Featured
-
-
Article |
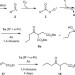 Hydroacylation of α,β-unsaturated esters via aerobic C–H activation
Hydroacylation of α,β-unsaturated esters via aerobic C–H activation
The development of benign methods for carbon–carbon bond formation is a continuing challenge. Here, a simple procedure for the hydroacylation of α,β-unsaturated esters is described, in which auto-oxidation of aldehydes and subsequent acyl radical addition to α,β-unsaturated esters occurs without the need for additional reagents.
- Vijay Chudasama
- , Richard J. Fitzmaurice
- & Stephen Caddick
-
Article |
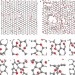 Structural evolution during the reduction of chemically derived graphene oxide
Structural evolution during the reduction of chemically derived graphene oxide
Thermal reduction of graphene oxide is an attractive route towards the preparation of graphene, but complete removal of residual oxygen is problematic. Now, molecular dynamics simulations elucidate the chemical changes involved in this process.
- Akbar Bagri
- , Cecilia Mattevi
- & Vivek B. Shenoy
-
News & Views |
 Forcing a molecule's hand
Forcing a molecule's hand
Ultrasound can be used to control molecular processes as delicate as rotation around a single carbon–carbon bond.
- S. Karthikeyan
- & Rint P. Sijbesma
-
Article |
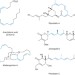 Convergent and stereospecific synthesis of complex skipped polyenes and polyunsaturated fatty acids
Convergent and stereospecific synthesis of complex skipped polyenes and polyunsaturated fatty acids
A convergent route to stereodefined skipped polyenes, which proceeds through the direct union of vinylcyclopropanes with alkynes, is described. Overall, C?C bond formation occurs in concert with the establishment of up to three stereodefined alkenes, through a tandem stereoselective metallacycle-mediated coupling/stereospecific fragmentation sequence.
- Todd K. Macklin
- & Glenn C. Micalizio
-
Article |
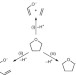 Cleave and capture chemistry illustrated through bimetallic-induced fragmentation of tetrahydrofuran
Cleave and capture chemistry illustrated through bimetallic-induced fragmentation of tetrahydrofuran
Tetrahydrofuran can be unexpectedly cleaved by bimetallic sodium–magnesium and sodium–manganese bases. At least six bonds are broken in the one-pot reaction, but all fragments are separately captured to form crystalline compounds.
- Robert E. Mulvey
- , Victoria L. Blair
- & Luca Russo
-
Article |
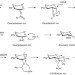 Chiral-auxiliary-mediated 1,2-cis-glycosylations for the solid-supported synthesis of a biologically important branched α-glucan
Chiral-auxiliary-mediated 1,2-cis-glycosylations for the solid-supported synthesis of a biologically important branched α-glucan
The difficulty in the stereoselective introduction of 1,2-cis-glycosides is a major stumbling block in the solid-supported synthesis of oligosaccharides. Now, this has been achieved for a biologically important glucoside containing multiple 1,2-cis-glycosidic linkages with complete anomeric control by using glycosyl donors having a participating (S)-(phenylthiomethyl)benzyl chiral auxiliary at C-2.
- Thomas J. Boltje
- , Jin-Hwan Kim
- & Geert-Jan Boons
-
Research Highlights |
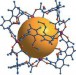 Hand-picked guests
Hand-picked guests
Metal–organic cages made with 1,1-biphenyl ligands act as hosts for enantioselective absorption.
- Neil Withers
-
Research Highlights |
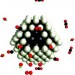 Poison control
Poison control
A combined theoretical and experimental approach has been used to improve catalysts for the selective oxidation of carbon monoxide in hydrogen feedstocks.
- Gavin Armstrong
-
News & Views |
 Catalysing dynamic libraries
Catalysing dynamic libraries
The composition of dynamic small-molecule libraries can be biased by the addition of a target compound — such as a protein — that binds selectively to one of the components in the mixture. The chemistry of the library must, however, be compatible with the target and it has now been shown that aniline-catalysed exchange of acylhydrazones fits the bill.
- Benjamin L. Miller
-
Article |
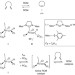 Mechanistic insights into the ruthenium-catalysed diene ring-closing metathesis reaction
Mechanistic insights into the ruthenium-catalysed diene ring-closing metathesis reaction
A kinetic and thermodynamic analysis of the ruthenium-catalysed ring-closing metathesis reaction has been achieved through the study of key intermediates accessible, for the first time, from 14-electron phosphonium alkylidene catalyst precursors. High intrinsic activities and a thermodynamic preference for ring-closing versus ring-opening reactions is observed.
- Edwin F. van der Eide
- & Warren E. Piers
-
Article |
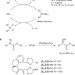 Enantioselective iron-catalysed O–H bond insertions
Enantioselective iron-catalysed O–H bond insertions
Iron is an abundant, low cost and environmentally benign metal. Here, iron complexes are shown to be the most effective catalysts for asymmetric O–H insertion reactions. These results should encourage the use of iron, rather than more traditional precious metals, in the development of greener organometallic catalysts for asymmetric transformations.
- Shou-Fei Zhu
- , Yan Cai
- & Qi-Lin Zhou
-
Article |
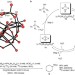 Efficient stereo- and regioselective hydroxylation of alkanes catalysed by a bulky polyoxometalate
Efficient stereo- and regioselective hydroxylation of alkanes catalysed by a bulky polyoxometalate
The ability to selectively transform the C–H bonds of simple alkanes to useful functional groups such as alcohols is a key step in the move away from petrochemical feedstocks. Now, it has been shown that the oxidation of alkanes can be catalysed by a bulky polyoxometalate species using hydrogen peroxide as a stoichiometric oxidant.
- Keigo Kamata
- , Kazuhiro Yonehara
- & Noritaka Mizuno
-
Article |
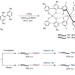 Ion-triggered spring-like motion of a double helicate accompanied by anisotropic twisting
Ion-triggered spring-like motion of a double helicate accompanied by anisotropic twisting
Helical molecules in biological systems commonly undergo extension, contraction and unidirectional twisting motions, but such twisting — promising for the construction of molecular machines — has rarely been achieved in synthetic systems. Now, a chiral double helix has been prepared whose spring-like motion is accompanied by an anisotropic twist under the control of sodium ions.
- Kazuhiro Miwa
- , Yoshio Furusho
- & Eiji Yashima
-
News & Views |
 Thinking inside and outside the box
Thinking inside and outside the box
Molecular 'boxes' can hold other molecules and often serve as the moving parts in molecular switches. The latest addition to this class of compounds is a simple-to-prepare positively charged macrocycle that can encircle molecular guests of appropriate size and charge — and offers new opportunities for assembling stimuli-responsive structures.
- Andrew I. Share
- & Amar H. Flood
-
Research Highlights |
 Pseudosymmetry solved
Pseudosymmetry solved
Two different but successful approaches to the synthesis of (+)-complanadine A will help in investigations of its interesting biological activity.
- Stephen Davey
-
Review Article |
 Induction of chiral porous solids containing only achiral building blocks
Induction of chiral porous solids containing only achiral building blocks
The synthesis or separation of chiral compounds is crucial for many areas of chemistry, with chiral solids having important roles as catalysts or separating agents. This Review covers recent progress and future avenues for developing methods of preparing chiral solids from achiral starting materials.
- Russell E. Morris
- & Xianhui Bu
-
Research Highlights |
Try the trication
A rationally designed highly electrophilic iridium complex excels at catalysing the formation of five-membered carbocycles.
- Stephen Davey
-
Research Highlights |
 Twice as nice
Twice as nice
A double-reductive alkylation of amides with two different organometallic compounds allows the direct formation of a tertiary alkylamines in one pot.
- Stephen Davey
-
Research Highlights |
 A solid combination
A solid combination
An efficient heterogeneous catalyst for the conversion of glucose into fructose has been prepared by incorporating tin centres into the framework of a zeolite.
- Anne Pichon
-
News & Views |
 Totally tubular peptide synthesis
Totally tubular peptide synthesis
The convergent total synthesis of the pore-forming polytheonamide B — a linear peptide natural product — pokes holes through perceived limitations in de novo peptide synthesis, and provides access to novel synthetic membrane channels.
- Craig J. Forsyth
-
News & Views |
 Without a trace
Without a trace
A strong acid catalyst and a small change to one reactant have transformed a 37-year-old 'curiosity' into a useful chemical reaction that has great potential in organic synthesis.
- Sarah E. Steinhardt
- & Christopher D. Vanderwal
-
Article |
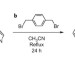 A ‘Texas-sized’ molecular box that forms an anion-induced supramolecular necklace
A ‘Texas-sized’ molecular box that forms an anion-induced supramolecular necklace
Macrocyles capable of hosting other molecules inside their hollow interiors have been used extensively to make threaded complexes and interlocked molecules. Now, a relatively large and flexible tetracationic macrocycle has been shown to bind anionic guests to form pseudorotaxanes that form extended structures in solution and the solid state.
- Han-Yuan Gong
- , Brett M. Rambo
- & Jonathan L. Sessler
-
Research Highlights |
 Turning on hydrocarbons
Turning on hydrocarbons
The intercalation of potassium into a simple aromatic hydrocarbon results in a new class of organic superconductors.
- Neil Withers
-
Article |
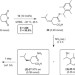 Synergistic organocatalysis in the kinetic resolution of secondary thiols with concomitant desymmetrization of an anhydride
Synergistic organocatalysis in the kinetic resolution of secondary thiols with concomitant desymmetrization of an anhydride
Chiral thiols and organosulfur compounds are important in many areas of chemistry but there are relatively few methods available for their efficient enantioselective synthesis. Here, a kinetic resolution of chiral thiols is reported along with a demonstration that a concomitant desymmetrization of the acylating agent is beneficial for the selectivity of both processes.
- Aldo Peschiulli
- , Barbara Procuranti
- & Stephen J. Connon
-
Research Highlights |
 Racemization on the rack
Racemization on the rack
Thermally stable stereoisomers can be interconverted by the application of a mechanical force using ultrasound irradiation.
- Stephen Davey
-
News & Views |
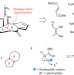 Stitching together palau'amine
Stitching together palau'amine
The long-awaited first total synthesis of the structurally intriguing natural product palau'amine has now been achieved. The synthesis features cascade reactions and an 'across ring' stitching of a 'macropalau'amine', and sets the bar for future efforts towards an enantioselective variant.
- Daniel Romo
-
News & Views |
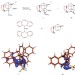 The same but different
The same but different
Electromerism is an unfamiliar concept to many chemists and refers to molecules that are not conventional isomers but instead differ in how the electrons are distributed across their structure. A novel example of such electromers has now been demonstrated.
- Thomas Bally
-
Article |
 Stereoinduction by distortional asymmetry
Stereoinduction by distortional asymmetry
Steric, torsion, stereoelectronic and polar effects are widely used to explain and predict the stereochemical outcome of synthetic organic reactions. Here, the asymmetric distortion of the reactant is considered and used to explain the observed stereoselectivity where these accepted models are unable to provide a clear prediction.
- Robert V. Kolakowski
- & Lawrence J. Williams
-
Article |
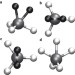 Quantum-induced symmetry breaking explains infrared spectra of CH5+ isotopologues
Quantum-induced symmetry breaking explains infrared spectra of CH5+ isotopologues
Stepwise deuteration of protonated methane CH5+ — a fluxional structure that undergoes ‘hydrogen scrambling’ — leads to dramatic changes in the infrared spectra of the isotopologues. The spectra can be assigned using ab initio quantum simulations that account for the non-classical occupation — by H and D atoms — of topologically different sites within the molecule.
- Sergei D. Ivanov
- , Oskar Asvany
- & Stephan Schlemmer
-
Article |
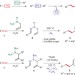 Triflimide-catalysed sigmatropic rearrangement of N-allylhydrazones as an example of a traceless bond construction
Triflimide-catalysed sigmatropic rearrangement of N-allylhydrazones as an example of a traceless bond construction
A triflimide-catalysed rearrangement of N-allylhydrazones has been developed that allows for the generation of a sigma bond between two unfunctionalized sp3 carbons such that no clear marker remains for how the bond was formed. This traceless bond construction offers new avenues for convergent fragment coupling in synthetic strategies.
- Devon A. Mundal
- , Christopher T. Avetta Jr
- & Regan J. Thomson
-
Article |
 Total synthesis of the large non-ribosomal peptide polytheonamide B
Total synthesis of the large non-ribosomal peptide polytheonamide B
Polytheonamide B is a large non-ribosomal peptide with very high bioactivity. The synthesis described here includes the first preparation of several non-proteinogenic amino acids and a general coupling strategy for large non-natural peptides. The synthesis is a key step necessary to understand and utilize the bioactivity of this and similar compounds.
- Masayuki Inoue
- , Naoki Shinohara
- & Shigeru Matsuoka
-
Review Article |
 Organocatalytic cascade reactions as a new tool in total synthesis
Organocatalytic cascade reactions as a new tool in total synthesis
The field of organocatalysis has grown rapidly in the past decade to become, along with metal catalysis and biocatalysis, a third pillar of asymmetric catalysis. Here, progress in the use of organocatalytic cascade reactions for total synthesis is reviewed. The elegance and efficiency of such cascades mean that they have emerged as a powerful tool in synthetic organic chemistry.
- Christoph Grondal
- , Matthieu Jeanty
- & Dieter Enders
-
Article |
 Exceptional ammonia uptake by a covalent organic framework
Exceptional ammonia uptake by a covalent organic framework
The uptake of ammonia by a covalent–organic framework (COF) containing a high density of Lewis-acidic boron sites has been found to be significantly greater than that exhibited by other state-of-the-art porous materials. The ammonia can be removed by heating under vacuum and the structural integrity of the COF is maintained during adsorption/desorption cycles.
- Christian J. Doonan
- , David J. Tranchemontagne
- & Omar M. Yaghi
-
Article |
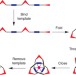 Synthesis of a molecular trefoil knot by folding and closing on an octahedral coordination template
Synthesis of a molecular trefoil knot by folding and closing on an octahedral coordination template
The synthesis of interlocked compounds such as catenanes and rotaxanes has undergone much development in recent years, but molecular knots are still relatively hard to make. It has now been shown that a linear bipyridine oligomer can fold around a single zinc-ion template to form a complex that can be cyclized to give a molecular trefoil knot.
- Jun Guo
- , Paul C. Mayers
- & Christopher A. Hunter
-
News & Views |
 Stereochemistry by remote control
Stereochemistry by remote control
The stereochemical lability of cycloalkylzinc reagents combined with a large difference in reactivity between epimers has been exploited to form a wide variety of interesting organic compounds with both high yields and diastereoselectivities.
- Frank Glorius
-
News & Views |
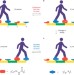 Tiny steps
Tiny steps
A molecular 'walker' can be made to move up and down a molecular 'track' by alternately locking and unlocking the two different types of covalent bonds that join the two components together. By changing the conditions under which one of the bond-forming/bond-breaking processes occurs, a directional bias for walking can be achieved.
- Sijbren Otto
-
Article |
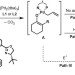 A palladium-catalysed enolate alkylation cascade for the formation of adjacent quaternary and tertiary stereocentres
A palladium-catalysed enolate alkylation cascade for the formation of adjacent quaternary and tertiary stereocentres
The selective construction of multiple adjacent stereocentres is an important challenge for synthetic organic methodology, and only a handful of catalytic methods exist that can forge adjacent quaternary and tertiary stereocentres. Here, a palladium-catalysed multiple-bond-forming cascade leads to the construction of such systems in high yield, diastereomeric ratio and enantiomeric excess.
- Jan Streuff
- , David E. White
- & Brian M. Stoltz
-
Article |
 Highly diastereoselective Csp3–Csp2 Negishi cross-coupling with 1,2-, 1,3- and 1,4-substituted cycloalkylzinc compounds
Highly diastereoselective Csp3–Csp2 Negishi cross-coupling with 1,2-, 1,3- and 1,4-substituted cycloalkylzinc compounds
Highly diastereoselective Negishi cross-coupling reactions between 2-, 3- and 4-substituted cycloalkylzinc reagents and aryl iodides are described. In all cases, the thermodynamically most stable diastereomers of the cross-coupling products were obtained. NMR spectroscopy and density functional theory calculations were performed in order to rationalize the observed stereoselectivities.
- Tobias Thaler
- , Benjamin Haag
- & Paul Knochel
-
Article |
 Random two-dimensional string networks based on divergent coordination assembly
Random two-dimensional string networks based on divergent coordination assembly
The bulk properties of materials that lack long-range order have been widely studied, but their local structures remain difficult to elucidate. Now, using scanning tunnelling microscopy, researchers have been able to look more closely at the structural motifs of robust, two-dimensional glassy networks assembled through metal–ligand interactions.
- Matthias Marschall
- , Joachim Reichert
- & Johannes V. Barth
-
Thesis |
 Life is the variety of spice
Life is the variety of spice
Bruce C. Gibb wonders why curry is not part of the chemistry curriculum.
- Bruce C. Gibb
-
News & Views |
 Internal construction
Internal construction
Among the wide variety of synthetic processes that chemists have developed, only a few can be carried out under physiological conditions. A condensation reaction that is controlled by the constituents of cells has led to the formation of nanostructures within living cells.
- Bing Xu

 Lewis acid-catalysed formation of two-dimensional phthalocyanine covalent organic frameworks
Lewis acid-catalysed formation of two-dimensional phthalocyanine covalent organic frameworks