Abstract
The control of regioselectivity in Heck-type reaction of unactivated alkenes represents a longstanding challenge due to several detachable hydrogens in β–H elimination step, which generally afford either one specific regioisomer or a mixture. Herein, a copper-catalyzed intermolecular Heck-type reaction of unactivated alkenes and N-fluoro-sulfonamides with divergent regioselectivities is reported. The complete switch of regioselectivity mainly depends on the choice of different additives. Employment of alcohol solvent gives access to vinyl products, while the addition of carboxylate leads to the formation of allylic products. In addition, exclusion of these two promoting factors results in β-lactams via a C–N reductive elimination. This protocol shows a broad substrate scope for both alkenes and structurally diverse N-fluoro-sulfonamides, producing the corresponding products with excellent regio- and stereoselectivities. Further control experiments and DFT calculations provide in-depth insights into the reaction mechanism, highlighting the distinct effect of the additives on a bidentate auxiliary-stabilized Cu(III) intermediate.
Similar content being viewed by others
Introduction
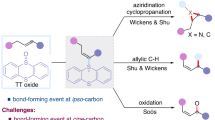
The intermolecular Mizoroki–Heck reaction is arguably one of the most significant methods for the construction of C–C bonds1,2,3,4,5,6. Electronically biased alkenes, such as styrene and acrylate derivatives are generally required to achieve both high reaction efficiency and excellent regioselectivity. In comparison, the coupling of unactivated alkenes has been far less investigated due to the intrinsic low reactivity of these olefins and the difficulty in controlling the linear regioselectivity7,8; the latter is attributed to the existence of several detachable β-hydrogens (Ha vs Hb) as well as the interconversion between vinyl and allylic products in the presence of fleeting metal hydride species (Fig. 1a)9,10. In the past few decades, unremitting efforts have been devoted to this field11,12. White et al. pioneered a substrate-chelated oxidative Heck reaction of phenylboronic acids and unactivated alkenes tethered with proximal heteroatoms for the regioselective synthesis of vinyl products13,14,15,16,17,18. Shortly thereafter, the Sigman group reported a catalyst-controlled β-H elimination process in both oxidative and classic Heck reactions of unactivated alkenes, giving good selectivity for the E-styrenyl products. But a mixture of vinyl and allylic products was obtained for γ-functionalized alkenes (R = CO2Me or Ph), and the ratios mainly depended on the electronic effect of the substituents on the aryl groups19,20,21. A significantly different Heck-type reaction was reported by the Gaunt group, wherein diaryliodonium salts coupled with unactivated alkenes via a carbocation intermediate, favoring unusual allylic products with moderate to good regio- and stereoselectivities22. Compared to aryl components, the intermolecular coupling of alkyl electrophiles and unactivated alkenes was previously regarded as an insurmountable obstacle8,23, considering the sluggish oxidative addition of low-valent transition metal to C(sp3) electrophiles and the facile β-H elimination24. This hurdle was overcome until very recently via electrophilic alkyl radicals stabilized by adjacent functionalities (e.g., F, ester, or cyano), pioneered by Buchwald25, Liu26, Wang27, Sodeoka28,29, Vincent30, Cho31,32, Gevorgyan33, Fu34, and Dong35. However, the lack of benzylic hydrogens renders the control of regioselectivity more challenging, and the preference for vinyl or allylic products heavily relies on different reaction partners (Fig. 1b). Despite the aforementioned encouraging advancements, only one specific regioisomer or a mixture of isomers are obtained in all successful cases; in contrast, a switch of regioselectivity in a single Heck operation of unactivated alkenes by simply adjusting the reaction conditions has not yet been realized.
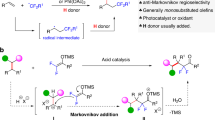
Our group has long sought alkyl electrophiles for the intermolecular Heck-type reactions of unactivated alkenes. In order to extend the scope of this reaction to commonly encountered nucleophilic alkyl radicals without the stabilization by an adjacent functionality, a bidentate directing group36,37,38,39 was employed in combination with copper salt to reduce π electron density of the electron-rich olefin moiety, making the polarity mismatched coupling of nucleophilic alkyl radicals with unactivated alkenes a reality40, and the vinyl products were obtained via concerted β–Hb elimination of the key Cu(III) intermediate A34,41. Nevertheless, a regioselectivity switch to Ha elimination remains a challenge (Fig. 2). Situated differently than the rotatable Hb, the location of Ha in a metallacyclic intermediate with locked conformation impedes the Ha elimination, both sterically and electronically42. Moreover, as a highly reactive species, the Cu(III) complex A is easy to undergo a reductive elimination to form C–N bond accompanying with the hydride elimination process43.
In recent years, the alkyl radicals generated from N-fluoro-amides via a sequence involving single-electron transfer (SET) with transition metal or photocatalyst, followed by 1,5-hydrogen atom transfer (HAT) have been well studied in the construction of C–C and C–X bonds44,45,46,47,48,49,50,51,52,53. However, the interaction of N-fluoro-amides with alkenes has rarely been explored. A seminal work was disclosed very recently by Li in benzylic radical-initiated difunctionalization of styrene derivatives54.
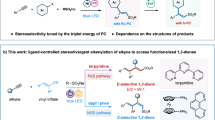
Herein, we report a copper-catalyzed intermolecular Heck-type reaction of unactivated alkenes and N-fluoro-sulfonamides with divergent regioselectivities by simply varying external additives. When the reactions were carried out in alcohol solvent, vinyl products B were selectively obtained via a hydroxyl-assisted concerted β–Hb elimination; while a complete regioselectivity switch to concerted β–Ha elimination (E2-type elimination) was achieved under acidic conditions in the presence of carboxylate, which accelerated the deprotonation of α-C–H through an amide-directed eight-membered metallacyclic transition state to deliver allylic products C. In addition, without these promoting factors, alternative β-lactams D could be formed through a reductive elimination of the Cu(III) intermediate A. Collectively, this protocol represented an actionable roadmap toward a broadly accessible solution to the assembly of branches of Heck-type reaction entities, with broad substrate scope for both terminal and internal alkenes, good E-stereoselectivity, and excellent functional-group tolerance.
Results
Screening of the reaction conditions
The study was initiated using 8-aminoquinoline (AQ) amide-tethered55 alkene 1a and N-fluoro-sulfonamides 2a bearing a δ-tertiary carbon as model substrates in the presence of Cu(TFA)2·H2O (Table 1). Screening began with an investigation of reaction solvent. For nonpolar solvent (entries 1–2), the reaction performed in DCE afforded reductive elimination product 5a in 42% yield, while in toluene a mixture of coupling products was obtained in 16% yield with allylic product 4a as the major isomer (r.r. = 1:3). Switching to polar solvent dioxane led to a significant boost in reductive elimination, providing β-lactam 5a in 73% (entry 3, as conditions C). However, CH3CN drastically attenuated the reaction profile, with the primary mass balance being unreacted starting material 1a and minor amounts of reductive elimination product 5a (entry 4). Delightedly, the reaction in NMP gave coupling products 3a and 4a as the major products with a good r.r. value of 10:1 (entry 5). The employment of alcohol as reaction solvent further benefitted the reaction efficiency and regioselectivity; in iBuOH, the vinyl product 3a was afforded in 65% yield in a highly regio- (r.r. > 20:1) and stereoselective (E/Z > 20:1) manner, and the reductive elimination process could be totally suppressed (entries 6–7). Then other copper salts were tested, and the results showed that Cu(OAc)2·H2O was the best choice to produce 3a in 72% yield, while Cu(NO3)2 proved to be completely ineffective (entries 8–11). Considering the important role of solvent for this reaction, some mixed solvents were evaluated (entries 12–14), among which an iBuOH/CH3CN combination could improve the yield to 82%, maintaining high regio- and E-stereoselectivities (r.r. > 20:1, E/Z > 20:1) (entry 14, as conditions A). The necessity of the copper catalyst for this transformation was demonstrated via a control experiment with the complete exclusion of copper salt, which only recovered the starting material 1a (entry 15). Other types of transition metals have also been tested, such as Ni(cod)2 and Co(acac)2, but no desired product could be detected (entries 16–17).
During the screening process, an intriguing result was observed when the reaction was conducted in toluene (entry 2). Despite low yield, the domination of 4a in coupling product distribution (r.r. = 1:3) meant the possibility of a regioselectivity switch to allylic products. Encouraged by this hypothesis, more nonpolar solvents were tested (entries 18–19), and the r.r. value could be elevated to 1:8 in benzene with a low overall yield of 15% for the coupling products. To improve the reaction efficiency, we envisaged that the weak α-acidity of the amide group might offer a handle to base-promoted E2-type elimination. In this context, we attempted the addition of a base to facilitate the concerted β–Ha elimination (entries 20–21). However, both the coupling and reductive elimination processes were completely suppressed; only the alkene substrate isomerization could be observed, and the α,β-unsaturated amide 1a′ was isolated in 90% yield in the presence of DBU. After an extensive screening, to our delight, metal carboxylates were found to be crucial to improve both the yield and r.r. value of the Heck-type reaction (entries 22–24). When the reaction was carried out in the presence of 0.4 equiv of Zn(OAc)2, allylic product 4a was obtained in 45% yield as a single regioisomer (r.r. > 20:1) along with the formation of β-lactam 5a in 32% yield. Increasing the loading of Zn(OAc)2 to 1.0 equiv partly suppressed the formation of β-lactam 5a (15%); however, the yield of the desired coupling product 4a (35%) did not increase accordingly and 28% of the alkene substrate was recovered (entry 25). The increased loading of Zn(OAc)2 accelerated the decomposition of N-fluoro-sulfonamide 2a (observed by TLC), which resulted in a decreased yield and a recovery of the alkene substrate. We also tested some other zinc salts, such as ZnBr2·TMEDA that has been used as an additive by Nakamura in iron-catalyzed C–H bond functionalization56, but it made little effect on the outcome (entry 26), showing a significant influence of the carboxylate on the reaction57. Further screening of the copper salts including Cu(OAc)2·H2O and Cu(CH3CN)4PF6 gave inferior yields of 4a, while alkene substrate 1a was unresponsive in the presence of other reductive metal catalyst44, e.g., Fe(OTf)2 (entries 27–29).
A major hurdle for the improvement of the yield of 4a was the competing reductive elimination pathway to β-lactam 5a. To overcome this issue, a variety of external additives were tested. Eventually, it was unexpectedly found that the addition of TFA would produce 4a in 62% yield, and the yield of β-lactam 5a decreased to 13% (entry 30). Increasing the amount of TFA from 1.0 to 2.0 equiv further improved the yield of 4a to 70%, and the reductive elimination process was completely supressed (entry 31, as conditions B). We have also tried to carry out the reaction with other Brønsted acids (entries 32–33). Whereas no better results were obtained, and when TfOH was added, the reaction system became messy with partial recovery of the alkene substrate (30%). It was noteworthy that treatment of alkenes bearing other bi- or monodentate auxiliaries rather than AQ amide58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75, 4-phenylbutene, or γ–δ unsaturated AQ amide under the conditions A–C all failed to give either coupling or reductive elimination products. The presence of AQ as a bidentate auxiliary could not only simulate the intermolecular reaction as an intramolecular variant to enhance the reactivity of nonactivated olefins, but also stabilize the high-valent copper species via a [5,5]-metallabicyclic intermediate to allow for further hydride elimination or reductive elimination. Moreover, we wondered whether an addition of external ligand would compete with AQ to coordinate with the metal center, and thus influence the reaction. In this context, several ligands were tested. To our delight, the external ligands had little effect on the regio- and diastereoselectivities of the Heck-type reactions, but the reaction yields decreased in varying degrees (for details, see Supplementary Fig. 1).
Substrate scope
With the optimized conditions in hand, the coupling reactions between various N-fluoro-sulfonamides and alkenyl amide 1a were investigated. As shown in Fig. 3, the N-fluoro-sulfonamides bearing tertiary δ-C–H bonds were first tested under conditions A or B to deliver the coupling products with quaternary carbon centers. The substituents on the phenyl ring of arylsulfonamide group had little impact on the reactivity to produce vinyl products 3b–3f under conditions A or allylic products 4b–4f under conditions B. Treatment of N-fluoro-2-naphthalene- or methane-sulfonamides with conditions A or B afforded 3g, 3h or 4g, 4h, respectively, with excellent regio- and E-stereoselectivities. The N-fluoro-sulfonamides containing branched alkyl chain were smoothly transformed to the corresponding vinyl products 3i–3m or allylic products 4i–4m. Of note, the tertiary δ-C–H bond could be selectively functionalized in the presence of a secondary δ′-C–H bond, affording products 3l or 4l in yields of 80 and 68%, respectively. Moreover, the coupling reactions were compatible with electronically diverse phenyl groups (3n, 3o or 4n, 4o) bearing either electron-donating (–OMe, 3p, 3s or 4p, 4s) or -withdrawing (–Br or –CF3, 3q, 3r or 4q, 4r) substituents. It should be mentioned that chlorine atom (3t or 4t), azide (3u or 4u), ether (3v or 4v), ester (3w, 3x or 4w, 4x), and cyclopropane (3y or 4y) were all well-tolerated without any sign of functional-group interconversion. Consistent with the aforementioned result, N-fluoro-sulfonamides underwent the coupling reactions preferentially at the tertiary δ-C–H bonds over the secondary ones (marked with an orange circle). Apart from δ,δ-dimethyl substituted fluorosulfonamides, the δ-methyl-δ-pentyl, δ-ethyl-δ-butyl, and δ,δ-cyclobutyl substituted N-fluoro-sulfonamides were found to couple with alkenyl amide 1a to afford vinyl products 3z–3ab or allylic products 4z–4ab. Subjection of N-fluoro-sulfonamides derived from lithocholic acid or Celecoxib to reaction conditions A produced 3ac and 3ad in 60 and 78% yields, respectively, while 4ac and 4ad were obtained in the yields of 52 and 67%, respectively, under reaction conditions B; showing the synthetic potential of this methodology for late-stage modification of bioactive molecules.
Conditions A (for vinyl coupling): 1a (0.20 mmol), 2 (0.50 mmol), and Cu(OAc)2·H2O (0.02 mmol) in a mixed iBuOH/CH3CN (2.5/0.5 mL) at 90 °C for 3 h. Conditions B (for allylic coupling): 1a (0.20 mmol), 2 (0.50 mmol), Cu(TFA)2·H2O (0.02 mmol), Zn(OAc)2 (0.08 mmol), and TFA (0.40 mmol) in benzene (2.0 mL) at 90 °C for 4 h. Isolated yields are shown. E/Z is >20:1 if not stated otherwise. TFA trifluoroacetic acid.
Subsequently, a variety of N-fluoro-sulfonamides bearing secondary δ-C–H bond were investigated under conditions D or B (Fig. 4). Both linear fluorosulfonamides with different alkyl chain lengths and the branched fluorosulfonamides worked well, and vinyl products 3ae–3aj were obtained as single regioisomers with moderate to good stereoselectivity (E/Z ranging from 5:1 to >20:1), while allylic products 4ae–4aj were isolated as E-isomers with excellent regioselectivity (r.r. ranging from 10:1 to >20:1). The secondary C–H bond could be activated prior to a primary one (marked with a purple circle) to give products 3ak or 4ak in yields of 65 and 62%, respectively. Benzylic δ-C–H bond proved to be available for the coupling reaction, giving rise to products 3al–3ap or 4al–4ap. With regard to the functional-group tolerance, all of the halogen atom, ester, and even terminal olefin could suffer from either conditions D or B to deliver vinyl products 3aq–3as or allylic products 4aq–4as, providing a useful handle for further manipulation. However, N-fluoro-sulfonamide bearing an azide group smoothly decomposed, and failed to give the corresponding allylic coupling product 4at under conditions B, a result different from that of 4u from tertiary δ-C–H bond. This may be attributed to the adverse effect of azide group on allylic coupling reactions (only 45% yield for 4u) as well as the less stability of secondary alkyl radical intermediates. Cyclic fluorosulfonamides with 5-, 6-, or even 7-membered ring could participate in the coupling reaction to provide 3au–3aw or 4au–4aw, and no diastereoselectivity was observed (d.r. = 2:1 or 1:1). When N-fluoro-sulfonamide derived from 1-C-ethylaminoadamantane was employed, coupling products 3ax or 4ax were obtained in 77 and 68% yields, respectively. For complex biologically relevant fluorosulfonamide, such as that from (+)-dehydroabietylamine, the coupling reaction occurred regioselectively at the C10 position to give 3ay or 4ay with complete diastereocontrol (d.r. > 20:1). Subjecting the N-fluoro-sulfonamide containing both secondary δ-C–H bond and tertiary ε-C–H bond to the coupling reaction conditions D or B led to a mixture of δ-coupling products 3az or 4az (via 1,5-HAT) and ε-coupling products 3az′ or 4az′ (via 1,6-HAT)76. Unfortunately, neither vinyl nor allylic product was observed for the reaction of N-butyl fluorosulfonamide, presumably due to the less stability of primary carbon radical77, which likely underwent decomposition prior to the addition to the olefin partner.
Conditions D (for vinyl coupling): 1a (0.20 mmol), 2 (0.50 mmol), and Cu(OAc)2·H2O (0.02 mmol) in iBuOH/dioxane (2.5/0.5 mL) at 90 °C for 3 h; conditions B (for allylic coupling): 1a (0.20 mmol), 2 (0.50 mmol), Cu(TFA)2·H2O (0.02 mmol), Zn(OAc)2 (0.08 mmol), and TFA (0.40 mmol) in benzene (2.0 mL) at 90 °C for 4 h. Isolated yields are shown. E/Z is >20:1 if not stated otherwise. TFA trifluoroacetic acid.
Encouraged by the aforementioned results, we then evaluated the scope of unactivated alkenes for the access to vinyl coupling products under slightly modified reaction conditions E, wherein isopropanol proved to be the best choice of solvent (Fig. 5). A variety of alkenyl amides bearing α-substituents were successfully converted into the corresponding vinyl products. α-Methyl or n-butyl substituted alkenes led to the formation of 6a and 6b in 84 and 88% yields, respectively, while bulky α-isopropyl substituent gave a diminished yield of 70% for 6c, all being produced as single regio- and E-stereoisomer. The alkenes bearing electronically diverse phenyl groups with either electron-donating (–OMe) or -withdrawing (–Br, –Cl) substituents provided excellent yields of the target vinyl products 6d–6g. The structure of 6d was further confirmed by X-ray crystallographic analysis (CCDC 2163753 contains the supplementary crystallographic data for compound 6d). Besides phenyl group, other aryl groups including naphthyl (6h, 6i) and thiophene-yl (6j) were compatible with the reaction conditions E. Significantly, the reaction proceeded well in the presence of diverse functionalities, such as acetal (6k), halogen atom (6l), ether (6m), cyclopropyl (6n), and even free hydroxyl (6o). For diene or enyne substrates, the β–γ double bond was selectively activated to produce 6p–6s in good yields, and the remaining γ–δ or δ–ε double bonds as well as the acetylene could serve as potential handles for further transformation. Subjection of the sterically hindered α,α-disubstituted alkenes to the reaction smoothly afforded 6t–6v in excellent yields, wherein the steric effect made little influence on the reaction outcome. For 1,1-disubstituted alkenes, the vinyl coupling products were obtained along with the regioisomers generated from β–elimination of the hydrogen atom on R2 alkyl chain, and the mixtures 6w (r.r. = 2:1) and 6x (r.r. = 2:3) were obtained in 82 and 65% yields, respectively. To our delight, the internal alkenes typically suffering from low substrate reactivity and poor product stereoselectivity78 proved to be suitable partners; both cyclic and Z-internal alkenes were transformed into the corresponding trisubstituted alkenes 6y–6aa with good reaction efficiency and excellent regio- and E-stereoselectivities.
Moving forward, we also tested the applicability of unactivated alkenes for the allylic coupling reaction under reaction conditions B. As shown in Fig. 6, the Z-internal alkene substrates could smoothly react with N-fluoro-sulfonamide 2a to produce the desired products 7a and 7b as single regio- and E-stereoisomer. For 1,1-disubstituted alkene, however, the reaction led to a slow decomposition of the alkene substrate, and the desired allylic coupling product 7c was not detected. It was noteworthy that when α-substituted alkene, for example, the α-methyl alkenyl amide was subjected to the reaction, product 7d was not obtained as expected. Instead, β-lactam 8 was isolated in 32% yield as a single trans-isomer with part of the alkene material recovered (30%). This may be attributed to the steric effect of the α substituent, which disfavors a coplanar transition state of the high-valent copper complex in concerted β–Ha elimination process, and thus alternative reductive elimination becomes the major reaction pathway to provide β-lactam 8 (for a detailed explanation by density functional theory (DFT) calculations, see Supplementary Fig. 2).
Conditions B: 1 (0.20 mmol), 2a (0.50 mmol), Cu(TFA)2·H2O (0.02 mmol), Zn(OAc)2 (0.08 mmol), and TFA (0.40 mmol) in benzene (2.0 mL) at 90 °C for 4 h. Conditions C: 1 (0.20 mmol), 2a (0.50 mmol), and Cu(TFA)2·H2O (0.02 mmol) in dioxane (3.0 mL) at 90 °C for 3 h. Isolated yields are shown. E/Z is >20:1 if not stated otherwise. TFA trifluoroacetic acid. aThe yield was recorded after a Boc-protection of the crude secondary amine product.
Of note, under reaction conditions C excluding the aforementioned promoting factors, the β-lactams 5 would dominate the product distribution through a reductive elimination of the high-valent copper intermediate79, a process previously observed independently by Zhao80,81, He/Chen/Wang82,83, and Quan/Liang/Wang43 in alkylamination of unactivated alkenes. As demonstrated in Fig. 6, we briefly investigated the scope for the synthesis of β-lactams. Both the α-substituted and internal alkenes could react with N-fluoro-sulfonamide 2a, and the corresponding β-lactams 5b–5e were obtained in yields ranging from 63 to 85% with excellent diastereoselectivity (d.r. > 20:1). Subsequently, alkenyl amide 1a was utilized as the alkene component and tested in combination with different N-fluoro-sulfonamides. Both linear and branched N-fluoro-sulfonamides bearing tertiary or secondary δ-C–H bonds were successfully converted into the corresponding β-lactams 5f–5j in good yields. This reaction showed good compatibility with the fluorosulfonamides derived from complex bioactive molecules. For example, treatment of the N-fluoro-sulfonamide prepared from (+)-dehydroabietylamine with alkenyl amide 1a under reaction conditions C provided the desired β-lactam 5k in 75% yield.
Control experiments
To better understand the reaction mechanism, several control experiments were performed (Fig. 7). The crossover experiments using N-fluoro-sulfonamides (2a or 2al) and the corresponding simple sulfonamides (9 or 10) were conducted under conditions A and D, respectively. No crossover products were observed (Fig. 7a). By employing NFSI as an external amidyl radical precursor84, we examined intermolecular coupling reactions between alkenyl amide 1a and simple sulfonamides 9 or 10 under conditions A and D, respectively, but neither vinyl coupling products 3a nor 3al were detected (Fig. 7b). These results could rule out the involvement of an intermolecular hydrogen atom abstraction process in the reaction. To further conform an intramolecular hydrogen migration process, deuterium-labeling experiments using external CD3OD were carried out with 1a and 2a under either conditions A′ or B′. As expected, no deuterium was incorporated into the vinyl or allylic coupling products (Fig. 7c).
a Crossover experiments. b With external amidyl radical precursor. c Deuterium-labeling experiments. d Radical trapping and radical clock experiments. e Evidence for no isomerization. f Extending to other alkyl electrophiles. TFA trifluoroacetic acid, NFSI N-fluorobenzenesulfonimide, TEMPO 2,2,6,6-tetramethyl-1-piperidinyloxy, BHT butylated hydroxytoluene.
When 3.0 equiv of either 2,2,6,6-tetramethylpiperidinooxy or butylated hydroxytoluene was added as a radical scavenger into the reaction of 1a and 2a, the desired coupling reactions were completely suppressed under either conditions A′′ or B′′. For radical clock experiments85, subjection of N-fluoro-sulfonamide 11 bearing a ε-ζ terminal double bond to the reaction with alkenyl amide 1a under conditions B generated diene 12, and the double bond migrated from ε-ζ to δ-ε position potentially via an π-allylic radical intermediate. Treatment of N-fluoro-sulfonamide 13 tethering a remote terminal double bond with 1a under conditions B provided allylic coupling product 14 in 63% yield via an intramolecular radical cyclization followed by an intermolecular Heck-type reaction. Moreover, when alkene substrate 15 containing a β-cyclopropyl moiety reacted with N-fluoro-sulfonamide 2a under conditions E, only trace amount of the desired coupling product 16 was detected, and instead acetate 17 and seven-membered lactam 18 were produced in 9 and 62% yields, respectively, via ring-opening of the cyclopropyl group. Taken together, these results demonstrated that the reaction involved alkyl radical intermediates (Fig. 7d).
Subjecting vinyl product 3a to either conditions A or B in the absence of N-fluoro-sulfonamide did not provide any allylic product 4a, only with the vast majority of the material recovered, which could exclude the possibility that the allylic products were generated from the corresponding vinyl products via olefin isomerization (Fig. 7e). Moreover, the present strategy could be further applied to other types of alkyl electrophiles. For example, the reaction of our previously employed cycloketone oxime ester41,83,86 with alkene 1a under conditions B delivered the desired allylic product 19 in 72% yield as a single regio- and E-stereoisomer, while β-lactam 20 was obtained in 68% yield under conditions C (Fig. 7f). Considering the distinct models to generate alkyl radicals from N-fluoro-sulfonamide and cycloketone oxime ester, these results have demonstrated that after initiating the SET process of radical precursors, the subsequent generation of alkyl radicals had limited effect on the following formation of Cu(III) intermediate and hydride elimination or reductive elimination.
Proposed reaction mechanism
According to the above control experiment results and precedent literature, a plausible mechanism for this intermolecular Heck-type reaction was proposed, as shown in Fig. 8. Firstly, the disproportionation reaction of Cu(II) catalyst generated a Cu(I) species87. A ligand exchange between Cu(I) catalyst and alkenyl amide 1a may occur to form complex I and release HX34. The coordination of C=C bond to Cu(I) center would reduce π electron density, and thus activate the olefin moiety. Subsequent SET of N-fluoro-sulfonamide 2a with copper complex I affords amidyl radical II and Cu(II) intermediate III88, and the released fluorine anion is proposed to be captured by the protons due to the strong interaction of H–F bond54,89,90. The resultant amidyl radical II is a high-energy species77,91,92, which undergoes an intramolecular HAT with remote δ-C(sp3)−H bond to form a carbon-centered radical IV93. Radical addition of IV to the olefin moiety of Cu(II) intermediate III results in the formation of a secondary carbon-centered radical, which can rapidly recombine with the nearby Cu(II) center to produce the key Cu(III) intermediate V34,41,43,45,46,49,50,53,80,81,82,83. At this point, there are three potential reaction pathways from V to provide different products depending on the choice of external additives. These three paths include a concerted β–Hb elimination (path 1) to form vinyl product 3a in alcohol solvent, a concerted β–Ha elimination (path 2) to afford allylic product 4a under acidic conditions in the presence of carboxylate, and a C–N reductive elimination (path 3) to generate β-lactam 5a without the aforementioned promoting factors. Along with the formation of the coupling or reductive elimination products, the Cu(I) catalyst can be regenerated to restart the catalytic cycle. Here, the origin of the complete switch of regioselectivity by the choice of different additives is worth investigating.
Theoretical calculations
To gain a deeper understanding of the divergent regioselectivities from Cu(III) species V, especially the key role of promoting factors, three possible reaction mechanisms with different additives have been evaluated through DFT calculations at the SMD94(iBuOH/benzene/dioxane)/(U)M0695/[6-311++G(d,p)/SDD96(Cu)]//SMD(iBuOH/benzene/dioxane)/(U)M06/[6-31G(d)/LanL2DZ97(Cu)] level (see the Supplementary Information for computational details). To ensure the rationality of the reaction mechanism prior to the regioselectivity-determining step from V, some elementary steps including ligand exchange, SET, 1,5-HAT, and radical addition were evaluated in alcohol solvent98. The DFT calculation results suggest that these four steps are accessible with the Gibbs free energy change (ΔG°) values of 3.7, –7.9, –5.8, and –27.2 kcal/mol, respectively (Fig. 8).
Figure 9 and Supplementary Fig. 3 show the Gibbs energy profiles of three regioselectivity-determining steps (concerted β–Ha elimination, concerted β–Hb elimination and C–N reductive elimination) under different reaction conditions. In the conditions A, iBuOH was employed as the reaction solvent based on entry 14 in Table 1. Initially, a Cu(III) complex 1′ can be formed through the ion exchange between counterion X of V and alkoxy group of iBuOH. As the most favorable path, the alkoxy-assisted concerted β–Hb elimination occurs through a five-membered-ring transition state TS1 to afford Cu(I) intermediate 2′ with the small Gibbs activation energy (ΔG°‡) and the large negative ΔG° values of 10.0 and −17.2 kcal/mol, respectively (Fig. 9a). The concerted β–Hb elimination can eventually lead to the formation of vinyl product 3a. In addition, we also located the alternative concerted β–Ha elimination and C–N reductive elimination, which lead to the allylic product 4a and the β-lactam product 5a, respectively. Although the participation of iBuOH can also promote these two reaction steps with the ΔG°‡ values of 24.9 and 21.0 kcal/mol, respectively, they are still much more unfavorable than the above-concerted β–Hb elimination.
In the conditions B, nonpolar benzene solvent was employed together with the presence of Zn(OAc)2 and TFA based on entry 29 in Table 1. A stable Cu(III) complex 5′ can be generated through the Lewis acid-base interaction between zinc and carbonyl (Fig. 9b). With the assistance of Zn(OAc)2, concerted β–Hb elimination (5′→TS4) and C–N reductive elimination (5′→TS6) from 5′ still require large ΔG°‡ values of 32.0 and 27.0 kcal/mol, respectively. However, the concerted β–Ha elimination easily occurs through an eight-membered-ring transition state TS5 to form an ionic Cu(I) complex 7′, which is eventually transformed into the allylic product 4a. The ΔG°‡ and ΔG° values of concerted β–Ha elimination are 18.1 and 2.2 kcal/mol, respectively. The calculation results suggest that Zn(OAc)2 additive promotes effectively the concerted β–Ha elimination. On one hand, as a Lewis acid, Zn(OAc)2 can reduce electron density of the electron-rich carbonyl group, improving the proton acidity of Ha. On the other hand, carboxylate anion can capture the protic Ha via a conformationally strained metallacyclic transition state. If omitting the assistance of Zn(OAc)2 in activating Ha, the energy barrier of concerted β–Hb elimination becomes larger by 12.1 kcal/mol (5′→TS7). The addition of TFA may accelerate the turnover of catalytic amount of Zn(OAc)2 via protonative demetalation, and thus suppress the competing reductive elimination process. It should be mentioned that when one Ha is replaced by an alkyl group, for example, a methyl, the concerted β–Ha elimination process switches to C–N reductive elimination (see Fig. 6). This is ascribed to the benefit of electron-donating alkyl group to reductive elimination, as well as the steric hindrance by α-methyl substitution, which is disadvantageous to a coplanar concerted β–Ha elimination process (for details, see Supplementary Fig. 2).
In the conditions C, dioxane was used as the reaction solvent, and the promoting factors in conditions A and B were excluded based on entry 3 in Table 1. Different from the above two conditions, the direct C–N reductive elimination becomes the most favorable reaction pathway in the absence of any external additives (for details, see Supplementary Fig. 3). Specifically, the C–N reductive elimination of 9′ occurs through a three-membered-ring transition state TS10 to form the three-coordinate Cu(I) intermediate 12′ with a ΔG°‡ value of 13.3 kcal/mol. Finally, the ligand exchange process can release the RE product 5a and regenerate the Cu(I) catalyst.
Overall, the complete switch of regioselectivity by the choice of different additives has been reproduced by the potential energy surface calculations. When alcohol solvent is employed, the alkoxy group can coordinate with a copper center, and act as a Brønsted base to facilitate the concerted β–Hb elimination, leading to an access to the vinyl coupling product. In contrast, when metal carboxylate is added to a nonpolar solvent, the metal can serve as a Lewis acid to reduce electron density of carbonyl moiety to increase the proton acidity of β–Ha, then achieves concerted β–Ha elimination through a carboxylate-participated eight-membered metallacyclic transition state to afford the allylic product. Moreover, the β-lactams dominate the product distribution through a favorable C–N reductive elimination of the Cu(III) intermediate in the absence of any aforementioned promoting factors.
Discussion
In summary, a divergent regioselective intermolecular Heck-type reaction was reported via a directed copper-catalyzed coupling of N-fluoro-sulfonamides and unactivated alkenes. The complete regioselectivity switch has been realized by simply varying the external additives. This protocol featured with broad substrate scope for both terminal and internal alkenes, excellent regio- and stereoselectivities, good functional-group tolerance, and a simple operational procedure. The N-fluoro-sulfonamides proceeded through SET with copper catalyst followed by an intramolecular HAT to generate the corresponding carbon-centered radical intermediates, which then participated in the intermolecular coupling with alkenes. Further control experiments and DFT calculations demonstrated the involvement of alkyl radical intermediates and illustrated a detailed reaction profile of the key Cu(III) intermediate, highlighting the distinct effect of the external additives. In alcohol solvent, a hydroxyl-assisted concerted β–Hb elimination led to the formation of vinyl coupling products, while the addition of metal carboxylate under acidic conditions resulted in a complete switch to concerted β–Ha elimination through a carboxylate-involved eight-membered metallacyclic transition state to deliver the allylic coupling products; without these promoting factors, C–N reductive elimination of the Cu(III) species dominated the product distribution to form β-lactams. Furthermore, the present strategy could be extended to other alkyl electrophiles, such as cycloketone oxime esters. This report reveals the major factors contributing to distinguishing the detachable β-hydrogens, offering a reference model for following research in Heck-type reaction.
Methods
Synthesis of vinyl product 3a
To a dry Schlenk flask were added 1a (42.4 mg, 0.20 mmol, 1.0 equiv), 2a (137 mg, 0.50 mmol, 2.5 equiv), Cu(OAc)2·H2O (4.0 mg, 0.02 mmol, 0.10 equiv), anhydrous iBuOH (2.5 mL), and anhydrous CH3CN (0.50 mL). The mixture was degassed for three times with argon and then stirred at 90 °C (oil bath) for 3 h. Once completion, the reaction was cooled to room temperature. The reaction mixture was filtered by celite, and the filtrate was concentrated in vacuo. Further purification by a flash column chromatography using eluents (PE/EA = 5:1) afforded the desired vinyl coupling product 3a as yellow oil (76.3 mg, 0.16 mmol, 82%, r.r. > 20:1, E/Z > 20:1).
Synthesis of allylic product 4a
To a dry Schlenk flask were added 1a (42.4 mg, 0.20 mmol, 1.0 equiv), 2a (137 mg, 0.50 mmol, 2.5 equiv), Cu(TFA)2·H2O (3.9 mg, 0.02 mmol, 0.10 equiv), Zn(OAc)2 (14.7 mg, 0.08 mmol, 0.40 equiv), TFA (30 uL, 0.40 mmol, 2.0 equiv), and anhydrous benzene (2.0 mL). The mixture was degassed for three times with argon and then stirred at 90 °C (oil bath) for 4 h. Once completion, the reaction was cooled to room temperature. The reaction mixture was filtered by celite, and the filtrate was concentrated in vacuo. Further purification by a flash column chromatography using eluents (PE/EA = 5:1) afforded the desired allylic product 4a as yellow oil (65.1 mg, 0.14 mmol, 70%, r.r. > 20:1, E/Z > 20:1).
Synthesis of β-lactam 5a
To a dry Schlenk flask were added 1a (42.4 mg, 0.20 mmol, 1.0 equiv), 2a (137 mg, 0.50 mmol, 2.5 equiv), Cu(TFA)2·H2O (3.9 mg, 0.02 mmol, 0.10 equiv), and anhydrous dioxane (3.0 mL). The mixture was degassed for three times with argon and then stirred at 90 °C (oil bath) for 3 h. Once completion, the reaction was cooled to room temperature. The reaction mixture was filtered by celite, and the filtrate was concentrated in vacuo. Further purification by a flash column chromatography using eluents (PE/EA = 5:1) afforded the desired β-lactam 5a as yellow oil (67.9 mg, 0.15 mmol, 73%).
Data availability
The authors declare that all data supporting the findings of this research are available within the article and its Supplementary Information. Cartesian coordinates of the calculated structures are available in Supplementary Data 1. Crystallographic data can be obtained free of charge for 6d (CCDC 2163753) from the Cambridge Crystallographic Data Centre (https://www.ccdc.cam.ac.uk/). Any further relevant data are available from the authors on request.
References
Mizoroki, T., Mori, K. & Ozaki, A. Arylation of olefin with aryl iodide catalyzed by palladium. Bull. Chem. Soc. Jpn. 44, 581–581 (1971).
Heck, R. F. & Nolley, J. P. Palladium-catalyzed vinylic hydrogen substitution reactions with aryl, benzyl, and styryl halides. J. Org. Chem. 37, 2320–2322 (1972).
Beletskaya, I. P. & Cheprakov, A. V. The Heck reaction as a sharpening stone of palladium catalysis. Chem. Rev. 100, 3009–3066 (2000).
Dounay, A. B. & Overman, L. E. The asymmetric intramolecular Heck reaction in natural product total synthesis. Chem. Rev. 103, 2945–2964 (2003).
Wang, S.-S. & Yang, G.-Y. Recent developments in low-cost TM-catalyzed Heck-type reactions (TM = transition metal, Ni, Co, Cu, and Fe). Catal. Sci. Technol. 6, 2862–2876 (2016).
Jagtap, S. Heck reaction-state of the art. Catalysts 7, 267 (2017).
Deb, A. & Maiti, D. Emergence of unactivated olefins for the synthesis of olefinated arenes. Eur. J. Org. Chem. 2017, 1239–1252 (2017).
Gao, S., Shi, L., Chang, L., Wang, B. & Fu, J. Recent developments in Heck-type reaction of unactivated alkenes and alkyl electrophiles. Synthesis 53, 861–878 (2021).
Bras, J. L. & Muzart, J. β-Elimination competitions leading to C=C bonds from alkylpalladium intermediates. Tetrahedron 68, 10065–10113 (2012).
Ruan, J. & Xiao, J. From α-arylation of olefins to acylation with aldehydes: a journey in regiocontrol of the Heck reaction. Acc. Chem. Res. 44, 614–626 (2011).
Heck, R. F. Electronic and steric effects in the olefin arylation and carboalkoxylation reactions with organopalladium compounds. J. Am. Chem. Soc. 93, 6896–6901 (1971).
Kikukawa, K., Nagira, K., Wada, F. & Matsuda, T. Reaction of diazonium salts with transition metals–III1: Palladium(0)-catalyzed arylation of unsaturated compounds with arenediazoium salts. Tetrahedron 37, 31–36 (1981).
Delcamp, J. H., Brucks, A. P. & White, M. C. A general and highly selective chelate-controlled intermolecular oxidative Heck reaction. J. Am. Chem. Soc. 130, 11270–11271 (2008).
Huffman, T. R., Wu, Y., Emmerich, A. & Shenvi, R. A. Intermolecular Heck coupling with hindered alkenes directed by potassium carboxylates. Angew. Chem. Int. Ed. 58, 2371–2376 (2019).
Romine, A. M., Yang, K. S., Karunananda, M. K., Chen, J. S. & Engle, K. M. Synthetic and mechanistic studies of a versatile heteroaryl thioether directing group for Pd(II) catalysis. ACS Catal. 9, 7626–7640 (2019).
Lv, H. et al. Nickel(0)-catalyzed intermolecular oxidative Heck arylation driven by transfer hydrogenation. Nat. Commun. 10, 5025 (2019).
Yang, S., Liu, L., Zhou, Z., Huang, Z. & Zhao, Y. Palladium-catalyzed direct C–H arylation of 3-butenoic acid derivatives. Org. Lett. 23, 296–299 (2021).
Landge, V. G. et al. Palladium-catalyzed γ,γ′-diarylation of free alkenyl amines. J. Am. Chem. Soc. 143, 10352–10360 (2021).
Werner, E. W. & Sigman, M. S. A highly selective and general palladium catalyst for the oxidative Heck reaction of electronically nonbiased olefins. J. Am. Chem. Soc. 132, 13981–13983 (2010).
Werner, E. W. & Sigman, M. S. Operationally simple and highly (E)-styrenyl-selective Heck reactions of electronically nonbiased olefins. J. Am. Chem. Soc. 133, 9692–9695 (2011).
Sigman, M. S. & Werner, E. W. Imparting catalyst control upon classical palladium-catalyzed alkenyl C−H bond functionalization reactions. Acc. Chem. Res. 45, 874–884 (2012).
Phipps, R. J., McMurray, L., Ritter, S., Duong, H. A. & Gaunt, M. J. Copper-catalyzed alkene arylation with diaryliodonium salts. J. Am. Chem. Soc. 134, 10773–10776 (2012).
Kurandina, D., Chuentragool, P. & Gevorgyan, V. Transition-metal-catalyzed alkyl Heck-type reactions. Synthesis 51, 985–1005 (2019).
Kwiatkowski, M. R. & Alexanian, E. J. Transition-metal (Pd, Ni, Mn)-catalyzed C−C bond constructions involving unactivated alkyl halides and fundamental synthetic building blocks. Acc. Chem. Res. 52, 1134–1144 (2019).
Parsons, A. T. & Buchwald, S. L. Copper-catalyzed trifluoromethylation of unactivated olefins. Angew. Chem. Int. Ed. 50, 9120–9123 (2011).
Xu, J. et al. Copper-catalyzed trifluoromethylation of terminal alkenes through allylic C–H bond activation. J. Am. Chem. Soc. 133, 15300–15303 (2011).
Wang, X. et al. Copper-catalyzed C(sp3)–C(sp3) bond formation using a hypervalent iodine reagent: an efficient allylic trifluoromethylation. J. Am. Chem. Soc. 133, 16410–16413 (2011).
Shimizu, R., Egami, H., Hamashima, Y. & Sodeoka, M. Copper-catalyzed trifluoromethylation of allylsilanes. Angew. Chem. Int. Ed. 51, 4577–4580 (2012).
Kawamura, S. & Sodeoka, M. Perfluoroalkylation of unactivated alkenes with acid anhydrides as the perfluoroalkyl source. Angew. Chem. Int. Ed. 55, 8740–8743 (2016).
Beniazza, R. et al. Copper(I)-photocatalyzed trifluoromethylation of alkenes. Chem. Commun. 51, 9571–9574 (2015).
Iqbal, N., Choi, S., Kim, E. & Cho, E. J. Trifluoromethylation of alkenes by visible light photoredox catalysis. J. Org. Chem. 77, 11383–11387 (2012).
Yu, C., Iqbal, N., Park, S. & Cho, E. J. Selective difluoroalkylation of alkenes by using visible light photoredox catalysis. Chem. Commun. 50, 12884–12887 (2014).
Kurandina, D., Rivas, M., Radzhabov, M. & Gevorgyan, V. Heck reaction of electronically diverse tertiary alkyl halides. Org. Lett. 20, 357–360 (2018).
Tang, C. et al. Directed copper-catalyzed intermolecular Heck-type reaction of unactivated olefins and alkyl halides. J. Am. Chem. Soc. 140, 16929–16935 (2018).
Wu, X., Riedel, J. & Dong, V. M. Transforming olefins into γ,δ-unsaturated nitriles through copper catalysis. Angew. Chem. Int. Ed. 56, 11589–11593 (2017).
Rouquet, G. & Chatani, N. Catalytic functionalization of C(sp2)–H and C(sp3)–H bonds by using bidentate directing groups. Angew. Chem. Int. Ed. 52, 11726–11743 (2013).
Daugulis, O., Roane, J. & Tran, L. D. Bidentate, monoanionic auxiliary-directed functionalization of carbon-hydrogen bonds. Acc. Chem. Res. 48, 1053–1064 (2015).
Rej, S., Ano, Y. & Chatani, N. Bidentate directing groups: an efficient tool in C–H bond functionalization chemistry for the expedient construction of C–C bonds. Chem. Rev. 120, 1788–1887 (2020).
Jeon, J., Lee, C., Park, I. & Hong, S. Regio- and stereoselective functionalization enabled by bidentate directing groups. Chem. Rec. 21, 3613–3627 (2021).
Wu, X. et al. Palladium-catalyzed intermolecular polarity-mismatched addition of unactivated alkyl radicals to unactivated alkenes. ACS Catal. 10, 14107–14116 (2020).
Deng, Y. et al. Directing-group-based strategy enabling intermolecular Heck-type reaction of cycloketone oxime esters and unactivated alkenes. Org. Lett. 22, 3524–3530 (2020).
Wang, Z., He, Z., Zhang, L. & Huang, Y. Iridium-catalyzed aerobic α,β-dehydrogenation of γ,δ-unsaturated amides and acids: activation of both α- and β‑C−H bonds through an allyl−iridium intermediate. J. Am. Chem. Soc. 140, 735–740 (2018).
Li, M. et al. Directed copper-catalyzed tandem radical cyclization reaction of alkyl bromides and unactivated olefins. Org. Lett. 24, 2738–2743 (2022).
Groendyke, B. J., AbuSalim, D. I. & Cook, S. P. Iron-catalyzed, fluoroamide-directed C−H fluorination. J. Am. Chem. Soc. 138, 12771–12774 (2016).
Li, Z., Wang, Q. & Zhu, J. Copper-catalyzed arylation of remote C(sp3)−H bonds in carboxamides and sulfonamides. Angew. Chem. Int. Ed. 57, 13288–13292 (2018).
Zhang, Z., Zhang, X. & Nagib, D. A. Chiral piperidines from acyclic amines via enantioselective, radical-mediated δ C−H cyanation. Chem 5, 3127–3134 (2019).
Liu, Z. et al. Copper-catalyzed remote C(sp3)−H trifluoromethylation of carboxamides and sulfonamides. Angew. Chem. Int. Ed. 58, 2510–2513 (2019).
Bafaluy, D. et al. Copper-catalyzed N–F bond activation for uniform intramolecular C–H amination to pyrrolidines and piperidines. Angew. Chem. Int. Ed. 58, 8912–8916 (2019).
Wang, C.-Y. et al. Enantioselective copper-catalyzed cyanation of remote C(sp3)–H bonds enabled by 1,5-hydrogen atom transfer. iScience 21, 490–498 (2019).
Zhang, Z.-H. et al. Copper-catalyzed enantioselective Sonogashira-type oxidative cross-coupling of unactivated C(sp3)–H bonds with alkynes. Nat. Commun. 10, 5689 (2019).
Guo, Q. et al. Visible-light promoted regioselective amination and alkylation of remote C(sp3)–H bonds. Nat. Commun. 11, 1463 (2020).
Wang, X. et al. Cu(II)-catalyzed N-directed distal C(sp3)–H heteroarylation of aliphatic N-fluorosulfonamides. Org. Lett. 24, 1055–1059 (2022).
Stateman, L. M., Dare, R. M., Paneque, A. N. & Nagib, D. A. Aza-heterocycles via copper-catalyzed, remote C–H desaturation of amines. Chem 8, 210–224 (2022).
Zhong, L.-J. et al. Intermolecular 1,2-difunctionalization of alkenes enabled by fluoroamide-directed remote benzyl C(sp3)−H functionalization. J. Am. Chem. Soc. 144, 339–348 (2022).
Zaitsev, V. G., Shabashov, D. & Daugulis, O. Highly regioselective arylation of sp3 C−H bonds catalyzed by palladium acetate. J. Am. Chem. Soc. 127, 13154–13155 (2005).
Shang, R., Ilies, L., Asako, S. & Nakamura, E. Iron-catalyzed C(sp2)–H bond functionalization with organoboron compounds. J. Am. Chem. Soc. 136, 14349–14352 (2014).
Tan, P. W., Mak, A. M., Sullivan, M. B., Dixon, D. J. & Seayad, J. Thioamide-directed cobalt(III)-catalyzed selective amidation of C(sp3)−H bonds. Angew. Chem. Int. Ed. 56, 16550–16554 (2017).
Gurak, J. A. Jr. & Engle, K. M. Regioselective hydroamination using a directed nucleopalladation/protodepalladation strategy. Synlett 28, 2057–2065 (2017).
Lin, C. & Shen, L. Recent progress in transition metal-catalyzed regioselective functionalization of unactivated alkenes/alkynes assisted by bidentate directing groups. ChemCatChem 11, 961–968 (2019).
Gurak, J. A. Jr., Yang, K. S., Liu, Z. & Engle, K. M. Directed, regiocontrolled hydroamination of unactivated alkenes via protodepalladation. J. Am. Chem. Soc. 138, 5805–5808 (2016).
Derosa, J., Tran, V. T., Boulous, M. N., Chen, J. S. & Engle, K. M. Nickel-catalyzed β,γ-dicarbofunctionalization of alkenyl carbonyl compounds via conjunctive cross-coupling. J. Am. Chem. Soc. 139, 10657–10660 (2017).
Tran, V. T., Gurak, J. A. Jr., Yang, K. S. & Engle, K. M. Activation of diverse carbon-heteroatom and carbon-carbon bonds via palladium(II)-catalysed β-X elimination. Nat. Chem. 10, 1126–1133 (2018).
Wang, H. et al. Palladium-catalyzed amidedirected enantioselective hydrocarbofunctionalization of unactivated alkenes using a chiral monodentate oxazoline ligand. J. Am. Chem. Soc. 140, 3542–3546 (2018).
Wang, C. et al. Palladium-catalyzed regiocontrollable reductive Heck reaction of unactivated aliphatic alkenes. J. Am. Chem. Soc. 140, 9332–9336 (2018).
Lv, H., Xiao, L.-J., Zhao, D. & Zhou, Q.-L. Nickel(0)-catalyzed linear-selective hydroarylation of unactivated alkenes and styrenes with aryl boronic acids. Chem. Sci. 9, 6839–6843 (2018).
Shen, H.-C. et al. Enantioselective addition of cyclic ketones to unactivated alkenes enabled by amine/Pd(II) cooperative catalysis. ACS Catal. 9, 791–797 (2019).
Schreib, B. S. & Carreira, E. M. Palladium-catalyzed regioselective C−H iodination of unactivated alkenes. J. Am. Chem. Soc. 141, 8758–8763 (2019).
Jeon, J. et al. Siteselective 1,1-difunctionalization of unactivated alkenes enabled by cationic palladium catalysis. J. Am. Chem. Soc. 141, 10048–10059 (2019).
Peng, J.-B. et al. Palladium-catalyzed regioselective carbonylative coupling/amination of aryl iodides with unactivated alkenes: efficient synthesis of β-aminoketones. ACS Catal. 9, 2977–2983 (2019).
Jeon, J., Lee, C., Seo, H. & Hong, S. NiH-catalyzed proximal-selective hydroamination of unactivated alkenes. J. Am. Chem. Soc. 142, 20470–20480 (2020).
Yang, T. et al. Chemoselective union of olefins, organohalides, and redox-active esters enables regioselective alkene dialkylation. J. Am. Chem. Soc. 142, 21410–21419 (2020).
Chen, X., Rao, W., Yang, T. & Koh, M. J. Alkyl halides as both hydride and alkyl sources in catalytic regioselective reductive olefin hydroalkylation. Nat. Commun. 11, 5857 (2020).
Lee, C., Seo, H., Jeon, J. & Hong, S. γ-Selective C(sp3)–H amination via controlled migratory hydroamination. Nat. Commun. 12, 5657 (2021).
Xie, L. et al. Directed nickel-catalyzed regio- and diastereoselective arylamination of unactivated alkenes. Nat. Commun. 12, 6280 (2021).
Jankins, T. C. et al. Low-valent tungsten redox catalysis enables controlled isomerization and cabonylative functionalization of alkenes. Nat. Chem. 14, 632–639 (2022).
Short, M. A., Blackburn, J. L. & Roizen, J. L. Modifying positional selectivity in C–H functionalization reactions with nitrogen-centered radicals: generalizable approaches to 1,6-hydrogen-atom transfer processes. Synlett 31, 102–116 (2020).
Luo, Y.-R. Handbook of Bond Dissociation Energies in Organic Compounds (CRC Press, 2003).
Nakashima, Y., Hirata, G., Sheppard, T. D. & Nishikata, T. The Mizoroki-Heck reaction with internal olefins: reactivities and stereoselectivities. Asian J. Org. Chem. 9, 480–491 (2020).
Hosseyni, S. & Jarrahpour, A. Recent advances in β-lactam synthesis. Org. Biomol. Chem. 16, 6840–6852 (2018).
Shi, P. et al. A practical copper-catalyzed approach to β-lactams via radical carboamination of alkenyl carbonyl compounds. Chem. Commun. 55, 10523–10526 (2019).
Gan, Z., Zhang, K., Shi, P., Zhao, Y. & Zeng, R. Copper(I)-catalyzed radical carboamination reaction of 8-aminoquinoline-oriented buteneamides with chloroform: synthesis of-β-lactams. RSC Adv. 11, 28081–28084 (2021).
Bai, Z. et al. Enantioselective alkylamination of unactivated alkenes under copper catalysis. J. Am. Chem. Soc. 143, 1195–1202 (2021).
Zhang, H. et al. β-Lactam synthesis via copper-catalyzed directed aminoalkylation of unactivated alkenes with cyclobutanone O-benzoyloximes. Org. Lett. 23, 3620–3625 (2021).
Zhang, W. et al. Enantioselective cyanation of benzylic C–H bonds via copper-catalyzed radical relay. Science 353, 1014–1018 (2016).
Griller, D. & Ingold, K. U. Free-radical clocks. Acc. Chem. Res. 13, 317–323 (1980).
Yang, D. et al. Directed cobalt-catalyzed anti-Markovnikov hydroalkylation of unactivated alkenes enabled by “Co–H” catalysis. Org. Lett. 22, 4333–4338 (2020).
Dong, J., Wang, F. & You, J. Copper-mediated tandem oxidative C(sp2)–H/C(sp)–H alkynylation and annulation of arenes with terminal alkynes. Org. Lett. 16, 2884–2887 (2014).
Muñoz-Molina, J. M., Belderrain, T. R. & Pérez, P. J. Recent advances in copper-catalyzed radical C–H bond activation using N–F reagents. Synthesis 53, 51–64 (2021).
Lee, J.-W. et al. Hydrogen-bond promoted nucleophilic fluorination: concept, mechanism and applications in positron emission tomography. Chem. Soc. Rev. 45, 4638–4650 (2016).
Liang, S., Hammond, G. B. & Xu, B. Hydrogen bonding: regulator for nucleophilic fluorination. Chem. Eur. J. 23, 17850–17861 (2017).
Xiong, T. & Zhang, Q. New amination strategies based on nitrogen-centered radical chemistry. Chem. Soc. Rev. 45, 3069–3087 (2016).
Kwon, K., Simons, R. T., Nandakumar, M. & Roizen, J. L. Strategies to generate nitrogen-centered radicals that may rely on photoredox catalysis: development in reaction methodology and applications in organic synthesis. Chem. Rev. 122, 2353–2428 (2022).
Chuentragool, P. et al. Aliphatic radical relay Heck reaction at unactivated C(sp3)−H sites of alcohols. Angew. Chem. Int. Ed. 58, 1794–1798 (2019).
Marenich, A. V., Cramer, C. J. & Truhlar, D. G. Universal solvation model based on solute electron density and on a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B. 113, 6378–6396 (2009).
Zhao, Y. & Truhlar, D. G. The M06 suite of density functionals for main group thermochemistry, thermochemical kinetics, noncovalent interactions, excited states, and transition elements: two new functionals and systematic testing of four M06-class functionals and 12 other functionals. Theor. Chem. Acc. 120, 215–241 (2008).
Andrae, D., Häußermann, U., Dolg, M., Stoll, H. & Preuß, H. Energy-adjusted ab initio pseudopotentials for the second and third row transition elements. Theor. Chem. Acc. 77, 123–141 (1990).
Hay, P. J. Ab initio effective core potentials for molecular calculations, potentials for K to Au including the outermost core orbitals. J. Chem. Phys. 82, 299–310 (1985).
Wang, R. et al. The role of AQ in the regioselectivity of strong alkyl C–O bond activation catalyzed by Pd(OAc)2: a density functional theory mechanistic study. Inorg. Chem. 60, 17555–17564 (2021).
Acknowledgements
We gratefully acknowledge the National Natural Science Foundation of China (21971034, for J.F.; 21702027, for J.F.; 22173016, for W.G.) and the Fundamental Research Funds for the Central Universities (2412019FZ017, for J.F.) for financial support.
Author information
Authors and Affiliations
Contributions
C.Z. and Y.L. designed and performed the experiments. Y.D. and W.G. performed the density functional theory calculations. M.L., D.X., and S.G. assisted in completing the experiments. Q.L. and Q.Z. participated in the data discussion. J.F. directed the project and wrote the manuscript. All the authors were involved in the interpretation of the results presented in the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Ming Lei, Jun-Long Niu, and Guozhu Zhang for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, C., Li, Y., Dong, Y. et al. Divergent regioselective Heck-type reaction of unactivated alkenes and N-fluoro-sulfonamides. Nat Commun 13, 6297 (2022). https://doi.org/10.1038/s41467-022-33996-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-022-33996-1
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.