Featured
-
-
Article
| Open Access Targeted sampling of natural product space to identify bioactive natural product-like polyketide macrolides
Targeted sampling of natural product space to identify bioactive natural product-like polyketide macrolides
Polyketide macrolides are of interest for drug discovery but their inherent structural and stereochemical complexity hinders the exploration of related regions of chemical space more broadly. Here, the authors designed in silico and synthesized a library of tetrahydrofuran-containing polyketide macrolides, and screened them against a panel of biological assays, identifying biologically active library members.
- Darryl M. Wilson
- , Daniel J. Driedger
- & Robert A. Britton
-
Article
| Open Access Biosynthesis of the highly oxygenated tetracyclic core skeleton of Taxol
Biosynthesis of the highly oxygenated tetracyclic core skeleton of Taxol
Despite intensive investigation, stepwise reactions from diol to Taxol tetracyclic core skeleton remain unclear. Here, authors fill this gap by identifying two P450s and confirming the reaction order.
- Chengshuai Yang
- , Yan Wang
- & Zhihua Zhou
-
Article
| Open Access Reconstitution of early paclitaxel biosynthetic network
Reconstitution of early paclitaxel biosynthetic network
Paclitaxel is an important anticancer drug whose biosynthetic pathway reconstruction is hindered by the propensity of heterologously expressed pathway cytochromes P450, including taxadiene 5α-hydroxylase (T5αH), to form multiple products. Here, the authors tune the promoter strength for T5αH expression in Nicotiana plants to increase the levels of paclitaxel precursor taxadien-5α-ol by three-fold and reconstitute the six step early biosynthetic pathway of paclitaxel.
- Jack Chun-Ting Liu
- , Ricardo De La Peña
- & Elizabeth S. Sattely
-
Article
| Open Access Collective total synthesis of stereoisomeric yohimbine alkaloids
Collective total synthesis of stereoisomeric yohimbine alkaloids
Nature generates the stereoisomeric yohimbine alkaloids using bioavailable monoterpene secolaganin as the building block. Here, the authors reset the stage by the development of a bioinspired coupling, in which the pentacyclic skeleton is constructed through enantioselective kinetic resolution of an achiral, easily accessible synthetic surrogate.
- Meiyi Tang
- , Haigen Lu
- & Liansuo Zu
-
Article
| Open Access Elucidation of unusual biosynthesis and DnaN-targeting mode of action of potent anti-tuberculosis antibiotics Mycoplanecins
Elucidation of unusual biosynthesis and DnaN-targeting mode of action of potent anti-tuberculosis antibiotics Mycoplanecins
Mycoplanecins show promising activity against tuberculosis. Here, the authors identify and study mycoplanecins’ biosynthesis, antibacterial effects, and binding mechanism to DnaN, suggesting potential for fighting multidrug-resistant tuberculosis.
- Chengzhang Fu
- , Yunkun Liu
- & Rolf Müller
-
Article
| Open Access Total syntheses of Tetrodotoxin and 9-epiTetrodotoxin
Total syntheses of Tetrodotoxin and 9-epiTetrodotoxin
Tetrodotoxin and congeners are specific voltage-gated sodium channel blockers that exhibit remarkable anesthetic and analgesic effects but total synthesis procedures are often limited by the scale. Here, the authors present a scalable asymmetric syntheses of Tetrodotoxin and 9-epiTetrodotoxin from the abundant chemical feedstock furfuryl alcohol.
- Peihao Chen
- , Jing Wang
- & Xiangbing Qi
-
Article
| Open Access C5 methylation confers accessibility, stability and selectivity to picrotoxinin
C5 methylation confers accessibility, stability and selectivity to picrotoxinin
Minor changes to complex structures can exert major influences on synthesis strategy and functional properties but synthetic difficulties can obstruct the exploration of natural product function. Here the authors explore two parallel series of picrotoxinin analogs and identify leads with selectivity between mammalian and insect ion channels.
- Guanghu Tong
- , Samantha Griffin
- & Ryan A. Shenvi
-
Article
| Open Access A naturally occurring polyacetylene isolated from carrots promotes health and delays signatures of aging
A naturally occurring polyacetylene isolated from carrots promotes health and delays signatures of aging
Ameliorating or preventing signatures of aging in humans using natural compounds is an exciting area of research. Here the authors isolate a previously unknown phytochemical from carrots which activates defence mechanisms against oxidative stress and extends lifespan in worms, and improves glucose metabolism, promotes exercise capacity, and protects from frailty at higher age in mice.
- Carolin Thomas
- , Reto Erni
- & Michael Ristow
-
Article
| Open Access Discovery of type II polyketide synthase-like enzymes for the biosynthesis of cispentacin
Discovery of type II polyketide synthase-like enzymes for the biosynthesis of cispentacin
Type II polyketide synthases (PKSs) normally synthesize polycyclic aromatic compounds, but the potential for the synthesis of further diverse skeletons remains under investigated. Here, the authors report the discovery of the type II PKS machinery for the biosynthesis of a five-membered nonaromatic skeleton contained in the nonproteinogenic amino acid cispentacin and the plant toxin coronatine.
- Genki Hibi
- , Taro Shiraishi
- & Tomohisa Kuzuyama
-
Article
| Open Access Synthesis of tertiary alkylphosphonate oligonucleotides through light-driven radical-polar crossover reactions
Synthesis of tertiary alkylphosphonate oligonucleotides through light-driven radical-polar crossover reactions
Alkylphosphonates are used as chargeneutral replacements for naturally-occurring phosphodiester backbones in nucleotides but the choice of alkyl moieties that can be attached to phosphorus atoms in these compounds is limited to methyl groups or primary/secondary alkyls. Here, the authors demonstrate the tertiary alkylation of the phosphorus atoms of phosphites bearing two 2’-deoxynuclosides using a carbocation generated via a light-driven radical-polar crossover mechanism
- Kenji Ota
- , Kazunori Nagao
- & Hirohisa Ohmiya
-
Article
| Open Access Cu-catalyzed asymmetric regiodivergent electrosynthesis and its application in the enantioselective total synthesis of (-)-fumimycin
Cu-catalyzed asymmetric regiodivergent electrosynthesis and its application in the enantioselective total synthesis of (-)-fumimycin
Quaternary amino acids are important building blocks and precursors of medicinal compounds. Here, the authors describe a copper-catalyzed regiodivergent electrochemical core structure-oriented crossdehydrogenative coupling reaction of Schiff bases and commercially available hydroquinones to obtain three classes of chiral quaternary amino acid derivatives
- Tian Xie
- , Jianming Huang
- & Chang Guo
-
Article
| Open Access Triepoxide formation by a flavin-dependent monooxygenase in monensin biosynthesis
Triepoxide formation by a flavin-dependent monooxygenase in monensin biosynthesis
MonCI, a flavin-dependent monooxygenase, transforms all three C = C groups in the polyene substrate into epoxides during monensin A biosynthesis. Here, the authors present the structural basis for this enzyme’s regio- and stereoselective epoxidation activity.
- Qian Wang
- , Ning Liu
- & Chu-Young Kim
-
Article
| Open Access Catalytic enantioselective reductive alkynylation of amides enables one-pot syntheses of pyrrolidine, piperidine and indolizidine alkaloids
Catalytic enantioselective reductive alkynylation of amides enables one-pot syntheses of pyrrolidine, piperidine and indolizidine alkaloids
Saturated α-alkyl aza-heterocycles are found in a wide array of bioactive molecules. Here, the authors disclosed a one-pot, catalytic enantioselective synthesis of pyrrolidine, piperidine and indolizidine alkaloids from amides and alkynes.
- Fang-Fang Xu
- , Jin-Quan Chen
- & Pei-Qiang Huang
-
Article
| Open Access O-methyltransferase-like enzyme catalyzed diazo installation in polyketide biosynthesis
O-methyltransferase-like enzyme catalyzed diazo installation in polyketide biosynthesis
Diazo compounds, such as kinamycin, are rare bioactive natural products whose assembly has been extensively studied, but the formation of the diazo group is elusive. Here, the authors report O-methyltransferase-like protein, AlpH, which is responsible for the l-glutamylhydrazine incorporation in kinamycin biosynthesis.
- Yuchun Zhao
- , Xiangyang Liu
- & Ming Jiang
-
Article
| Open Access Molecular insights into the catalytic promiscuity of a bacterial diterpene synthase
Molecular insights into the catalytic promiscuity of a bacterial diterpene synthase
Diterpene synthase VenA catalyses the synthesis of venezuelaene A with a unique 5-5-6-7 tetracyclic skeleton from geranylgeranyl pyrophosphate. Here, the authors report crystal structures of apo- and holo-VenA, provide mechanistic insights into its substrate selectivity and promiscuity, and engineer VenA into a sesterterpene synthase.
- Zhong Li
- , Lilan Zhang
- & Shengying Li
-
Article
| Open Access Discovery of extended product structural space of the fungal dioxygenase AsqJ
Discovery of extended product structural space of the fungal dioxygenase AsqJ
The fungal dioxygenase AsqJ catalyses the conversion of benzo[1,4]diazepine-2,5-diones into quinolone antibiotics, and can also catalyse the production of quinazolinones. Here, the authors perform comprehensive substrate promiscuity mapping of AsqJ revealing its large tolerance towards various substrates, giving biocatalytic access to a plethora of biomedically valuable heterocyclic frameworks.
- Manuel Einsiedler
- & Tobias A. M. Gulder
-
Article
| Open Access A flavin-monooxygenase catalyzing oxepinone formation and the complete biosynthesis of vibralactone
A flavin-monooxygenase catalyzing oxepinone formation and the complete biosynthesis of vibralactone
Vibralactone is a strong lipase inhibitor and a bicyclic β-lactone containing an oxepinone ring, whose biosynthetic construction was unknown. Here the authors identify an oxepinone-building flavin monooxygenase VibO that is involved in the biosynthesis of vibralactone, and determine its X-ray crystal structure.
- Ke-Na Feng
- , Yue Zhang
- & Ying Zeng
-
Article
| Open Access Discovery and biosynthesis of tricyclic copper-binding ribosomal peptides containing histidine-to-butyrine crosslinks
Discovery and biosynthesis of tricyclic copper-binding ribosomal peptides containing histidine-to-butyrine crosslinks
Cyclic peptides are important bioactive compounds and drugs, synthesised by enzymatic side-chain macrocyclization of ribosomal peptides, which rarely involves histidine residues. Here, the authors report the discovery and biosynthesis of tricyclic lanthipeptide noursin, constrained by a tri amino acid labionin crosslink and histidine-to-butyrine crosslink, which is important for copper binding of noursin.
- Yuqing Li
- , Yeying Ma
- & Huan Wang
-
Article
| Open Access Unveiling an indole alkaloid diketopiperazine biosynthetic pathway that features a unique stereoisomerase and multifunctional methyltransferase
Unveiling an indole alkaloid diketopiperazine biosynthetic pathway that features a unique stereoisomerase and multifunctional methyltransferase
Diketopiperazine (DKP) natural products have diverse structures and biological functions. Here, the authors elucidate the biosynthetic pathway for indole alkaloid DKP nocardioazine B which includes DKP stereoisomerization by an unusual aspartate/glutamate racemase homolog and N- and C-methylation by a dual function methyltransferase.
- Garrett Deletti
- , Sajan D. Green
- & Amy L. Lane
-
Perspective
| Open Access A roadmap to establish a comprehensive platform for sustainable manufacturing of natural products in yeast
A roadmap to establish a comprehensive platform for sustainable manufacturing of natural products in yeast
Yeasts, Saccharomyces cerevisiae and Pichia pastoris, are promising chassis for the production of nature products (NPs). Here, the author discusses establishing a comprehensive platform for sustainable production of NPs via system-associated optimization at genetics, temporal controllers, productivity screening, and scalability levels.
- Gita Naseri
-
Article
| Open Access An α-chloroaldehyde-based formal synthesis of eribulin
An α-chloroaldehyde-based formal synthesis of eribulin
Eribulin is an anticancer drug and the most structurally complex non-peptidic drug made by chemical synthesis. Here, the authors report a streamlined synthesis of eribulin that creates new opportunities for drug discovery and development.
- Anissa Kaghad
- , Dimitrios Panagopoulos
- & Robert Britton
-
Article
| Open Access Genome mining unveils a class of ribosomal peptides with two amino termini
Genome mining unveils a class of ribosomal peptides with two amino termini
RiPP discovery has expanded the scope of post-translational modification chemistry, but genome mining of RiPP classes remains an unsolved challenge. Here, the authors employed bioinformatics and synthetic biology approaches to discover and characterize an unknown class of RiPPs, defined by an unusual amino-modified C-terminus.
- Hengqian Ren
- , Shravan R. Dommaraju
- & Huimin Zhao
-
Article
| Open Access Decrypting the programming of β-methylation in virginiamycin M biosynthesis
Decrypting the programming of β-methylation in virginiamycin M biosynthesis
Biosynthesis of complex polyketides by polyketide synthases often relies on trans-acting enzymes to modify the intermediates. Here, the authors elucidate how β-methylation enzymes identify their substrates. The recognition is imperfect, resulting in a doubly β-methylated virginiamycin derivative.
- Sabrina Collin
- , Russell J. Cox
- & Arnaud Gruez
-
Article
| Open Access Enzyme-like polyene cyclizations catalyzed by dynamic, self-assembled, supramolecular fluoro alcohol-amine clusters
Enzyme-like polyene cyclizations catalyzed by dynamic, self-assembled, supramolecular fluoro alcohol-amine clusters
Terpene cyclases enable the synthesis of (poly)cyclic carbon frameworks via ring closure of linear polyenes. Here, the authors report in-situ formed fluorinated-alcohol-amine supramolecular clusters that mimic terpene cyclases for shape-controlled polyene cyclizations.
- Andreas M. Arnold
- , Philipp Dullinger
- & Tanja Gulder
-
Article
| Open Access Insights into azalomycin F assembly-line contribute to evolution-guided polyketide synthase engineering and identification of intermodular recognition
Insights into azalomycin F assembly-line contribute to evolution-guided polyketide synthase engineering and identification of intermodular recognition
Intermodular recognition in polyketide synthase (PKS) is a key prerequisite for catalysis and assembly-line engineering. Here, the authors present a specific domain interaction between modules and an evolution-oriented strategy for PKS engineering derived from the enoylreduction module of azalomycin F.
- Guifa Zhai
- , Yan Zhu
- & Yuhui Sun
-
Article
| Open Access Uncovering a miltiradiene biosynthetic gene cluster in the Lamiaceae reveals a dynamic evolutionary trajectory
Uncovering a miltiradiene biosynthetic gene cluster in the Lamiaceae reveals a dynamic evolutionary trajectory
A diterpenoid biosynthetic gene cluster (BGC) has been identified in a few species in the Lamiaceae (mint) family, but its origin and evolution remain unclear. Here, the authors report assembly of genomes of three species within the family and reveal the dynamic evolutionary trajectory of the BGC.
- Abigail E. Bryson
- , Emily R. Lanier
- & Björn Hamberger
-
Article
| Open Access Synthesis of macrocyclic nucleoside antibacterials and their interactions with MraY
Synthesis of macrocyclic nucleoside antibacterials and their interactions with MraY
MraY is a membrane enzyme required for bacterial cell wall synthesis. Here, the authors modify sphaerimicins as antibacterials targeting it via structure-based design and synthesis through two key reactions, showing a platform for further development of MraY inhibitors as antibacterials.
- Takeshi Nakaya
- , Miyuki Yabe
- & Satoshi Ichikawa
-
Article
| Open Access Biosynthesis of mushroom-derived type II ganoderic acids by engineered yeast
Biosynthesis of mushroom-derived type II ganoderic acids by engineered yeast
The biosynthetic pathway of type II ganoderic acids (GAs) in Ganoderma lucidum, a traditional medicinal mushroom, is unknown. Here, the authors assemble the genome of type II GAs accumulating accession, identify CYPs involving in type II GAs biosynthesis, and achieve their production in engineered baker’s yeast.
- Wei Yuan
- , Chenjian Jiang
- & Han Xiao
-
Article
| Open Access Organocatalyst-mediated five-pot synthesis of (–)-quinine
Organocatalyst-mediated five-pot synthesis of (–)-quinine
Syntheses of quinine have fascinated organic chemists for over a hundred years. Here, the authors developed an organocatalyst-mediated pot-economical enantioselective total synthesis using five reaction vessels.
- Takahiro Terunuma
- & Yujiro Hayashi
-
Article
| Open Access Modular total syntheses of trans-clerodanes and sesquiterpene (hydro)quinones via tail-to-head cyclization and reductive coupling strategies
Modular total syntheses of trans-clerodanes and sesquiterpene (hydro)quinones via tail-to-head cyclization and reductive coupling strategies
trans-Clerodanes and sesquiterpene (hydro)quinones are different types of natural products featuring the same trans-decalin core. Here, the authors report a modular synthetic route to this family by tail-to-head cyclization and reductive coupling strategies.
- Wenming Zhu
- , Qishuang Yin
- & Ming Yang
-
Article
| Open Access The occurrence of ansamers in the synthesis of cyclic peptides
The occurrence of ansamers in the synthesis of cyclic peptides
The occurrence of isomers of the bicyclic octapeptide α-amanitin, which presents a macrolactam with a tryptathionine cross-link forming a handle, has been reported under the term of atropoisomers. Here, the authors synthesize its analogs and analyse their isomers, proposing and describing for them the term ansamer.
- Guiyang Yao
- , Simone Kosol
- & Roderich D. Süssmuth
-
Article
| Open Access Collective total synthesis of C4-oxygenated securinine-type alkaloids via stereocontrolled diversifications on the piperidine core
Collective total synthesis of C4-oxygenated securinine-type alkaloids via stereocontrolled diversifications on the piperidine core
A general synthetic solution toward high-oxidation state securinega alkaloids could be useful for systematic biological studies. Here, the authors report a divergent synthesis of C4-oxygenated securinega alkaloids via stereocontrolled diversifications on the piperidine core.
- Sangbin Park
- , Gyumin Kang
- & Sunkyu Han
-
Article
| Open Access Chimeric natural products derived from medermycin and the nature-inspired construction of their polycyclic skeletons
Chimeric natural products derived from medermycin and the nature-inspired construction of their polycyclic skeletons
Nonenzymatic reactions play an important part in the formation of some natural products possessing complex skeletons. Here, the authors report the discovery of eight chimeric medermycin-type natural products and their nonenzymatic construction.
- Shupeng Yin
- , Zhi Liu
- & Peng Fu
-
Article
| Open Access Tripterygium wilfordii cytochrome P450s catalyze the methyl shift and epoxidations in the biosynthesis of triptonide
Tripterygium wilfordii cytochrome P450s catalyze the methyl shift and epoxidations in the biosynthesis of triptonide
How triptonide is made in the medicinal plant Tripterygium wilfordii is largely unknown. Here, the authors report the identification and characterization of a suite of cytochrome P450s and show their function in catalyzing the formation of triptonide from miltriadiene in tobacco and baker’s yeast.
- Nikolaj Lervad Hansen
- , Louise Kjaerulff
- & Johan Andersen-Ranberg
-
Article
| Open Access A cryptic third active site in cyanophycin synthetase creates primers for polymerization
A cryptic third active site in cyanophycin synthetase creates primers for polymerization
Cyanophycin synthetase CphA1 polymerizes Asp and Arg into the nitrogen reserve polymer cyanophycin using two active sites. Sharon et al. show CphA1 has a cryptic 3rd active site that cleaves cyanophycin into primers for self-sufficient biosynthesis.
- Itai Sharon
- , Sharon Pinus
- & T. Martin Schmeing
-
Article
| Open Access Taming the radical cation intermediate enabled one-step access to structurally diverse lignans
Taming the radical cation intermediate enabled one-step access to structurally diverse lignans
Lignans, in spite of their structural diversity, are all biosynthetically derived from coniferyl alcohol. Here, the authors describe a divergent and stereoselective chemical synthesis of three types of lignans from biomass-derived monolignols, exploiting the different reaction manifolds of highly reactive radical cation intermediate.
- Jia-Chen Xiang
- , Cédric Fung
- & Jieping Zhu
-
Article
| Open Access An efficient approach to angular tricyclic molecular architecture via Nazarov-like cyclization and double ring-expansion cascade
An efficient approach to angular tricyclic molecular architecture via Nazarov-like cyclization and double ring-expansion cascade
Effective methods for accessing polycarbocycles are scarcely reported and generally involve complex, multistep transformations, thus restricting practical utility. Here, the authors construct tri-to-penta- cyclic frameworks containing at least two quaternary carbon centers via a tandem Nazarov-like cyclization / double cycloexpansion of simple 1,3-dicyclobutylidene ketone substrates.
- Yun-Peng Wang
- , Kun Fang
- & Tian Ke
-
Article
| Open Access Structure units oriented approach towards collective synthesis of sarpagine-ajmaline-koumine type alkaloids
Structure units oriented approach towards collective synthesis of sarpagine-ajmaline-koumine type alkaloids
The sarpagine-ajmaline-koumine type alkaloids are among the most important groups of monoterpenoid indole alkaloids, having notable biological activity. Here the authors perform divergent syntheses of 14 natural products in this family from a common intermediate.
- Wen Chen
- , Yonghui Ma
- & Hongbin Zhang
-
Article
| Open Access Reconstituting the complete biosynthesis of D-lysergic acid in yeast
Reconstituting the complete biosynthesis of D-lysergic acid in yeast
The ergot alkaloids are a class of natural products known for their pharmacologically privileged molecular structure that are used in the treatment of neurological ailments. Here the authors report on the production of the ergot (fungus)-derived therapeutic precursor, D-lysergic acid (DLA), in baker’s yeast.
- Garrett Wong
- , Li Rong Lim
- & Wen Shan Yew
-
Article
| Open Access Removal of lycopene substrate inhibition enables high carotenoid productivity in Yarrowia lipolytica
Removal of lycopene substrate inhibition enables high carotenoid productivity in Yarrowia lipolytica
Substrate inhibition has not been widely studied in the context of synthetic biology and metabolic engineering. Here, the authors report removal of lycopene substrate inhibition by two different strategies and enable high carotenoid productivity in Yarrowia lipolytica.
- Yongshuo Ma
- , Nian Liu
- & Gregory Stephanopoulos
-
Article
| Open Access Structures and function of a tailoring oxidase in complex with a nonribosomal peptide synthetase module
Structures and function of a tailoring oxidase in complex with a nonribosomal peptide synthetase module
Nonribosomal peptide synthetases work with additional enzymes to synthesise secondary metabolites and therapeutics. Here, the authors explore bacillamide D synthesis and show the oxidase action is done while the intermediate is attached to the synthetase and replicate this with an oxidase bound synthetase for bioengineering applications.
- Camille Marie Fortinez
- , Kristjan Bloudoff
- & T. Martin Schmeing
-
Article
| Open Access Computation-guided asymmetric total syntheses of resveratrol dimers
Computation-guided asymmetric total syntheses of resveratrol dimers
Although computational simulation-based natural product syntheses are in their initial stages of development, this concept can potentially become an indispensable resource in the field of organic synthesis. Here the authors report asymmetric total syntheses of several resveratrol dimers based on a comprehensive computational simulation of their biosynthetic pathways.
- Masaya Nakajima
- , Yusuke Adachi
- & Tetsuhiro Nemoto
-
Article
| Open Access Synthesis and structure elucidation of the human tRNA nucleoside mannosyl-queuosine
Synthesis and structure elucidation of the human tRNA nucleoside mannosyl-queuosine
Mannosyl-queuosine (manQ) is a non-canonical RNA nucleoside present in the anticodon loop of certain tRNAs. Here, the authors use a combination of total synthesis and mass spectrometry to contradict the literature-reported structure and show that manQ features an alpha-allyl connectivity of its mannose moiety.
- Markus Hillmeier
- , Mirko Wagner
- & Thomas Carell
-
Article
| Open Access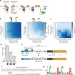 Selection for constrained peptides that bind to a single target protein
Selection for constrained peptides that bind to a single target protein
Peptide secondary metabolites have a diverse range of functions. Here the authors present a method to design and screen a large library of modified peptides in E. coli against a target of interest.
- Andrew M. King
- , Daniel A. Anderson
- & Christopher A. Voigt
-
Article
| Open Access Thiocysteine lyases as polyketide synthase domains installing hydropersulfide into natural products and a hydropersulfide methyltransferase
Thiocysteine lyases as polyketide synthase domains installing hydropersulfide into natural products and a hydropersulfide methyltransferase
Enzymes installing an intact hydropersulfide (-SSH) group into natural products have so far not been identified. Here, the authors report the characterization of an S-adenosyl methionine-dependent hydropersulfide methyltransferase (GnmP) for guangnanmycin biosynthesis, and identification of three SH domains within several NRPS-PKS assembly lines as thiocysteine lyases.
- Song Meng
- , Andrew D. Steele
- & Ben Shen
-
Article
| Open Access Facile generation of bridged medium-sized polycyclic systems by rhodium-catalysed intramolecular (3+2) dipolar cycloadditions
Facile generation of bridged medium-sized polycyclic systems by rhodium-catalysed intramolecular (3+2) dipolar cycloadditions
The bridged medium-sized ring bicyclo[m.n.2] family of natural products are commonly found but difficult to synthesize efficiently. Here the authors present a cascade reaction to form the carbon skeleton, via a [3+2] cycloaddition of a captured azavinyl carbene intermediate.
- Bao-Long Hou
- , Jonathan J. Wong
- & Chuang-Chuang Li
-
Article
| Open Access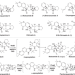 Structural basis of the stereoselective formation of the spirooxindole ring in the biosynthesis of citrinadins
Structural basis of the stereoselective formation of the spirooxindole ring in the biosynthesis of citrinadins
Prenylated indole alkaloids contain spirooxindole rings with a 3R or 3S carbon stereocenter, which determines their bioactivities, but the biocatalytic mechanism controlling the 3R- or 3S-spirooxindole formation was unclear. Here, the authors report the biochemical and structural characterization of the oxygenase/semipinacolase CtdE that catalyses the 3S-spirooxindole construction in the biosynthesis of 21R-citrinadin A.
- Zhiwen Liu
- , Fanglong Zhao
- & Xue Gao
-
Article
| Open Access Highly selective and robust single-atom catalyst Ru1/NC for reductive amination of aldehydes/ketones
Highly selective and robust single-atom catalyst Ru1/NC for reductive amination of aldehydes/ketones
Single-atom catalyst (SAC) has emerged as a frontier in heterogeneous catalysis yet its robustness remains a critical concern. Here, a highly active, selective and robust Ru1-N3 SAC is explored for a challenging reaction, reductive amination of aldehydes/ketones for synthesis of primary amines.
- Haifeng Qi
- , Ji Yang
- & Tao Zhang
-
Article
| Open Access Flavin-dependent halogenases catalyze enantioselective olefin halocyclization
Flavin-dependent halogenases catalyze enantioselective olefin halocyclization
Catalytic enantioselective halocyclization of alkenes is an important bond forming tool and a key step in natural product biosynthesis, but so far no examples of the enzymatic counterpart of this reaction on simple achiral olefins have been reported. Here, the authors describe examples of engineered flavin-dependent halogenases that catalyze halolactonization of olefins with high enantioselectivity and near-native catalytic activity.
- Dibyendu Mondal
- , Brian F. Fisher
- & Jared C. Lewis

 Enantioselective total synthesis of (‒)-lucidumone enabled by tandem prins cyclization/cycloetherification sequence
Enantioselective total synthesis of (‒)-lucidumone enabled by tandem prins cyclization/cycloetherification sequence