Centrosome amplification causes microcephaly
Centrosome amplification causes microcephaly
Centrosome amplification is often seen in cancer cells and may cause aneuploidy. Basto and colleagues unexpectedly find that centrosome amplification (induced by Plk4 overexpression) in the mouse central nervous system causes microcephaly due to depletion of the neural stem cell pool through aneuploidy and cell death.
- Véronique Marthiens
- , Maria A. Rujano
- & Renata Basto
-
-
Article |
 Telomerase directly regulates NF-κB-dependent transcription
Telomerase directly regulates NF-κB-dependent transcription
Both telomerase activity and NF-κB-driven inflammation occur in tumours, and NF-κB is known to upregulate telomerase levels. Tergaonkar and colleagues now find evidence for a reciprocal direct regulation of NF-κB-dependent gene transcription by telomerase, through an interaction between telomerase and the NF-κB p65 subunit.
- Arkasubhra Ghosh
- , Gaye Saginc
- & Vinay Tergaonkar
-
-
News & Views |
 T time for point centromeres
T time for point centromeres
The diverse nature of eukaryotic centromere structure has led to a prevailing view that the kinetochore–chromatin interface is fundamentally different in distinct species. Two studies now challenge this dogma with the identification of budding yeast homologues of the vertebrate centromere DNA-binding proteins CENP-T and CENP-W.
- Karen E. Gascoigne
- & Iain M. Cheeseman
-
Article |
 CENP-T proteins are conserved centromere receptors of the Ndc80 complex
CENP-T proteins are conserved centromere receptors of the Ndc80 complex
Multi-protein kinetochore complexes bind to the centromeric region of chromosomes to ensure accurate spindle attachment and chromosome segregation, although centromere organization differs widely between species. Westermann and colleagues now identify the budding yeast protein Cnn1 as the orthologue of mammalian CENP-T. They show that it binds to the Ndc80 kinetochore complex and functions in chromosome segregation, illustrating a conserved role for this protein.
- Alexander Schleiffer
- , Michael Maier
- & Stefan Westermann
-
Article |
 Increased chromosome mobility facilitates homology search during recombination
Increased chromosome mobility facilitates homology search during recombination
Repair of double-strand breaks (DSBs) by homologous recombination is thought to involve the movement of damaged chromosomes to facilitate pairing of homologues. Rothstein and colleagues have now followed the movement of damaged loci in diploid yeast by time-lapse microscopy, revealing the dynamics of damage-induced movement and the requirement for repair proteins in this process.
- Judith Miné-Hattab
- & Rodney Rothstein
-
News & Views |
 Signalling the end of the line
Signalling the end of the line
Cellular senescence is a stable proliferation arrest induced by triggers such as short telomeres, activated oncogenes and genotoxic stress. Two studies show that cellular senescence induced by genotoxic stress depends on chronic DNA-damage signalling from irreparable damage to telomeres. Hence, dysfunctional or damaged telomeres are the initiators of multiple modes of senescence.
- John van Tuyn
- & Peter D. Adams
-
Article |
 Telomeric DNA damage is irreparable and causes persistent DNA-damage-response activation
Telomeric DNA damage is irreparable and causes persistent DNA-damage-response activation
D’Adda di Fagagna and colleagues observe that, after genotoxic treatment of cells and mice, unrepaired DNA-damage foci and DNA-damage signalling persist at telomeres. They show that introducing the telomeric protein TRF2 near a double-strand break elsewhere on the chromosomes prevents repair. Unrepaired foci are also observed at telomeres of ageing animals, suggesting a role for TRF2 in senescence establishment.
- Marzia Fumagalli
- , Francesca Rossiello
- & Fabrizio d’Adda di Fagagna
-
-
Letter |
 DNA-damage response and repair activities at uncapped telomeres depend on RNF8
DNA-damage response and repair activities at uncapped telomeres depend on RNF8
The E3 ligase RNF8 is recruited to double-strand breaks in DNA to promote repair. Jacobs and colleagues discovered that RNF8 also goes to unprotected telomeres, where it mediates non-homologous end-joining of chromosome ends and contributes to telomere-induced genomic instability.
- Marieke H. Peuscher
- & Jacqueline J. L. Jacobs
-
Letter |
 The PIAS homologue Siz2 regulates perinuclear telomere position and telomerase activity in budding yeast
The PIAS homologue Siz2 regulates perinuclear telomere position and telomerase activity in budding yeast
Budding yeast telomeres are anchored to the nuclear envelope. Siz2-mediated SUMOylation of Yku70/80 and Sir4, both known to mediate telomere anchoring, is found to promote telomere localisation at the nuclear envelope and restrain their elongation.
- Helder C. Ferreira
- , Brian Luke
- & Susan M. Gasser
-
News & Views |
 A GTPase switch maintains CENP-A at centromeric chromatin
A GTPase switch maintains CENP-A at centromeric chromatin
The histone H3 variant CENP-A defines centromeric chromatin, but it has been unclear how CENP-A is stably maintained at centromeres. It has now been shown that the CENP-A licensing factor HsKNL2 and the small GTPases activating protein MgcRacGAP cooperate to promote the stability of newly loaded CENP-A at centromeres.
- Lisa Prendergast
- & Kevin F. Sullivan
-
Article |
 A small GTPase molecular switch regulates epigenetic centromere maintenance by stabilizing newly incorporated CENP-A
A small GTPase molecular switch regulates epigenetic centromere maintenance by stabilizing newly incorporated CENP-A
The histone H3 variant CENP-A defines centromeric chromatin, but it is not known how it is maintained at these loci. The CENP-A licensing factor HsKNL2, the small GTPases Cdc42 and Rac, and their regulators Ect2 and MgcRacGAP, coordinately promote the stability of newly loaded CENP-A at centromeres.
- Anaïck Lagana
- , Jonas F. Dorn
- & Paul S. Maddox
-
Article |
 Positive feedback between p53 and TRF2 during telomere-damage signalling and cellular senescence
Positive feedback between p53 and TRF2 during telomere-damage signalling and cellular senescence
Uncapped telomeres activate a p53-mediated DNA damage response to elicit cellular senescence. In turn, p53 negatively modulates telomere capping by promoting ubiquitin-mediated degradation of the TRF2 shelterin component.
- Kaori Fujita
- , Izumi Horikawa
- & Curtis C. Harris
-
Perspective |
 Centromere tension: a divisive issue
Centromere tension: a divisive issue
It has been proposed that the spindle assembly checkpoint detects both unattached kinetochores and lack of tension between sister kinetochores when sister chromatids are not attached to opposite spindle poles. However, here we argue that there is only one signal — whether kinetochores are attached to microtubules or not — and this has implications for our understanding of both chromosome segregation and the control of genomic stability.
- Alexey Khodjakov
- & Jonathon Pines
-
News & Views |
 Meiotic kinetochores get pushed aside by a CLS act
Meiotic kinetochores get pushed aside by a CLS act
Kinetochores link microtubules to DNA and provide force critical for chromosome segregation in mitosis. New data show that kinetochores are not necessary for acentrosomal meiotic chromosome segregation in Caenorhabditis elegans. Instead, CLS-2 (CLASP) generates a mid-zone bundle of microtubules that are suggested to act in pushing the chromosomes apart.
- Xue Han
- & Martin Srayko
-
Letter |
 A kinetochore-independent mechanism drives anaphase chromosome separation during acentrosomal meiosis
A kinetochore-independent mechanism drives anaphase chromosome separation during acentrosomal meiosis
How acentrosomal meiotic spindles separate chromosomes in anaphase has been unclear. Although kinetochores are required for the bi-orientation of chromosomes, chromosome separation may instead be driven by CLASP/CLS-2 mediated microtubule assembly.
- Julien Dumont
- , Karen Oegema
- & Arshad Desai

 Adding a transcription-coupled repair pathway
Adding a transcription-coupled repair pathway