Article
|
Open Access
Featured
-
-
Article
| Open Access Modulating stereoselectivity in allylic C(sp3)-H bond arylations via nickel and photoredox catalysis
Modulating stereoselectivity in allylic C(sp3)-H bond arylations via nickel and photoredox catalysis
The selective construction of C-C double bonds in alkenes, which can be one of two isomers, is a fundamental focus of organic chemistry. Here, the authors systematically investigated the stereoselectivity in allylic C(sp3)-H bond arylations and demonstrated a ligand switch could allow a stereodivergent synthesis of silyl enol ethers.
- Long Huang
- , Marcin Szewczyk
- & Magnus Rueping
-
Article
| Open Access The rocky road to organics needs drying
The rocky road to organics needs drying
How complex organics form in a prebiotic world remains a missing key to establish where life emerged. The authors present a road to abiotic organic synthesis and diversification in hydrothermal contexts involving magmatism and rock hydration.
- Muriel Andreani
- , Gilles Montagnac
- & Bénédicte Ménez
-
Article
| Open Access Preferential molecular recognition of heterochiral guests within a cyclophane receptor
Preferential molecular recognition of heterochiral guests within a cyclophane receptor
The design of synthetic supramolecular receptors capable of enantioselective molecular recognition is challenging. Here the authors report a perylene bisimide-based receptor that forms heterochiral host-guest complexes upon aromatic guests encapsulation; guest recognition can be modulated by peripheral substitution of the binding site.
- Manuel Weh
- , Kazutaka Shoyama
- & Frank Würthner
-
Article
| Open Access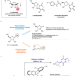 Additive-controlled asymmetric iodocyclization enables enantioselective access to both α- and β-nucleosides
Additive-controlled asymmetric iodocyclization enables enantioselective access to both α- and β-nucleosides
Nucleosides and their analogs are pharmacologically important molecules. Here, the authors report an additive-controlled stereodivergent iodocyclization method for the selective synthesis of α- or β-nucleosides.
- Qi Wang
- , Jiayi Mu
- & Fener Chen
-
Article
| Open Access Site- and enantioselective cross-coupling of saturated N-heterocycles with carboxylic acids by cooperative Ni/photoredox catalysis
Site- and enantioselective cross-coupling of saturated N-heterocycles with carboxylic acids by cooperative Ni/photoredox catalysis
Chiral α-acylated N-heterocycles are commonly found in pharmaceutical drugs, natural products, and catalysts for asymmetric synthesis. Here, the authors describe a cooperative nickel/photoredox catalysis strategy for the site- and enantioselective cross-coupling of saturated azacycles and carboxylic acids to access such valuable heterocyclic molecules.
- Xiaomin Shu
- , De Zhong
- & Haohua Huo
-
Article
| Open Access Formation and degradation of strongly reducing cyanoarene-based radical anions towards efficient radical anion-mediated photoredox catalysis
Formation and degradation of strongly reducing cyanoarene-based radical anions towards efficient radical anion-mediated photoredox catalysis
Cyanoarene-based photocatalysts are widely used due to their catalytic performance, but the formation of the active species and its potential degradation pathways are poorly understood. Here, the authors investigate these pathways under commonly-used photoredox-mediated reaction conditions.
- Yonghwan Kwon
- , Jungwook Lee
- & Min Sang Kwon
-
Article
| Open Access Ligand-controlled stereodivergent alkenylation of alkynes to access functionalized trans- and cis-1,3-dienes
Ligand-controlled stereodivergent alkenylation of alkynes to access functionalized trans- and cis-1,3-dienes
The stereoselective synthesis of 1,3-dienes represents a challenge in organic synthesis. Here, the authors report a ligand-controlled stereodivergent access to both Z- and E-sulfonyl-1,3-dienes via photoredox and nickel catalysis.
- Tianyu Long
- , Chen Zhu
- & Lingling Chu
-
Article
| Open Access Transition-metal free C–N bond formation from alkyl iodides and diazonium salts via halogen-atom transfer
Transition-metal free C–N bond formation from alkyl iodides and diazonium salts via halogen-atom transfer
The pursuit of efficient C–N bond formation is a prime focus of synthetic organic chemistry. Here, the authors documented a base promoted amination between alkyl iodides and diazonium salts via halogen-atom transfer (XAT) process.
- Jing Zhang
- , Min Jiang
- & Xinxin Shao
-
Article
| Open Access Iron-mediated ligand-to-metal charge transfer enables 1,2-diazidation of alkenes
Iron-mediated ligand-to-metal charge transfer enables 1,2-diazidation of alkenes
Alkene diazidations represent a promising strategy for the synthesis of 1,2-diamines. Here, the authors report a protocol that enables alkene diazidation via iron-mediated ligand-to-metal charge transfer, providing a versatile platform to access structurally diverse diazides without using external oxidants.
- Muliang Zhang
- , Jinghui Zhang
- & Yumeng Shi
-
Article
| Open Access Photochemical diazidation of alkenes enabled by ligand-to-metal charge transfer and radical ligand transfer
Photochemical diazidation of alkenes enabled by ligand-to-metal charge transfer and radical ligand transfer
The difunctionalization of alkenes allows for efficient construction of molecular complexity. Combining Ligand-to-Metal Charge transfer (LMCT) and Radical Ligand Transfer (RLT), the authors show a photochemical diazidation using iron salts, allowing rapid preparation of vicinal diamines and other privileged synthetic motifs.
- Kang-Jie Bian
- , Shih-Chieh Kao
- & Julian G. West
-
Article
| Open Access Ni-catalyzed benzylic β-C(sp3)–H bond activation of formamides
Ni-catalyzed benzylic β-C(sp3)–H bond activation of formamides
The development of 3d-metal-catalyzed β-C–H bond activation via 4-membered metallacycles remains an elusive challenge. Here, the authors report a Ni-Al bimetal-catalyzed β-C(sp3)–H bond activation of formamides via 4-membered nickelacycles.
- Rong-Hua Wang
- , Wei-Wei Xu
- & Mengchun Ye
-
Article
| Open Access Synthesis of macrocyclic nucleoside antibacterials and their interactions with MraY
Synthesis of macrocyclic nucleoside antibacterials and their interactions with MraY
MraY is a membrane enzyme required for bacterial cell wall synthesis. Here, the authors modify sphaerimicins as antibacterials targeting it via structure-based design and synthesis through two key reactions, showing a platform for further development of MraY inhibitors as antibacterials.
- Takeshi Nakaya
- , Miyuki Yabe
- & Satoshi Ichikawa
-
Article
| Open Access Rational enzyme design for enabling biocatalytic Baldwin cyclization and asymmetric synthesis of chiral heterocycles
Rational enzyme design for enabling biocatalytic Baldwin cyclization and asymmetric synthesis of chiral heterocycles
Chiral heterocyclic compounds are privileged structures in medicinal chemistry. Here, the authors report an in silico strategy for the enzymatic synthesis of pharmaceutically significant chiral N- and O-heterocycles via Baldwin cyclization of hydroxy- and amino-substituted epoxides and oxetanes using epoxide hydrolase mutants.
- Jun-Kuan Li
- , Ge Qu
- & Zhoutong Sun
-
Article
| Open Access Stereodivergent synthesis of chiral succinimides via Rh-catalyzed asymmetric transfer hydrogenation
Stereodivergent synthesis of chiral succinimides via Rh-catalyzed asymmetric transfer hydrogenation
Chiral succinimides are ubiquitous in bioactive compounds. Here, authors report a stereodivergent method for accessing 3,4-disubstituted succinimides, and the origin of stereoselectivity is elucidated by experimental and theoretical investigations.
- Fangyuan Wang
- , Zongpeng Zhang
- & Xumu Zhang
-
Article
| Open Access Chemoselective carbene insertion into the N−H bonds of NH3·H2O
Chemoselective carbene insertion into the N−H bonds of NH3·H2O
Converting low-cost inorganic chemicals into value-added organic chemicals is a longstanding goal in chemistry. Here the authors describe a silver-catalysed chemoselective carbene N−H insertion reaction, providing access to primary amines from aqueous ammonia and diazo compounds.
- Zhaohong Liu
- , Yong Yang
- & Xihe Bi
-
Article
| Open Access Catalytic direct hydrocarboxylation of styrenes with CO2 and H2
Catalytic direct hydrocarboxylation of styrenes with CO2 and H2
Hydrocarboxylation of olefins with CO2 provides a useful approach for the synthesis of aliphatic carboxylic acids. Here, the authors report a rhodium-catalysed hydrocarboxylation of styrenes with CO2, using hydrogen as the terminal reductant.
- Yushu Jin
- , Joaquim Caner
- & Nobuharu Iwasawa
-
Article
| Open Access Organocatalyst-mediated five-pot synthesis of (–)-quinine
Organocatalyst-mediated five-pot synthesis of (–)-quinine
Syntheses of quinine have fascinated organic chemists for over a hundred years. Here, the authors developed an organocatalyst-mediated pot-economical enantioselective total synthesis using five reaction vessels.
- Takahiro Terunuma
- & Yujiro Hayashi
-
Article
| Open Access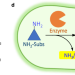 A growth selection system for the directed evolution of amine-forming or converting enzymes
A growth selection system for the directed evolution of amine-forming or converting enzymes
Fast screening of enzymes is key for directed evolution of industrial biocatalysts. Here, the authors report a simple, high-throughput, and low-equipment-dependent growth selection system for engineering three enzymes for synthesis of chiral amines.
- Shuke Wu
- , Chao Xiang
- & Uwe T. Bornscheuer
-
Article
| Open Access Photoinduced β-fragmentation of aliphatic alcohol derivatives for forging C–C bonds
Photoinduced β-fragmentation of aliphatic alcohol derivatives for forging C–C bonds
Transformations of alcohols, which are ubiquitous in chemistry and are native functionalities in many natural products and bioactive molecules, are cornerstones of organic synthesis. Here the authors describe photocatalyzed cross-couplings of activated alcohols with α-amino acids, providing a direct approach for a variety of unnatural amino acids.
- Yiman Gao
- , Jie Liu
- & Lingchao Cai
-
Article
| Open Access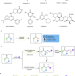 Azoarene activation for Schmidt-type reaction and mechanistic insights
Azoarene activation for Schmidt-type reaction and mechanistic insights
The Schmidt reaction enables nitrogen insertion across an aldehyde or ketone substrate by frequently using azide reagents. Here, the authors show a Schmidt reaction of aryldiazonium salts as a replacement of azide reagents to access skeletally diverse cyclic lactams.
- Fan-Tao Meng
- , Ya-Nan Wang
- & Bo Jiang
-
Article
| Open Access Paired electrolysis-enabled nickel-catalyzed enantioselective reductive cross-coupling between α-chloroesters and aryl bromides
Paired electrolysis-enabled nickel-catalyzed enantioselective reductive cross-coupling between α-chloroesters and aryl bromides
Precise matching of reaction rate and electric potential of electrodes in paired electrolysis is challenging. Here, the authors develop paired-electrolysis-enabled nickel-catalyzed enantioselective reductive cross-coupling of α-chloroesters and aryl bromides.
- Dong Liu
- , Zhao-Ran Liu
- & Tian-Sheng Mei
-
Article
| Open Access Chiral aldehyde-nickel dual catalysis enables asymmetric α−propargylation of amino acids and stereodivergent synthesis of NP25302
Chiral aldehyde-nickel dual catalysis enables asymmetric α−propargylation of amino acids and stereodivergent synthesis of NP25302
The combination of transition metal catalysis and organocatalysis can afford both good reactivity and selectivity. Here, the authors present an α − propargylation of N-unprotected amino acid esters with propargylic alcohol derivatives via dual nickel and chiral aldehyde catalysis.
- Fang Zhu
- , Chao-Xing Li
- & Qi-Xiang Guo
-
Article
| Open Access How mono- and diphosphine ligands alter regioselectivity of the Rh-catalyzed annulative cleavage of bicyclo[1.1.0]butanes
How mono- and diphosphine ligands alter regioselectivity of the Rh-catalyzed annulative cleavage of bicyclo[1.1.0]butanes
Ligands alter the regioselectivity of Rh(I)- catalyzed cycloisomerizations of bicyclobutanes. Here, mechanistic studies performed by the authors pinpoint a concerted C−C bond cleavage and carbenoid formation responsible for the divergence.
- Pan-Pan Chen
- , Peter Wipf
- & K. N. Houk
-
Article
| Open Access A theory-driven synthesis of symmetric and unsymmetric 1,2-bis(diphenylphosphino)ethane analogues via radical difunctionalization of ethylene
A theory-driven synthesis of symmetric and unsymmetric 1,2-bis(diphenylphosphino)ethane analogues via radical difunctionalization of ethylene
DPPEs are fundamental bidentate ligands with a C2-alkyl-linker chain for many transition-metal-catalyzed reactions. Here, authors utilize the AFIR method to develop a practical synthetic method for both symmetric and unsymmetric DPPEs with ethylene.
- Hideaki Takano
- , Hitomi Katsuyama
- & Tsuyoshi Mita
-
Article
| Open Access Photocatalytic direct borylation of carboxylic acids
Photocatalytic direct borylation of carboxylic acids
Boronic acids are useful reagents in organic synthesis and thus methods to prepare these compounds under mild conditions are desirable. Here, the authors synthesize boronic acids via photocatalytic decarboxylation of benzoic acids followed by borylation; guanidine-type additives are used to assist the process.
- Qiang Wei
- , Yuhsuan Lee
- & Zhibo Liu
-
Article
| Open Access Regioselective Ni-Catalyzed reductive alkylsilylation of acrylonitrile with unactivated alkyl bromides and chlorosilanes
Regioselective Ni-Catalyzed reductive alkylsilylation of acrylonitrile with unactivated alkyl bromides and chlorosilanes
Synthetic methodologies that enable the formation of C‒Si bonds in organic molecules are desirable. Here, the authors report the nickel-catalyzed reductive alkylsilylation of acrylonitrile with unactivated alkyl bromides and chlorosilanes.
- Jinwei Sun
- , Yongze Zhou
- & Xuan Zhang
-
Article
| Open Access Decarboxylative oxidation-enabled consecutive C-C bond cleavage
Decarboxylative oxidation-enabled consecutive C-C bond cleavage
The selective cleavage of inert C(sp3)-C(sp3) bonds and their subsequent functionalization is an important goal in synthetic organic chemistry. Here, the authors developed consecutive C–C bond cleavage from stable trisubstituted acids via photocatalysis and copper catalysis.
- Ruining Li
- , Ya Dong
- & Zhankui Sun
-
Article
| Open Access Accessing chiral sulfones bearing quaternary carbon stereocenters via photoinduced radical sulfur dioxide insertion and Truce–Smiles rearrangement
Accessing chiral sulfones bearing quaternary carbon stereocenters via photoinduced radical sulfur dioxide insertion and Truce–Smiles rearrangement
The preparation of chiral sulfones featuring quaternary carbon stereocenters remains challenging. Here the authors report the synthesis of such compounds via a multicomponent reaction involving a photoinduced radical sulfur dioxide insertion followed by an asymmetric Truce–Smiles rearrangement.
- Jiapian Huang
- , Fei Liu
- & Jie Wu
-
Article
| Open Access An enantioselective four-component reaction via assembling two reaction intermediates
An enantioselective four-component reaction via assembling two reaction intermediates
The interception of one chemical intermediate with another intermediate is very challenging, as intermediates may be incompatible or too reactive, but could result in a streamlined method of organic synthesis. Here, the authors demonstrate enantioselective four-component reactions via the coupling of two intermediates.
- Sifan Yu
- , Wenju Chang
- & Wenhao Hu
-
Article
| Open Access Enantioselective Ugi and Ugi-azide reactions catalyzed by anionic stereogenic-at-cobalt(III) complexes
Enantioselective Ugi and Ugi-azide reactions catalyzed by anionic stereogenic-at-cobalt(III) complexes
Ugi reactions are well known multicomponent reactions that allow the preparation of a variety of chemical compounds including peptides, heterocyclic compounds, or natural products. Here, the authors report enantioselective Ugi reactions that enable the synthesis of chiral α-acylamino amides and α-aminotetrazoles catalyzed by stereogenic-at-cobalt(III) complexes.
- Bing-Bing Sun
- , Kun Liu
- & Jie Yu
-
Article
| Open Access Nickel-catalyzed switchable 1,3-dienylation and enantioselective allenylation of phosphine oxides
Nickel-catalyzed switchable 1,3-dienylation and enantioselective allenylation of phosphine oxides
Asymmetric catalytic methods for the divergent construction of phosphoryl derivatives are not widely available. Here, the authors show nickel-catalyzed dienylation and enantioselective allenylation of phosphine oxides with high chemo-, regio-, and enantioselectivity.
- Jiayin Zhang
- , Xihao Chang
- & Chang Guo
-
Article
| Open Access Asymmetric construction of allylicstereogenic carbon center featuring atrifluoromethyl group via enantioselective reductive fluoroalkylation
Asymmetric construction of allylicstereogenic carbon center featuring atrifluoromethyl group via enantioselective reductive fluoroalkylation
The efficient construction of trifluoromethylated alkanes bearing a CF3 chiral center is of great importance in organic synthesis. Here, the authors disclose the enantioselective syntheses of α-trifluoromethylated allylic alkanes via reductive trifluoroalkylation.
- Ruo-Xing Jin
- , Bing-Bing Wu
- & Xi-Sheng Wang
-
Article
| Open Access Azide-functionalized ligand enabling organic–inorganic hybrid dielectric for high-performance solution-processed oxide transistors
Azide-functionalized ligand enabling organic–inorganic hybrid dielectric for high-performance solution-processed oxide transistors
Low cost and low power consumption of transistors are needed for the development of internet of things. Here Chung et al. develop a high performance n-type oxide thin film transistor by introducing a ligand for crosslinking nanoparticles and polymers, obtaining a near-ideal hybrid dielectric layer.
- Juhyeok Lee
- , Syed Zahid Hassan
- & Dae Sung Chung
-
Article
| Open Access Ligand-enabled Ni-catalyzed hydroarylation and hydroalkenylation of internal alkenes with organoborons
Ligand-enabled Ni-catalyzed hydroarylation and hydroalkenylation of internal alkenes with organoborons
The hydrocarbofunctionalization of alkenes represents one of the most efficient methods for generating complex products from simple starting materials. Here, the authors report a bulky β-diketone-ligand-enabled hydroarylation and hydroalkenylation of internal alkenes.
- Dao-Ming Wang
- , Li-Qin She
- & Peng Wang
-
Article
| Open Access Site-selective photocatalytic functionalization of peptides and proteins at selenocysteine
Site-selective photocatalytic functionalization of peptides and proteins at selenocysteine
The modification of peptides and proteins for application in drug discovery and chemical biology is currently a rapidly growing field of research. Here, the authors report a photocatalytic diselenide contraction method for the dimerization and site-specific functionalisation of peptides and protein.
- Luke J. Dowman
- , Sameer S. Kulkarni
- & Richard J. Payne
-
Article
| Open Access Enantioselective synthesis of N-alkylindoles enabled by nickel-catalyzed C-C coupling
Enantioselective synthesis of N-alkylindoles enabled by nickel-catalyzed C-C coupling
Enantioenriched N-alkylindole compounds, in which nitrogen is bound to a stereogenic sp3 carbon, are target molecules in the fields of biological, medicinal, and organic chemistry. Here the authors show a nickel-catalyzed C-C coupling protocol to synthesize these molecules from N-indolylsubstituted alkenes.
- Lun Li
- , Jiangtao Ren
- & Deyun Qian
-
Article
| Open Access Sila-spirocyclization involving unstrained C(sp3)−Si bond cleavage
Sila-spirocyclization involving unstrained C(sp3)−Si bond cleavage
Methods that enable the cleavage of Si−C(sp3) bonds catalytically remain rare. Here the authors report the synthesis of spirosilacycles via palladium-catalysed spiro-sila-cyclization of trialkylsilane derivatives; the reaction proceeds via cleavage of a C(sp3)−Si bond and insertion of an alkylpalladium complex.
- Yufeng Shi
- , Xiaonan Shi
- & Dongbing Zhao
-
Article
| Open Access Modular total syntheses of trans-clerodanes and sesquiterpene (hydro)quinones via tail-to-head cyclization and reductive coupling strategies
Modular total syntheses of trans-clerodanes and sesquiterpene (hydro)quinones via tail-to-head cyclization and reductive coupling strategies
trans-Clerodanes and sesquiterpene (hydro)quinones are different types of natural products featuring the same trans-decalin core. Here, the authors report a modular synthetic route to this family by tail-to-head cyclization and reductive coupling strategies.
- Wenming Zhu
- , Qishuang Yin
- & Ming Yang
-
Article
| Open Access Synthesis of I(III)/S(VI) reagents and their reactivity in photochemical cycloaddition reactions with unsaturated bonds
Synthesis of I(III)/S(VI) reagents and their reactivity in photochemical cycloaddition reactions with unsaturated bonds
Novel methodologies for introducing sulfoxonium groups into organic molecules are desirable. Here the authors report the synthesis of an iodonium-sulfoxonium ylide reagent that enables the preparation of sulfoxonium-containing cyclic structures upon reaction with alkenes, alkynes, nitriles or allenes.
- Li Li
- , Kun Deng
- & Yong Huang
-
Article
| Open Access The occurrence of ansamers in the synthesis of cyclic peptides
The occurrence of ansamers in the synthesis of cyclic peptides
The occurrence of isomers of the bicyclic octapeptide α-amanitin, which presents a macrolactam with a tryptathionine cross-link forming a handle, has been reported under the term of atropoisomers. Here, the authors synthesize its analogs and analyse their isomers, proposing and describing for them the term ansamer.
- Guiyang Yao
- , Simone Kosol
- & Roderich D. Süssmuth
-
Article
| Open Access Development of long lifespan high-energy aqueous organic||iodine rechargeable batteries
Development of long lifespan high-energy aqueous organic||iodine rechargeable batteries
Aqueous I2-based batteries are a promising system for cost-effective and environmentally-friendly electricity storage. Here, the authors propose a high-capacity and long-lasting aqueous I2 battery system using an electrochemically active organic molecule at the negative electrode.
- Zishuai Zhang
- , Yilong Zhu
- & Yan Huang
-
Article
| Open Access Dearomatization of aromatic asmic isocyanides to complex cyclohexadienes
Dearomatization of aromatic asmic isocyanides to complex cyclohexadienes
As aromatic compounds can be obtained easily, their functionalization is a cornerstone of organic synthesis. Here, the authors report a dearomatization of aromatic isocyanides, installing three carbon-carbon bonds, two quaternary stereocenters, and three orthogonal functionalities in one operation.
- Bilal Altundas
- , Embarek Alwedi
- & Fraser F. Fleming
-
Article
| Open Access Synthesis of N-acyl sulfenamides via copper catalysis and their use as S-sulfenylating reagents of thiols
Synthesis of N-acyl sulfenamides via copper catalysis and their use as S-sulfenylating reagents of thiols
Sulfenamides are organosulfur reagents that feature labile S–N bonds and have potential applications in chemical synthesis. Here, the authors report the preparation of N-acyl sulfenamides via copper-catalyzed S-amidation of thiols with dioxazolones; These N-acyl sulfenamides can be used to prepare unsymmetrical disulfides via reaction with thiols.
- Ziqian Bai
- , Shiyang Zhu
- & Gong Chen
-
Article
| Open Access Divergent regioselective Heck-type reaction of unactivated alkenes and N-fluoro-sulfonamides
Divergent regioselective Heck-type reaction of unactivated alkenes and N-fluoro-sulfonamides
The control of regioselectivity in Heck-type reactions of unactivated alkenes is challenging. Here, the authors realize regiodivergent Heck-type reactions of unactivated alkenes and N-fluoro-sulfonamides.
- Chunyang Zhao
- , Yang Li
- & Junkai Fu
-
Article
| Open Access Temporally programmed polymer – solvent interactions using a chemical reaction network
Temporally programmed polymer – solvent interactions using a chemical reaction network
Out of equilibrium operation of chemical reaction networks. (CRNs) enables materials to autonomously respond to their environment by activation and deactivation of intermolecular interactions but the deactivation to noninteracting building blocks remains understudied. Here, the authors develop a new, modular CRN that enables control over the deactivation kinetics.
- Benjamin Klemm
- , Reece W. Lewis
- & Rienk Eelkema
-
Article
| Open Access Coupling photocatalytic water oxidation with reductive transformations of organic molecules
Coupling photocatalytic water oxidation with reductive transformations of organic molecules
While reductive coupling strategies in organic synthesis are crucial, most require additional sacrificial or toxic reagents. Here, authors demonstrate water as mild reducing agent in the photochemical reduction of organic compounds paired with photocatalytic water oxidation.
- Xinzhe Tian
- , Yinggang Guo
- & Xin Zheng
-
Article
| Open Access Membrane-based TBADT recovery as a strategy to increase the sustainability of continuous-flow photocatalytic HAT transformations
Membrane-based TBADT recovery as a strategy to increase the sustainability of continuous-flow photocatalytic HAT transformations
Decatungstate-enabled Hydrogen Atom Transfer (HAT) photocatalysis of hydrocarbon scaffolds enabled rapid access to new C(sp3)-enriched chemical space. Here, the authors present in-line TBADT recovery with organic solvent nanofiltration (OSN) as an answer to the concerns surrounding the scale-up of decatungstate HAT reactions, i.e., catalyst loading and cost.
- Zhenghui Wen
- , Diego Pintossi
- & Timothy Noël
-
Article
| Open Access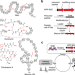 A scalable platform to discover antimicrobials of ribosomal origin
A scalable platform to discover antimicrobials of ribosomal origin
Ribosomally synthesized and post-translationally modified peptides are a source of antimicrobials. Here, the authors report a platform for the rapid evaluation and characterization of biosynthetic gene clusters that enables the identification of 30 structurally diverse modified peptides, including three showing antimicrobial activities.
- Richard S. Ayikpoe
- , Chengyou Shi
- & Huimin Zhao
-
Article
| Open Access Cascade cyclization of alkene-tethered acylsilanes and allylic sulfones enabled by unproductive energy transfer photocatalysis
Cascade cyclization of alkene-tethered acylsilanes and allylic sulfones enabled by unproductive energy transfer photocatalysis
Acylsilanes decompose under light irradiation, and this limits their use in light-induced organic transformations. Here the authors report a strategy to inhibit the light-induced decomposition of acylsilanes, enabling the photochemical synthesis of cyclopentanol derivatives from alkene-tethered acylsilanes and allylic sulfones.
- Yunxiao Zhang
- , Yizhi Zhang
- & Xiao Shen

 Nickel-catalyzed divergent Mizoroki–Heck reaction of 1,3-dienes
Nickel-catalyzed divergent Mizoroki–Heck reaction of 1,3-dienes