Featured
-
-
News & Views |
 CENP-T bears the load in mitosis
CENP-T bears the load in mitosis
During mitosis, the kinetochore connects chromosomes to spindle microtubules and enables chromosome segregation. A genetic study in vertebrate cells demonstrates phosphorylation-regulated plasticity of kinetochore assembly and highlights the role of the centromere protein T in load-bearing kinetochore–microtubule attachment.
- Yang Yang
- & Hongtao Yu
-
Article |
 Multiple phosphorylations control recruitment of the KMN network onto kinetochores
Multiple phosphorylations control recruitment of the KMN network onto kinetochores
Hara et al. find that the recruitment of the KMN kinetochore protein network is mainly dependent on the CENP-T pathway and promoted by Cdk1-mediated phosphorylation of CENP-T in chicken DT40 cells.
- Masatoshi Hara
- , Mariko Ariyoshi
- & Tatsuo Fukagawa
-
Article |
 Dynamic kinetochore size regulation promotes microtubule capture and chromosome biorientation in mitosis
Dynamic kinetochore size regulation promotes microtubule capture and chromosome biorientation in mitosis
Sacristan et al. show that the dynein adaptor Spindly facilitates oligomerisation of the RZZ complex to expand the kinetochore, after which Spindly-associated dynein compacts the kinetochore to allow for faithful chromosome segregation.
- Carlos Sacristan
- , Misbha Ud Din Ahmad
- & Geert JPL Kops
-
Article |
 How the kinetochore couples microtubule force and centromere stretch to move chromosomes
How the kinetochore couples microtubule force and centromere stretch to move chromosomes
Using mathematical simulations and a FRET tension sensor inserted into the microtubule-binding complex Ndc80, Suzuki and colleagues obtain insights into how force is generated at the budding yeast kinetochore.
- Aussie Suzuki
- , Benjamin L. Badger
- & Kerry Bloom
-
Article |
 Adaptive changes in the kinetochore architecture facilitate proper spindle assembly
Adaptive changes in the kinetochore architecture facilitate proper spindle assembly
Using light- and electron microscopy, Khodjakov, Mogilner and colleagues find that the shape of the kinetochore undergoes dramatic changes during mitosis. Computational analysis suggests that this facilitates correct chromosome attachment.
- Valentin Magidson
- , Raja Paul
- & Alexey Khodjakov
-
Article |
 The kinetochore encodes a mechanical switch to disrupt spindle assembly checkpoint signalling
The kinetochore encodes a mechanical switch to disrupt spindle assembly checkpoint signalling
Joglekar and colleagues show that in budding yeast, kinetochore–microtubule attachment mediates silencing of the spindle assembly checkpoint through physical separation of Mps1 kinase from its substrate Spc105.
- Pavithra Aravamudhan
- , Alan A. Goldfarb
- & Ajit P. Joglekar
-
Letter |
 Kinetochore components are required for central spindle assembly
Kinetochore components are required for central spindle assembly
Using C. elegans zygotes, Dumont and colleagues find that kinetochore proteins, including KNL-1, participate in assembly of central spindle microtubules for cytokinesis.
- Gilliane Maton
- , Frances Edwards
- & Julien Dumont
-
Article |
 Kinetochore–microtubule error correction is driven by differentially regulated interaction modes
Kinetochore–microtubule error correction is driven by differentially regulated interaction modes
Tanaka and colleagues find that aurora B regulates end-on versus lateral kinetochore–microtubule attachments differently to ensure that chromosomes orient correctly for segregation in mitosis.
- Maria Kalantzaki
- , Etsushi Kitamura
- & Tomoyuki U. Tanaka
-
Letter |
 Negative feedback at kinetochores underlies a responsive spindle checkpoint signal
Negative feedback at kinetochores underlies a responsive spindle checkpoint signal
Saurin, Kops and colleagues suggest that rapid spindle assembly checkpoint (SAC) responsiveness is mediated by a mechanism in which active SAC recruits PP2A, leading to PP1 recruitment, which in turn displaces PP2A and shuts off the SAC.
- Wilco Nijenhuis
- , Giulia Vallardi
- & Adrian T. Saurin
-
Letter |
 Kinetochore motors drive congression of peripheral polar chromosomes by overcoming random arm-ejection forces
Kinetochore motors drive congression of peripheral polar chromosomes by overcoming random arm-ejection forces
By live imaging and laser microsurgery, Maiato and colleagues characterize how the chromokinesin, dynein and CENP-E motor proteins cooperate to congress chromosomes peripheral to the spindle poles of the metaphase plate in mitosis.
- Marin Barisic
- , Paulo Aguiar
- & Helder Maiato
-
News & Views |
 Swapping CENP-A at the centromere
Swapping CENP-A at the centromere
Faithful genome segregation depends on the functions of the eukaryotic centromere, which is characterized by the histone variant CENP-A. Gene replacement in human cells and fission yeast has now been used to show how CENP-A biochemically encodes centromere identity, as well as reveal an unexpected role for CENP-B in centromere function.
- Bradley T. French
- & Aaron F. Straight
-
Article |
 A two-step mechanism for epigenetic specification of centromere identity and function
A two-step mechanism for epigenetic specification of centromere identity and function
The centromere-specific histone H3 variant CENP-A is sufficient for centromere specification in many species. Cleveland and colleagues have used an elegant gene targeting strategy to define a two-step mechanism for how CENP-A acts in centromere targeting and kinetochore assembly and function.
- Daniele Fachinetti
- , H. Diego Folco
- & Don W. Cleveland
-
Article |
 Microtubules and Alp7–Alp14 (TACC–TOG) reposition chromosomes before meiotic segregation
Microtubules and Alp7–Alp14 (TACC–TOG) reposition chromosomes before meiotic segregation
Chromosome segregation requires the capture of kinetochores by microtubules, a process that in yeast mitosis is facilitated by kinetochores being tethered to spindle poles. Sato and colleagues find that in meiosis I, when kinetochores are not tethered to poles, a microtubule array associated with TACC (Alp7) and TOG proteins (Alp14 and Dis1) retrieves kinetochores in a Polo-kinase-dependent manner.
- Yasutaka Kakui
- , Masamitsu Sato
- & Masayuki Yamamoto
-
News & Views |
 Cullin' PLK1 from kinetochores
Cullin' PLK1 from kinetochores
To ensure proper attachment of all chromosomes to the spindle, PLK1 has to associate with kinetochores during prometaphase and must be released from these sites before sister chromatid separation can begin. The monoubiquitylation of PLK1 by the ubiquitin ligase CUL3–KLHL22 is now identified as a critical step in promoting the release of PLK1 from kinetochores, pushing non-proteolytic ubiquitylation into the limelight of cell division research.
- Colleen A. McGourty
- & Michael Rape
-
Letter |
 Pivoting of microtubules around the spindle pole accelerates kinetochore capture
Pivoting of microtubules around the spindle pole accelerates kinetochore capture
To segregate chromosomes, spindle microtubules must attach to chromosomes through kinetochores, in a process involving several types of microtubule behaviour. Tolic-Norrelykke and colleagues find that fission yeast microtubules rapidly rotate around the spindle poles, and mathematical modelling confirms that this random microtubule movement facilitates kinetochore capture.
- Iana Kalinina
- , Amitabha Nandi
- & Iva M. Tolić-Nørrelykke
-
Letter |
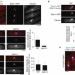 MPS1/Mph1 phosphorylates the kinetochore protein KNL1/Spc7 to recruit SAC components
MPS1/Mph1 phosphorylates the kinetochore protein KNL1/Spc7 to recruit SAC components
The kinase MPS1 is a conserved and essential component of the spindle assembly checkpoint (SAC), but its relevant substrate in this context has remained uncertain. Watanabe and colleagues now show that, in fission yeast and human cells, MPS1 (Mph1 in fission yeast) phosphorylates the kinetochore protein KNL1 (Spc7), leading to kinetochore recruitment of BUB1, an event required for SAC activation.
- Yuya Yamagishi
- , Ching-Hui Yang
- & Yoshinori Watanabe
-
News & Views |
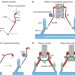 Cdt1 throws kinetochore–microtubule attachments for a loop
Cdt1 throws kinetochore–microtubule attachments for a loop
The Ndc80 complex links spindle microtubules to the kinetochore to ensure the proper segregation of chromosomes during mitosis. Analysis of the replication licensing factor Cdt1 during mitosis now reveals a cooperative role with the Ndc80 complex in establishing stable microtubule attachments to the spindle.
- Daniel R. Matson
- & P. Todd Stukenberg
-
Article |
 Cnn1 inhibits the interactions between the KMN complexes of the yeast kinetochore
Cnn1 inhibits the interactions between the KMN complexes of the yeast kinetochore
The kinetochore is a multiprotein complex that tethers chromosomes to the mitotic spindle for accurate chromosome segregation. De Wulf and colleagues now show in budding yeast that the protein Cnn1 functions at the kinetochore and is recruited to the inner kinetochore, in a manner dependent on its phosphorylation mediated by the Cdc28, Mps1 and Ipl1 kinases.
- Lucy J. Bock
- , Cinzia Pagliuca
- & Peter De Wulf
-
News & Views |
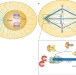 A new cap for kinetochore fibre minus ends
A new cap for kinetochore fibre minus ends
In mitotic spindles, each sister chromatid is directly attached to a spindle pole through microtubule bundles known as kinetochore fibres. Microspherule protein 1 (MCRS1) is now shown to support spindle assembly by localizing to the minus ends of kinetochore fibres and protecting them from depolymerization.
- Sabine Petry
- & Ronald D. Vale
-
Article |
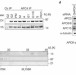 APC15 drives the turnover of MCC-CDC20 to make the spindle assembly checkpoint responsive to kinetochore attachment
APC15 drives the turnover of MCC-CDC20 to make the spindle assembly checkpoint responsive to kinetochore attachment
The spindle assembly checkpoint halts cell-cycle progression in the presence of unattached kinetochores by preventing activation of APC/C. Pines and colleagues find that APC15 has a critical role in regulating APC/C activation by promoting release of the inhibitory MCC complex from APC/C once the spindle assembly checkpoint is satisfied.
- Jörg Mansfeld
- , Philippe Collin
- & Jonathon Pines
-
Letter |
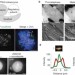 Formation of stable attachments between kinetochores and microtubules depends on the B56-PP2A phosphatase
Formation of stable attachments between kinetochores and microtubules depends on the B56-PP2A phosphatase
The microtubules that attach kinetochores to chromosomes (K-fibres) are stabilized in prometaphase to allow for accurate chromosome segregation. Kapoor and colleagues find that the B56-PP2A phosphatase stabilizes K-fibres potentially by counteracting the phosphorylation of kinetochore substrates that is mediated by Aurora B and Plk1.
- Emily A. Foley
- , Maria Maldonado
- & Tarun M. Kapoor
-
Article |
 Heterochromatin boundaries are hotspots for de novo kinetochore formation
Heterochromatin boundaries are hotspots for de novo kinetochore formation
In most eukaryotes, the centromere is defined by epigenetic marks such as the histone H3 variant CENH3/CENP-A/CID. Ectopic induction of kinetochores in Drosophila S2 cells by CID overexpression leads to kinetochore assembly specifically in silent intergenic regions bordering heterochomratin, demonstrating a role for these domains in centromere identity.
- Agata M. Olszak
- , Dominic van Essen
- & Patrick Heun
-
Letter |
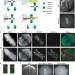 Constitutive Mad1 targeting to kinetochores uncouples checkpoint signalling from chromosome biorientation
Constitutive Mad1 targeting to kinetochores uncouples checkpoint signalling from chromosome biorientation
Targeting Mad1 to kinetochores reveals that Mad1 localization is sufficient to induce mitotic-checkpoint-dependent arrest, and that Aurora B, Mps1 and BubR1 are required for checkpoint maintenance.
- Maria Maldonado
- & Tarun M. Kapoor
-
News & Views |
 Meiotic kinetochores get pushed aside by a CLS act
Meiotic kinetochores get pushed aside by a CLS act
Kinetochores link microtubules to DNA and provide force critical for chromosome segregation in mitosis. New data show that kinetochores are not necessary for acentrosomal meiotic chromosome segregation in Caenorhabditis elegans. Instead, CLS-2 (CLASP) generates a mid-zone bundle of microtubules that are suggested to act in pushing the chromosomes apart.
- Xue Han
- & Martin Srayko
-
Letter |
 A kinetochore-independent mechanism drives anaphase chromosome separation during acentrosomal meiosis
A kinetochore-independent mechanism drives anaphase chromosome separation during acentrosomal meiosis
How acentrosomal meiotic spindles separate chromosomes in anaphase has been unclear. Although kinetochores are required for the bi-orientation of chromosomes, chromosome separation may instead be driven by CLASP/CLS-2 mediated microtubule assembly.
- Julien Dumont
- , Karen Oegema
- & Arshad Desai
-
Article |
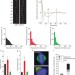 Molecular control of kinetochore-microtubule dynamics and chromosome oscillations
Molecular control of kinetochore-microtubule dynamics and chromosome oscillations
The core kinetochore protein CENP-H is shown to be required for the organization of the metaphase plate. CENP-H regulates microtubule plus-end dynamics, and chromosome oscillations, which are shown to be essential for chromosome congression.
- Ana C. Amaro
- , Catarina P. Samora
- & Patrick Meraldi
-
Letter |
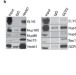 The Nup107-160 complex and γ-TuRC regulate microtubule polymerization at kinetochores
The Nup107-160 complex and γ-TuRC regulate microtubule polymerization at kinetochores
The γ-tubulin ring complex (γ-TuRC) nucleates microtubules. The nuclear pore subcomplex Nup107-160 is found to interact and cooperate with γ-TuRC to nucleate microtubules at kinetochores, thereby promoting spindle assembly.
- Ram Kumar Mishra
- , Papia Chakraborty
- & Mary Dasso

 Higher-order protein assembly controls kinetochore formation
Higher-order protein assembly controls kinetochore formation