Abstract
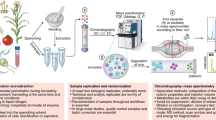
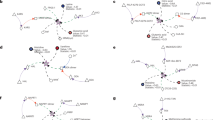
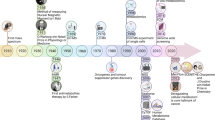
Metabolites, the chemical entities that are transformed during metabolism, provide a functional readout of cellular biochemistry. With emerging technologies in mass spectrometry, thousands of metabolites can now be quantitatively measured from minimal amounts of biological material, which has thereby enabled systems-level analyses. By performing global metabolite profiling, also known as untargeted metabolomics, new discoveries linking cellular pathways to biological mechanism are being revealed and are shaping our understanding of cell biology, physiology and medicine.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Smith, C. A., Want, E. J., O'Maille, G., Abagyan, R. & Siuzdak, G. XCMS: processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Anal. Chem. 78, 779–787 (2006).
Patti, G. J. et al. Nanostructure-initiator mass spectrometry (NIMS) imaging of brain cholesterol metabolites in Smith-Lemli-Opitz syndrome. Neuroscience 170, 858–864 (2010).
Koizumi, S. et al. Imaging mass spectrometry revealed the production of lyso-phosphatidylcholine in the injured ischemic rat brain. Neuroscience 168, 219–225 (2010).
Baker, M. Metabolomics: from small molecules to big ideas. Nature Methods 8, 117–121 (2011).
Kind, T., Scholz, M. & Fiehn, O. How large is the metabolome? A critical analysis of data exchange practices in chemistry. PLoS ONE 4, e5440 (2009).
Dudley, E., Yousef, M., Wang, Y. & Griffiths, W. J. Targeted metabolomics and mass spectrometry. Adv. Protein Chem. Struct. Biol. 80, 45–83 (2010).
Nicholson, J. K., Connelly, J., Lindon, J. C. & Holmes, E. Metabonomics: a platform for studying drug toxicity and gene function. Nature Rev. Drug Discov. 1, 153–161 (2002).
Dirren, H., Robinson, A. B. & Pauling, L. Sex-related patterns in the profiles of human urinary amino acids. Clin. Chem. 21, 1970–1975 (1975).
Davison, J. M. & Cheyne, G. A. History of the measurement of glucose in urine: a cautionary tale. Med. Hist. 18, 194–197 (1974).
Robinson, A. B. & Robinson, N. E. Origins of metabolic profiling. Methods Mol. Biol. 708, 1–23 (2011).
Dalgliesh, C. E., Horning, E. C., Horning, M. G., Knox, K. L. & Yarger, K. A gas-liquid-chromatographic procedure for separating a wide range of metabolites occuring in urine or tissue extracts. Biochem. J. 101, 792–810 (1966).
Eknoyan, G. Santorio Sanctorius (1561–1636) —founding father of metabolic balance studies. Am. J. Nephrol. 19, 226–233 (1999).
Astarita, G., Ahmed, F. & Piomelli, D. Lipidomic analysis of biological samples by liquid chromatography coupled to mass spectrometry. Methods Mol. Biol. 579, 201–219 (2009).
Want, E. J. et al. Solvent-dependent metabolite distribution, clustering, and protein extraction for serum profiling with mass spectrometry. Anal. Chem. 78, 743–752 (2006).
Yanes, O., Tautenhahn, R., Patti, G. J. & Siuzdak, G. Expanding coverage of the metabolome for global metabolite profiling. Anal. Chem. 83, 2152–2161 (2011).
Pike, L. S., Smift, A. L., Croteau, N. J., Ferrick, D. A. & Wu, M. Inhibition of fatty acid oxidation by etomoxir impairs NADPH production and increases reactive oxygen species resulting in ATP depletion and cell death in human glioblastoma cells. Biochim. Biophys. Acta 1807, 726–734 (2011).
Buxton, D. B., Schwaiger, M., Nguyen, A., Phelps, M. E. & Schelbert, H. R. Radiolabeled acetate as a tracer of myocardial tricarboxylic acid cycle flux. Circ. Res. 63, 628–634 (1988).
Bajad, S. U. et al. Separation and quantitation of water soluble cellular metabolites by hydrophilic interaction chromatography-tandem mass spectrometry. J. Chromatogr. A 1125, 76–88 (2006).
Bennett, B. D. et al. Absolute metabolite concentrations and implied enzyme active site occupancy in Escherichia coli. Nature Chem. Biol. 5, 593–599 (2009).
Buescher, J. M., Moco, S., Sauer, U. & Zamboni, N. Ultrahigh performance liquid chromatography-tandem mass spectrometry method for fast and robust quantification of anionic and aromatic metabolites. Anal. Chem. 82, 4403–4412 (2010).
Sabatine, M. S. et al. Metabolomic identification of novel biomarkers of myocardial ischemia. Circulation 112, 3868–3875 (2005).
Wang, T. J. et al. Metabolite profiles and the risk of developing diabetes. Nature Med. 17, 448–453 (2011).
Shaham, O. et al. Metabolic profiling of the human response to a glucose challenge reveals distinct axes of insulin sensitivity. Mol. Syst. Biol. 4, 214 (2008).
Barton, R. H., Nicholson, J. K., Elliott, P. & Holmes, E. High-throughput 1H NMR-based metabolic analysis of human serum and urine for large-scale epidemiological studies: validation study. Int. J. Epidemiol. 37 (Suppl. 1), I31–I40 (2008).
Buscher, J. M., Czernik, D., Ewald, J. C., Sauer, U. & Zamboni, N. Cross-platform comparison of methods for quantitative metabolomics of primary metabolism. Anal. Chem. 81, 2135–2143 (2009).
Jonsson, P. et al. Extraction, interpretation and validation of information for comparing samples in metabolic LC/MS data sets. Analyst 130, 701–707 (2005).
Nordstrom, A., Want, E., Northen, T., Lehtio, J. & Siuzdak, G. Multiple ionization mass spectrometry strategy used to reveal the complexity of metabolomics. Anal. Chem. 80, 421–429 (2008).
Wikoff, W. R. et al. Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc. Natl Acad. Sci. USA 106, 3698–3703 (2009).
Yanes, O. et al. Metabolic oxidation regulates embryonic stem cell differentiation. Nature Chem. Biol. 6, 411–417 (2010).
Baran, R. et al. MathDAMP: a package for differential analysis of metabolite profiles. BMC Bioinformatics 7, 530 (2006).
Lommen, A. MetAlign: interface-driven, versatile metabolomics tool for hyphenated full-scan mass spectrometry data preprocessing. Anal. Chem. 81, 3079–3086 (2009).
Katajamaa, M., Miettinen, J. & Oresic, M. MZmine: toolbox for processing and visualization of mass spectrometry based molecular profile data. Bioinformatics 22, 634–636 (2006).
Katajamaa, M. & Oresic, M. Processing methods for differential analysis of LC/MS profile data. BMC Bioinformatics 6, 179 (2005).
Tautenhahn, R., Bottcher, C. & Neumann, S. Highly sensitive feature detection for high resolution LC/MS. BMC Bioinformatics 9, 504 (2008).
Beadle, G. W. & Tatum, E. L. Genetic control of biochemical reactions in neurospora. Proc. Natl Acad. Sci. USA 27, 499–506 (1941).
Tatum, E. L. & Beadle, G. W. Genetic control of biochemical reactions in neurospora: an “Aminobenzoicless” mutant. Proc. Natl Acad. Sci. USA 28, 234–243 (1942).
Suhre, K. et al. Human metabolic individuality in biomedical and pharmaceutical research. Nature 477, 54–60 (2011).
Nicholson, G. et al. Human metabolic profiles are stably controlled by genetic and environmental variation. Mol. Syst. Biol. 7, 525 (2011).
Fiehn, O. Combining genomics, metabolome analysis, and biochemical modelling to understand metabolic networks. Comp. Funct. Genomics 2, 155–168 (2001).
Trauger, S. A. et al. Correlating the transcriptome, proteome, and metabolome in the environmental adaptation of a hyperthermophile. J. Proteome Res. 7, 1027–1035 (2008).
Kirschner, M. W. The meaning of systems biology. Cell 121, 503–504 (2005).
Kenyon, C., Chang, J., Gensch, E., Rudner, A. & Tabtiang, R. A C. elegans mutant that lives twice as long as wild type. Nature 366, 461–464 (1993).
Dong, M. Q. et al. Quantitative mass spectrometry identifies insulin signaling targets in C. elegans. Science 317, 660–663 (2007).
Kang, S., Bader, A. G. & Vogt, P. K. Phosphatidylinositol 3-kinase mutations identified in human cancer are oncogenic. Proc. Natl Acad. Sci. USA 102, 802–807 (2005).
Villas-Boas, S. G., Hojer-Pedersen, J., Akesson, M., Smedsgaard, J. & Nielsen, J. Global metabolite analysis of yeast: evaluation of sample preparation methods. Yeast 22, 1155–1169 (2005).
Wittmann, C., Kromer, J. O., Kiefer, P., Binz, T. & Heinzle, E. Impact of the cold shock phenomenon on quantification of intracellular metabolites in bacteria. Anal. Biochem. 327, 135–139 (2004).
Patti, G. J. Separation strategies for untargeted metabolomics. J. Sep. Sci. 34, 3460–3469 (2011).
Lu, W. et al. Metabolomic analysis via reversed-phase ion-pairing liquid chromatography coupled to a stand alone orbitrap mass spectrometer. Anal. Chem. 82, 3212–3221 (2010).
Han, X. & Gross, R. W. Shotgun lipidomics: electrospray ionization mass spectrometric analysis and quantitation of cellular lipidomes directly from crude extracts of biological samples. Mass Spectrom. Rev. 24, 367–412 (2005).
Wolters, D. A., Washburn, M. P. & Yates, J. R. 3rd. An automated multidimensional protein identification technology for shotgun proteomics. Anal. Chem. 73, 5683–5690 (2001).
Melamud, E., Vastag, L. & Rabinowitz, J. D. Metabolomic analysis and visualization engine for LC-MS data. Anal. Chem. 82, 9818–9826 (2010).
Wishart, D. S. et al. HMDB: a knowledgebase for the human metabolome. Nucleic Acids Res. 37, D603–D610 (2009).
Forsythe, I. J. & Wishart, D. S. Exploring human metabolites using the human metabolome database. Curr. Protoc. Bioinformatics Ch. 14, Unit 14.8 (2009).
Smith, C. A. et al. METLIN: a metabolite mass spectral database. Ther. Drug Monit. 27, 747–751 (2005).
Patti, G. J., Tautenhahn, R. & Siuzdak, G. Meta-analysis of untargeted metabolomic data from multiple profiling experiments. Nature Protoc. 7, 508–516 (2012).
Tautenhahn, R. et al. metaXCMS: second-order analysis of untargeted metabolomics data. Anal. Chem. 83, 696–700 (2011).
Bottomley, P. A., Hardy, C. J. & Roemer, P. B. Phosphate metabolite imaging and concentration measurements in human heart by nuclear magnetic resonance. Magn. Reson. Med. 14, 425–434 (1990).
Golman, K., Zandt, R. I., Lerche, M., Pehrson, R. & Ardenkjaer-Larsen, J. H. Metabolic imaging by hyperpolarized 13C magnetic resonance imaging for in vivo tumor diagnosis. Cancer Res. 66, 10855–10860 (2006).
Cakmakci, H., Pekcevik, Y., Yis, U., Unalp, A. & Kurul, S. Diagnostic value of proton MR spectroscopy and diffusion-weighted MR imaging in childhood inherited neurometabolic brain diseases and review of the literature. Eur. J. Radiol. 74, e161–e171 (2010).
Greving, M. P., Patti, G. J. & Siuzdak, G. Nanostructure-initiator mass spectrometry metabolite analysis and imaging. Anal. Chem. 83, 2–7 (2011).
Northen, T. R. et al. Clathrate nanostructures for mass spectrometry. Nature 449, 1033–1036 (2007).
Woo, H. K., Northen, T. R., Yanes, O. & Siuzdak, G. Nanostructure-initiator mass spectrometry: a protocol for preparing and applying NIMS surfaces for high-sensitivity mass analysis. Nature Protoc. 3, 1341–1349 (2008).
Rabinowitz, J. D., Purdy, J. G., Vastag, L., Shenk, T. & Koyuncu, E. Metabolomics in drug target discovery. Cold Spring Harb. Symp. Quant. Biol. 23 Nov 2011 (doi:10.1101/sqb.2011.76.010694).
Dang, L. et al. Cancer-associated IDH1 mutations produce 2-hydroxyglutarate. Nature 462, 739–744 (2009).
Patti, G. J. et al. Metabolomics implicates altered sphingolipids in chronic pain of neuropathic origin. Nature Chem. Biol. 8, 232–234 (2012).
Saghatelian, A. et al. Assignment of endogenous substrates to enzymes by global metabolite profiling. Biochemistry 43, 14332–14339 (2004).
Clasquin, M. F. et al. Riboneogenesis in yeast. Cell 145, 969–980 (2011).
de Carvalho, L. P. et al. Activity-based metabolomic profiling of enzymatic function: identification of Rv1248c as a mycobacterial 2-hydroxy-3-oxoadipate synthase. Chem. Biol. 17, 323–332 (2010).
Feynman, R. P., Leighton, R. B. & Sands, M. L. The Feynman Lectures on Physics (Addison-Wesley Publishing Company, Boston, 1963).
Segre, E. From X-rays to Quarks: Modern Physicists and Their Discoveries (Dover Publications, New York, 2007).
Acknowledgements
The authors gratefully acknowledge that their work is supported by the California Institute of Regenerative Medicine (grant TR1-01219; G.S.), the US National Institutes of Health (grants R24 EY017540-04, P30 MH062261-10 and P01 DA026146-02; G.S.), the US Department of Energy (grants FG02-07ER64325 and DE-AC0205CH11231; G.S.), the US National Institutes of Health/National Institute on Aging (L30 AG0 038036; G.J.P.) and start-up funds from Washington University (G.J.P.).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Imaging mass spectrometry
-
A surface-based approach in which molecules such as metabolites and proteins are spatially analysed in biological specimens. Common imaging mass spectrometry techniques are matrix-assisted laser desorption ionization (MALDI), secondary ion mass spectrometry (SIMS) and nanostructure-initiator mass spectrometry (NIMS).
- Ion trap
-
A type of mass spectrometer that traps ions by using electric or magnetic fields. Once trapped, the ions are analysed to determine their mass-to-charge ratios. Tandem mass spectrometry can also be performed on selected ions by isolating them in the trap and then subjecting them to dissociation.
- Matrix-assisted laser desorption ionization
-
(MALDI). A surface-based mass spectrometry approach in which analytes are embedded in a chemical matrix that absorbs energy from an ultraviolet laser, resulting in analyte desorption and ionization.
- MS1 mode
-
The mode of a mass spectrometer in which only the mass-to-charge ratio of the intact ion is measured. In these experiments no tandem mass spectrometry is performed.
- Nanostructure-initiator mass spectrometry
-
(NIMS). A nanostructure surface-based mass spectrometry approach that does not require a matrix. NIMS is commonly used for metabolomic studies and metabolite imaging.
- Nuclear magnetic resonance
-
(NMR). An analytical technique that exploits the different responses to radiofrequency stimuli by chemically distinct atomic nuclei in an external magnetic field to provide information about the structure and dynamics of molecules.
- Quadrupole time-of-flight
-
(QTOF). A mass spectrometer commonly used to perform untargeted metabolomics. By using the quadrupole as a focusing lens, the time-of-flight mass analyser can be used to acquire profiling data. Alternatively, the quadrupole can be used to select ions for tandem mass spectrometry experiments.
- Tandem mass spectrometry
-
(MS/MS). A type of mass spectrometry in which ions are selectively isolated and then fragmented. The mass-to-charge ratio of each molecular fragment is measured and used for structural characterization.
- Triple quadrupole
-
(QqQ). A type of mass spectrometer that is often used for targeted studies owing to its sensitivity and specificity. The QqQ analyser consists of a quadrupole, a quadrupole collision cell and a second quadrupole, respectively. The first selects and analyses ions of interest, the second is used as a collision cell for fragmentation and the third analyses fragments.
Rights and permissions
About this article
Cite this article
Patti, G., Yanes, O. & Siuzdak, G. Metabolomics: the apogee of the omics trilogy. Nat Rev Mol Cell Biol 13, 263–269 (2012). https://doi.org/10.1038/nrm3314
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrm3314
This article is cited by
-
Identification of blood metabolites associated with risk of Alzheimer’s disease by integrating genomics and metabolomics data
Molecular Psychiatry (2024)
-
Urine and serum metabolomic analysis of endometrial cancer diagnosis and classification based on ultra-performance liquid chromatography mass spectrometry
Metabolomics (2024)
-
Untargeted serum metabolomic profiles and breast density in young women
Cancer Causes & Control (2024)
-
Global untargeted and individual targeted plasma metabolomics of breast cancer recurrence modified by hormone receptors
Breast Cancer (2024)
-
The hidden treasures in endophytic fungi: a comprehensive review on the diversity of fungal bioactive metabolites, usual analytical methodologies, and applications
Archives of Microbiology (2024)