Featured
-
-
News & Views |
 Mapping protein–protein contacts
Mapping protein–protein contacts
Obtaining detailed structural information about the interactions between amyloid-forming proteins and inhibitors can be extremely difficult. Two-dimensional infrared spectroscopy has now risen to this challenge to show the mapping of protein–protein contact sites in real time.
- Minhaeng Cho
-
-
Article |
 Protein fold determined by paramagnetic magic-angle spinning solid-state NMR spectroscopy
Protein fold determined by paramagnetic magic-angle spinning solid-state NMR spectroscopy
Despite recent progress, solving protein structures using solid-state NMR spectroscopy is not routine. Now, a method for the rapid determination of global protein fold is reported, based on measurements of 15N longitudinal paramagnetic relaxation enhancements in several protein variants modified with covalently attached cysteine–EDTA–Cu2+ tags.
- Ishita Sengupta
- , Philippe S. Nadaud
- & Christopher P. Jaroniec
-
Article |
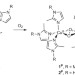 Self-assembly of the oxy-tyrosinase core and the fundamental components of phenolic hydroxylation
Self-assembly of the oxy-tyrosinase core and the fundamental components of phenolic hydroxylation
A functional active-site mimic of the oxy-tyrosinase enzyme forms through self-assembly of monodentate imidazole ligands, copper(I) and oxygen at −125 °C. The fidelity of this copper–dioxygen complex to the native enzyme, its inherent stability and hydroxylation reactivity suggest that an organizational role of the protein matrix suffices to realize function.
- Cooper Citek
- , Christopher T. Lyons
- & T. Daniel P. Stack
-
-
News & Views |
 Lighting up with azobenzenes
Lighting up with azobenzenes
The chemical introduction of a photoswitchable ligand into ion channel structures should make it possible to study the diverse roles of neurotransmitters and receptors in the brain.
- G. Andrew Woolley
-
Article |
 Optimizing the specificity of nucleic acid hybridization
Optimizing the specificity of nucleic acid hybridization
High-fidelity pairing of nucleic acid polymers is important in the development of sensors and for the application of DNA nanotechnology. Here, a set of hybridization probes is described that discriminates single-base changes with high specificity. The probes function robustly across many different temperatures, salinities and nucleic acid concentrations.
- David Yu Zhang
- , Sherry Xi Chen
- & Peng Yin
-
Article |
 Darwinian evolution of an alternative genetic system provides support for TNA as an RNA progenitor
Darwinian evolution of an alternative genetic system provides support for TNA as an RNA progenitor
The pre-RNA-world hypothesis postulates that RNA was preceded in the evolution of life by a simpler genetic material. Here, Darwinian evolution methods were used to generate a threose nucleic acid (TNA) aptamer. This result provides evidence that TNA could have served as an ancestral genetic system during an early stage of life.
- Hanyang Yu
- , Su Zhang
- & John C. Chaput
-
Article |
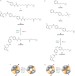 Optochemical control of genetically engineered neuronal nicotinic acetylcholine receptors
Optochemical control of genetically engineered neuronal nicotinic acetylcholine receptors
Light-sensitive ligands can be used to regulate neurobiological receptors with high spatiotemporal precision. Here, the optochemical control of neuronal nicotinic acetylcholine receptors, using both photoswitchable tethered agonists and antagonists, is described. These rationally designed hybrid photoreceptors will facilitate the investigation of the physiological and pathological functions of nicotinic receptors in the brain.
- Ivan Tochitsky
- , Matthew R. Banghart
- & Dirk Trauner
-
News & Views |
 Adding a second dimension
Adding a second dimension
Mutating RNA one nucleotide at a time and measuring the impact of this on its chemical reactivity provides a strategy for determining its three-dimensional structure, and from there, hopefully, its function.
- Katja Petzold
- & Hashim M. Al-Hashimi
-
Article |
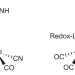 Combining acid–base, redox and substrate binding functionalities to give a complete model for the [FeFe]-hydrogenase
Combining acid–base, redox and substrate binding functionalities to give a complete model for the [FeFe]-hydrogenase
The fastest catalysts in nature for producing and oxidizing hydrogen are [FeFe]-hydrogenases, which make use of an extra one-electron redox equivalent from an iron-sulfur cluster that is outside the core. Now, a ferrocene-based ligand that oxidizes at mild potential performs this cluster's role in an excellent synthetic hydrogenase model.
- James M. Camara
- & Thomas B. Rauchfuss
-
Article |
 A two-dimensional mutate-and-map strategy for non-coding RNA structure
A two-dimensional mutate-and-map strategy for non-coding RNA structure
Non-coding RNAs are ubiquitous biomolecules with intricate three-dimensional folds that are difficult to characterize. This Article presents an information-rich strategy for inferring RNA structure by combining nucleotide-by-nucleotide mutagenesis with single-nucleotide-resolution chemical mapping.
- Wipapat Kladwang
- , Christopher C. VanLang
- & Rhiju Das
-
News & Views |
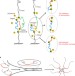 Breaking out of the cage
Breaking out of the cage
Activating caged reactive sites in proteins using mechanical force provides a powerful approach in the study of chemical reactions, and provides greater insight into which reactions are possible and their rates.
- Chia-Ching Chou
- & Markus J. Buehler
-
Article |
 Direct observation of disulfide isomerization in a single protein
Direct observation of disulfide isomerization in a single protein
Multiple redox reaction pathways exist in proteins containing several cysteines. A technique termed mechanical uncaging is now demonstrated, allowing the release of a single reactive cysteine within a protein and the unequivocal observation of subsequent thiol/disulfide exchanges. Mechanical uncaging of reactive groups is useful for studying chemical kinetics in a synchronized manner.
- Jorge Alegre-Cebollada
- , Pallav Kosuri
- & Julio M. Fernández
-
News & Views |
 Untying a nanoscale knot
Untying a nanoscale knot
Mechanical unfolding of a single DNA G-quadruplex structure with and without a stabilizing ligand can be used to calculate the binding strength of the ligand and could help to identify drugs to target these important biological assemblies.
- Micah J. McCauley
- & Mark C. Williams
-
News & Views |
 Spinning into focus
Spinning into focus
Transient sedimentation of proteins inside a solid-state NMR rotor under fast magic-angle spinning offers a promising solution to the challenge of determining the structures of high-molecular-weight proteins with atomic resolution. This opens new opportunities for structural analysis of large macromolecules and macromolecular assemblies.
- Tatyana Polenova
-
Article |
 Reversible bond formation enables the replication and amplification of a crosslinking salen complex as an orthogonal base pair
Reversible bond formation enables the replication and amplification of a crosslinking salen complex as an orthogonal base pair
Adding one further base pair to the classic Watson–Crick scheme not only expands the genetic code but also offers opportunities to modify the structure and function of DNA. It has now been shown that an artificial metal–salen base pair can be enzymatically incorporated into DNA duplexes and even amplified by PCR.
- Corinna Kaul
- , Markus Müller
- & Thomas Carell
-
News & Views |
 The many faces of a proton
The many faces of a proton
A joint X-ray/neutron diffraction study has enabled the direct observation of a hydronium ion coordinated by three amino-acid residues of an enzyme. This sighting will affect our views on how enzymes transport and use protons.
- Victor L. Davidson
-
Article |
 Regio- and stereoselectivity of P450-catalysed hydroxylation of steroids controlled by laboratory evolution
Regio- and stereoselectivity of P450-catalysed hydroxylation of steroids controlled by laboratory evolution
Selective reaction of one C–H bond among many in complex organic molecules is a grand challenge for organic chemistry. Here, starting from an enzyme that oxidizes two positions in a steroid without bias, laboratory evolution is used to prepare mutants that can regio- and stereoselectively oxidize either position.
- Sabrina Kille
- , Felipe E. Zilly
- & Manfred T. Reetz
-
Article |
 A route to enantiopure RNA precursors from nearly racemic starting materials
A route to enantiopure RNA precursors from nearly racemic starting materials
A drawback of recently reported prebiotic routes to RNA is a requirement for enantioenriched reactants. Here, the presence of a slightly enantioenriched amino acid in the reaction mixture is shown to drive the formation of enantiopure RNA precursors. This provides a plausible scenario in which single-handed biological molecules were formed prior to the emergence of self-replicating informational polymers.
- Jason E. Hein
- , Eric Tse
- & Donna G. Blackmond
-
Article |
 Efficient enzyme-free copying of all four nucleobases templated by immobilized RNA
Efficient enzyme-free copying of all four nucleobases templated by immobilized RNA
A long-standing problem with the RNA-world hypothesis is that enzyme-free replication of RNA strands has not been demonstrated. Here, immobilization of a template strand and periodic replacement of a solution containing activated nucleotides allows the copying of unmodified RNA sequences containing any of the four natural nucleobases in near-quantitative yield.
- Christopher Deck
- , Mario Jauker
- & Clemens Richert
-
Review Article |
 Contemporary strategies for peptide macrocyclization
Contemporary strategies for peptide macrocyclization
Peptide macrocycles have a number of important applications. Among other things, the reduced conformational freedom of the cyclic structure enables strong binding to the extended contact regions of protein–protein complexes. Here, emerging methods directed towards the synthesis of these valuable molecules are reviewed.
- Christopher J. White
- & Andrei K. Yudin
-
Article |
 Water-oxidation catalysis by manganese in a geochemical-like cycle
Water-oxidation catalysis by manganese in a geochemical-like cycle
The development of an effective, cheap and robust water oxidation catalyst is crucial to the development of solar water-splitting technology. A manganese catalyst inspired by the water oxidizing centre of photosystem II is now shown to dissociate and reform into nanoparticles resembling the known natural oxidant mineral birnessite.
- Rosalie K. Hocking
- , Robin Brimblecombe
- & Leone Spiccia
-
Article |
 Platinum nanocrystals selectively shaped using facet-specific peptide sequences
Platinum nanocrystals selectively shaped using facet-specific peptide sequences
Crystal growth in nature is controlled by biomolecules to produce precisely engineered crystal shapes. Now, facet-specific peptide sequences that have been rationally selected through a biomimetic evolution process are used as regulating agents for predictable synthesis of platinum nanocrystals with selectively exposed crystal surfaces.
- Chin-Yi Chiu
- , Yujing Li
- & Yu Huang
-
Article |
 Stereoselective C–C bond formation catalysed by engineered carboxymethylproline synthases
Stereoselective C–C bond formation catalysed by engineered carboxymethylproline synthases
The reaction of enols and enolates with electrophiles is used extensively in synthesis. Here, protein engineering — substituting amino acid residues in an enzyme active site — is used to produce biocatalysts for the control of enolate chemistry. The adapted enzymes enable stereoselective C–C bond formation yielding N-heterocycles in high diastereomeric excess by the reaction of trisubstituted-enolates.
- Refaat B. Hamed
- , J. Ruben Gomez-Castellanos
- & Christopher J. Schofield
-
Article |
 DNA charge transport over 34 nm
DNA charge transport over 34 nm
The potential for using molecules as wires in nanoscale electronics is somewhat tempered by the challenges in making long and uniform structures. Now, it has been shown that DNA — which is easily synthesized to precise lengths — can conduct charge over 34 nm on multiplexed gold electrodes, a distance that surpasses most reports of molecular wires.
- Jason D. Slinker
- , Natalie B. Muren
- & Jacqueline K. Barton
-
Article |
 Ion mobility–mass spectrometry reveals a conformational conversion from random assembly to β-sheet in amyloid fibril formation
Ion mobility–mass spectrometry reveals a conformational conversion from random assembly to β-sheet in amyloid fibril formation
Amyloid cascades leading to peptide β-sheet fibrils are central to many diseases. Intermediate assemblies were recently identified as the toxic agents, but obtaining structural details of these early oligomers has largely been unsuccessful with traditional techniques. Here, ion mobility methods provide evidence for structural transitions from random to β-sheet assembly.
- Christian Bleiholder
- , Nicholas F. Dupuis
- & Michael T. Bowers
-
Article |
 Supramolecular fishing for plasma membrane proteins using an ultrastable synthetic host–guest binding pair
Supramolecular fishing for plasma membrane proteins using an ultrastable synthetic host–guest binding pair
Characterization of plasma membrane proteins is important in understanding fundamental biological processes and developing new drugs, but their separation remains a challenge. Now, a synthetic receptor–ligand pair based on ferrocene derivatives and cucurbit[7]uril is shown to have exceptionally high binding affinity, and enables membrane proteins to be isolated efficiently without any contamination from naturally biotinylated molecules.
- Don-Wook Lee
- , Kyeng Min Park
- & Kimoon Kim
-
Article |
 Interrogating viral capsid assembly with ion mobility–mass spectrometry
Interrogating viral capsid assembly with ion mobility–mass spectrometry
Although most proteins fulfil their role as part of large protein complexes, little is known about the pathways of complex assembly. Here, ion mobility–mass spectrometry is used to monitor and structurally characterize the assembly intermediates of viral protein shells, called capsids, of two major human pathogens, norovirus and hepatitis B virus.
- Charlotte Uetrecht
- , Ioana M. Barbu
- & Albert J. R. Heck
-
Research Highlights |
 Caught in the act
Caught in the act
The isolation of pure quantities of the active intermediate in cytochrome P450 enzymes — known as compound I — has led to this elusive species being characterized in detail.
- Neil Withers
-
News & Views |
 Detection by failure
Detection by failure
Testing for enzymes is important for diagnosing various medical conditions but can be problematic because of the complexity of physiological media such as blood. Now, a method of detecting phospholipases has been developed that neatly couples their concentration with the aggregation of gold nanoparticles.
- Nicholas A. Melosh
-
Article |
 Mineralization of the metre-long biosilica structures of glass sponges is templated on hydroxylated collagen
Mineralization of the metre-long biosilica structures of glass sponges is templated on hydroxylated collagen
Understanding how biosilica forms is crucial to our knowledge of this important biomaterial. Now, the role of collagen as structuring agent for the metre-long spicules of a primitive glass sponge has been revealed and found to have an unusual peptide motif.
- Hermann Ehrlich
- , Rainer Deutzmann
- & Matthew J. Collins
-
Article |
 Crystallographic snapshots of the reaction of aromatic C–H with O2 catalysed by a protein-bound iron complex
Crystallographic snapshots of the reaction of aromatic C–H with O2 catalysed by a protein-bound iron complex
Crystallographically studying chemical reactions is difficult because the intermediates are rarely stable enough to be crystallized. Now, protein crystals have been used to trap four reaction intermediates in an aromatic dihydroxylation, showing that an iron peroxo complex is observed.
- Christine Cavazza
- , Constance Bochot
- & Stéphane Ménage
-
Research Highlights |
 Under a graphene cover
Under a graphene cover
Layers of water adsorbed on a mica surface have been trapped under a graphene sheet and their structure determined by atomic force microscopy.
- Anne Pichon
-
News & Views |
 Redox control of oxoiron(IV)
Redox control of oxoiron(IV)
The oxygen-evolving centre in the protein complex photosystem II involves Ca2+ within a high-valent oxomanganese cluster, but the role of Ca2+ has yet to be clarified. Interestingly, the electron-transfer behaviour of a non-haem oxoiron(IV) complex has now been found to be significantly altered by Lewis acid metal-ion binding to the oxo atom.
- Kenneth D. Karlin
-
Research Highlights |
Nitrogenase branches out
A vanadium-containing nitrogenase enzyme can reduce carbon monoxide to ethane and propane.
- Neil Withers
-
News & Views |
 A new phase for protein chemistry
A new phase for protein chemistry
Proteins are most at home in water, although it has been known for some time that they can remain functional in non-aqueous environments. Researchers have now shown that in solvent-free melts, the oxygen-binding protein myoglobin adopts a near-native structure and retains its biological activity.
- Douglas S. Clark
-
Article |
 Crystal structure of a metal ion-bound oxoiron(IV) complex and implications for biological electron transfer
Crystal structure of a metal ion-bound oxoiron(IV) complex and implications for biological electron transfer
The interactions of metal ions with metaloxo species are crucial in many important biological processes, such as the oxygen-evolving complex (OEC) in photosystem II, but their exact function remains elusive. Now, the binding of metal ions to a non-haem oxoiron complex has been studied and the observed changes to its electron-transfer properties provide insights into the active site of the OEC.
- Shunichi Fukuzumi
- , Yuma Morimoto
- & Wonwoo Nam
-
Article |
 Electrically induced bonding of DNA to gold
Electrically induced bonding of DNA to gold
The assembly and evaluation of molecular structures on surfaces has been boosted by advances in single-molecule techniques. The development of such methods are continued here, showing that double-stranded DNA, bound to the tip of an atomic force microscope, can be deposited on the surface of a gold electrode using an electrical trigger.
- Matthias Erdmann
- , Ralf David
- & Hermann E. Gaub
-
News & Views |
 Anion transport as easy as pi
Anion transport as easy as pi
A systematic study that combines both theory and experiment now provides direct evidence for the existence of anion–π interactions in compounds that facilitate the transport of anions across phospholipid membranes. This study offers new insight into the factors that affect the strength, selectivity and functional relevance of anion–π interactions.
- Jeffery T. Davis
-
News & Views |
 Rewiring nanostructures
Rewiring nanostructures
The cell's dynamic skeleton, a tightly regulated network of protein fibres, continues to provide inspiration for the design of synthetic nanostructures. Genetic engineering has now been used to encode non-biological functionality within these structures.
- Rein V. Ulijn
- & Pier-Francesco Caponi
-
Article |
 Reversible dioxygen binding in solvent-free liquid myoglobin
Reversible dioxygen binding in solvent-free liquid myoglobin
Freeze-drying of aqueous myoglobin–polymer surfactant nanoconjugates affords a water-free solid that melts at room temperature to produce a viscous solventless liquid protein that exhibits near-native secondary structure and reversible dioxygen binding. The results challenge the accepted role of solvent molecules in mediating protein structure and function, and offer new opportunities in protein-based nanoscience and bionanotechnology.
- Adam W. Perriman
- , Alex P. S. Brogan
- & Stephen Mann
-
-
Research Highlights |
 Environmental issues
Environmental issues
A computational investigation into how environmental factors influence phosphoester hydrolysis reveals differences between the mechanisms observed in aqueous solution and within an enzyme active site.
- Gavin Armstrong
-
Article |
 Direct detection of CH/π interactions in proteins
Direct detection of CH/π interactions in proteins
Weakly polar XH/π interactions are thought to be capable of influencing both the structure and function of proteins, but such interactions are usually identified from three-dimensional structural models. Now, using NMR spectroscopy and isotopic labelling, it has been shown that individual methyl/π interactions can be detected directly in proteins by measuring weak scalar couplings between the nuclei involved.
- Michael J. Plevin
- , David L. Bryce
- & Jérôme Boisbouvier
-
Research Highlights |
 Behold the fold
Behold the fold
Folding techniques have been used on printed titanium hydride sheets to create three-dimensional structures.
- Neil Withers
-
Research Highlights |
 Peptides make the difference
Peptides make the difference
Microporous crystals formed by hydrogen-bonded dipeptides show different permeabilities for argon, nitrogen and oxygen.
- Anne Pichon
-
Research Highlights |
 Sticky situation
Sticky situation
The sliding movement of the actin–myosin protein conjugate responsible for contracting muscles can be inhibited using a dendrimer that glues them together.
- Gavin Armstrong
-
Research Highlights |
 X-ray ordering
X-ray ordering
Exposing supramolecular filaments to X-rays results in spontaneous and reversible crystalline ordering.
- Neil Withers
Browse broader subjects
Browse narrower subjects
- Biocatalysis
- Biogeochemistry
- Bioinorganic chemistry
- Biophysical chemistry
- Carbohydrates
- Chemical modification
- Cytokines
- DNA
- Enzyme mechanisms
- Enzymes
- Glycobiology
- Glycomics
- Histocytochemistry
- Hormones
- Immunochemistry
- Ion channels
- Isoenzymes
- Kinases
- Lipidomics
- Lipids
- Metabolomics
- Metals
- Neurochemistry
- Peptides
- Prions
- Proteases
- Protein folding
- Proteins
- Proteolysis
- Proteomics
- RNA
- Structural biology

 Protein camouflage in cytochrome c–calixarene complexes
Protein camouflage in cytochrome c–calixarene complexes Carbon in the middle
Carbon in the middle Protein under glass
Protein under glass