News & Views |
Featured
-
-
Article |
 TBK1 and IKKε prevent TNF-induced cell death by RIPK1 phosphorylation
TBK1 and IKKε prevent TNF-induced cell death by RIPK1 phosphorylation
Lafont et al. uncover a checkpoint mediated by TBK1 and IKKε, which phosphorylate RIPK1 in the TNFR1-SC. TBK1 and IKKε recruitment depends on M1 ubiquitylation and NEMO to restrict TNF-induced cell death.
- Elodie Lafont
- , Peter Draber
- & Henning Walczak
-
News & Views |
 Metabolic control of ferroptosis in cancer
Metabolic control of ferroptosis in cancer
Ferroptosis is a regulated non-apoptotic form of cell death and its functional role in tumorigenesis remains elusive. A study now shows that the tumour suppressor BAP1 enhances ferroptosis by modulating expression of the cystine transporter SLC7A11, leading to improved control of tumour growth.
- Michael P. Murphy
-
Review Article |
 Life, death and autophagy
Life, death and autophagy
In this Review, Doherty and Baehrecke discuss the multiple roles of autophagy during cell survival and cell death. They cover the interplay between autophagy, apoptosis and necrosis, as well as engulfment and inflammation.
- Johnna Doherty
- & Eric H. Baehrecke
-
Article |
 BAP1 links metabolic regulation of ferroptosis to tumour suppression
BAP1 links metabolic regulation of ferroptosis to tumour suppression
Zhang et al. show that BAP1 suppresses SLC7A11 expression and cystine uptake, thereby promoting ferroptosis and inhibiting tumour growth.
- Yilei Zhang
- , Jiejun Shi
- & Boyi Gan
-
Letter |
 EFF-1 fusogen promotes phagosome sealing during cell process clearance in Caenorhabditis elegans
EFF-1 fusogen promotes phagosome sealing during cell process clearance in Caenorhabditis elegans
Ghose et al. show that EFF-1 fusogen generates a sealed phagosome during engulfment of cells with long processes in Caenorhabditis elegans, and different mechanisms are required for soma, distal and proximal process clearance.
- Piya Ghose
- , Alina Rashid
- & Shai Shaham
-
Article |
 Diminished apoptotic priming and ATM signalling confer a survival advantage onto aged haematopoietic stem cells in response to DNA damage
Diminished apoptotic priming and ATM signalling confer a survival advantage onto aged haematopoietic stem cells in response to DNA damage
Gutierrez-Martinez et al. show that an impaired DNA damage response and reduced apoptotic priming in old haematopoietic stem cells (HSCs) contribute to the survival and expansion of damaged HSCs in the bone marrow of aged mice.
- Paula Gutierrez-Martinez
- , Leah Hogdal
- & Derrick J. Rossi
-
Article |
 RIPK1-mediated induction of mitophagy compromises the viability of extracellular-matrix-detached cells
RIPK1-mediated induction of mitophagy compromises the viability of extracellular-matrix-detached cells
Hawk et al. show that RIPK1 activation during extracellular matrix detachment induces mitophagy through mitochondrial phosphatase PGAM5 to increase reactive oxygen species and non-apoptotic cell death, and that antagonizing RIPK1/PGAM5 enhances tumour formation.
- Mark A. Hawk
- , Cassandra L. Gorsuch
- & Zachary T. Schafer
-
Article |
 RIP3 targets pyruvate dehydrogenase complex to increase aerobic respiration in TNF-induced necroptosis
RIP3 targets pyruvate dehydrogenase complex to increase aerobic respiration in TNF-induced necroptosis
RIP3 regulates mitochondrial metabolism. Yang et al. show that RIP3 activates the pyruvate dehydrogenase complex to enhance aerobic respiration and increase mitochondrial ROS during necroptosis, and MLKL is required for RIP3 translocation to mitochondria.
- Zhentao Yang
- , Yan Wang
- & Jiahuai Han
-
News & Views |
 MK2 balances inflammation and cell death
MK2 balances inflammation and cell death
The cytokine tumour necrosis factor (TNF) and the toll-like receptors (TLRs) coordinate immune responses by activating inflammatory transcriptional programs, but these signals can also trigger cell death. Recent studies identify the MAP kinase substrate MK2 as a key player in determining whether cells live or die in response to TNF and TLR signalling.
- Andrew Oberst
-
Article |
 Extra-mitochondrial prosurvival BCL-2 proteins regulate gene transcription by inhibiting the SUFU tumour suppressor
Extra-mitochondrial prosurvival BCL-2 proteins regulate gene transcription by inhibiting the SUFU tumour suppressor
Wu et al. show that prosurvival BCL-2 proteins disrupt SUFU repression of GLI activity, leading to GLI target gene expression. BH3 mimetics disable GLI transcriptional activity driven by HH pathway mutations.
- Xiaofeng Wu
- , Li-shu Zhang
- & Lawrence Lum
-
Article |
 MK2 phosphorylation of RIPK1 regulates TNF-mediated cell death
MK2 phosphorylation of RIPK1 regulates TNF-mediated cell death
Dondelinger et al. and Menon et al. show that MAPKAP kinase-2 (MK2) phosphorylates RIPK1 to regulate TNF-mediated cell death as well as RIPK1 signalling in inflammation and bacterial infection.
- Yves Dondelinger
- , Tom Delanghe
- & Mathieu J. M. Bertrand
-
Article |
 p38MAPK/MK2-dependent phosphorylation controls cytotoxic RIPK1 signalling in inflammation and infection
p38MAPK/MK2-dependent phosphorylation controls cytotoxic RIPK1 signalling in inflammation and infection
Dondelinger et al. and Menon et al. show that MAPKAP kinase-2 (MK2) phosphorylates RIPK1 to regulate TNF-mediated cell death as well as RIPK1 signalling in inflammation and bacterial infection.
- Manoj B. Menon
- , Julia Gropengießer
- & Klaus Ruckdeschel
-
News & Views |
 A caspase-independent way to kill cancer cells
A caspase-independent way to kill cancer cells
Cancer treatments often focus on killing tumour cells through apoptosis, which is thought to typically require mitochondrial outer membrane permeabilization (MOMP) and subsequent caspase activation. A study now shows that MOMP can trigger TNF-dependent, but caspase-independent cell death, suggesting a different approach to improve cancer therapy.
- Brent E. Fitzwalter
- & Andrew Thorburn
-
Article |
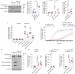 Mitochondrial permeabilization engages NF-κB-dependent anti-tumour activity under caspase deficiency
Mitochondrial permeabilization engages NF-κB-dependent anti-tumour activity under caspase deficiency
Tait and colleagues show that caspase-independent cell death induced by mitochondrial permeabilization stimulates NF-κB activity through downregulation of inhibitor of apoptosis, and enhances anti-tumour effects.
- Evangelos Giampazolias
- , Barbara Zunino
- & Stephen W. G. Tait
-
News & Views |
 PARL paves the way to apoptosis
PARL paves the way to apoptosis
Although the mitochondrial inner membrane rhomboid peptidase PARL is known to participate in critical signalling cascades, its role in apoptosis has remained unclear. PARL is now shown to process the mitochondrial pro-apoptotic protein Smac (also known as DIABLO) for its subsequent release into the cytosol where it antagonizes XIAP-mediated caspase inhibition to promote apoptosis.
- Naotada Ishihara
- & Katsuyoshi Mihara
-
Article |
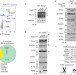 PARL mediates Smac proteolytic maturation in mitochondria to promote apoptosis
PARL mediates Smac proteolytic maturation in mitochondria to promote apoptosis
Saita et al. show that PARL cleaves Smac (also known as DIABLO) to generate an IAP-binding motif required for its apoptotic activity, identifying PARL-mediated Smac processing as a pro-apoptotic mitochondrial pathway.
- Shotaro Saita
- , Hendrik Nolte
- & Thomas Langer
-
Article |
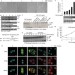 Cathepsin-B-mediated cleavage of Disabled-2 regulates TGF-β-induced autophagy
Cathepsin-B-mediated cleavage of Disabled-2 regulates TGF-β-induced autophagy
Jiang et al. show that Disabled-2 (Dab2) regulates the switch between autophagy and apoptosis in TGB-β-treated cells, through regulation of the Beclin-1–Vps34 interaction.
- Yong Jiang
- , Alec N. Woosley
- & Philip H. Howe
-
Article |
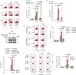 CHIP controls necroptosis through ubiquitylation- and lysosome-dependent degradation of RIPK3
CHIP controls necroptosis through ubiquitylation- and lysosome-dependent degradation of RIPK3
Receptor-interacting protein kinase 3 (RIPK3) is a key regulator of necroptosis. Seo et al. show that the E3 ligase CHIP mediates ubiquitylation and lysosomal degradation of RIPK3, thus regulating both necrosome formation and necroptosis.
- Jinho Seo
- , Eun-Woo Lee
- & Jaewhan Song
-
Article |
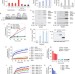 An interconnected hierarchical model of cell death regulation by the BCL-2 family
An interconnected hierarchical model of cell death regulation by the BCL-2 family
Chen et al. produced a quadruple knockout of all the known BH3-only proteins in order to characterize their pro-apoptotic functions. They establish that NOXA is a direct activator of BAX/BAK and not merely a sensitizer as previously suggested.
- Hui-Chen Chen
- , Masayuki Kanai
- & Emily H. Cheng
-
Article |
 AMPK and PFKFB3 mediate glycolysis and survival in response to mitophagy during mitotic arrest
AMPK and PFKFB3 mediate glycolysis and survival in response to mitophagy during mitotic arrest
Malumbres and colleagues reveal that mitotic arrest is accompanied by reduced mitochondrial mass and oxidative respiration resulting in activation of AMPK and induction of glycolysis to promote cell survival.
- Elena Doménech
- , Carolina Maestre
- & Marcos Malumbres
-
Review Article |
 Proteostasis control by the unfolded protein response
Proteostasis control by the unfolded protein response
- Claudio Hetz
- , Eric Chevet
- & Scott A. Oakes
-
Article |
 Autophagy and mTORC1 regulate the stochastic phase of somatic cell reprogramming
Autophagy and mTORC1 regulate the stochastic phase of somatic cell reprogramming
Qin, Pei and colleagues report that autophagy is induced early during somatic cell reprogramming into induced pluripotent stem cells. mTORC1 and autophagy control reprogramming efficiency by modulating mitochondrial architecture and p62 levels.
- Yasong Wu
- , Yuan Li
- & Duanqing Pei
-
Article |
 Ppm1b negatively regulates necroptosis through dephosphorylating Rip3
Ppm1b negatively regulates necroptosis through dephosphorylating Rip3
Han and colleagues reveal that the phosphatase Ppm1b dephosphorylates the kinase Rip3 to negatively regulate necroptosis in vitro and in vivo.
- Wanze Chen
- , Jianfeng Wu
- & Jiahuai Han
-
Article |
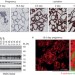 EGF-mediated induction of Mcl-1 at the switch to lactation is essential for alveolar cell survival
EGF-mediated induction of Mcl-1 at the switch to lactation is essential for alveolar cell survival
Visvader and colleagues report that in the mouse mammary gland, EGF and mTOR signalling induce expression of the anti-apoptotic Bcl2 family member Mcl-1 and show that this is required for the survival of milk-producing mammary epithelial cells.
- Nai Yang Fu
- , Anne C. Rios
- & Jane E. Visvader
-
News & Views |
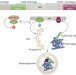 Huntingtin facilitates selective autophagy
Huntingtin facilitates selective autophagy
Selective autophagy is essential for maintaining cellular homeostasis under different growth conditions. Huntingtin, mutated versions of which have been implicated in Huntington disease, is now shown to act as a scaffold protein that couples the induction of autophagy and the selective recruitment of cargo into autophagosomes.
- Amir Gelman
- , Moran Rawet-Slobodkin
- & Zvulun Elazar
-
Article |
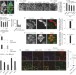 Lysosomal calcium signalling regulates autophagy through calcineurin and TFEB
Lysosomal calcium signalling regulates autophagy through calcineurin and TFEB
Medina, Ballabio and colleagues report that calcium release from the lysosome stimulates calcineurin, which dephosphorylates and activates TFEB. These findings reveal a central role for calcium signalling in autophagy and lysosome homeostasis.
- Diego L. Medina
- , Simone Di Paola
- & Andrea Ballabio
-
Article |
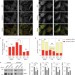 ATG12–ATG3 interacts with Alix to promote basal autophagic flux and late endosome function
ATG12–ATG3 interacts with Alix to promote basal autophagic flux and late endosome function
Debnath and colleagues report that the E2-like enzyme ATG3 is a target for ATG12 conjugation. ATG12–ATG3 conjugates interact with the ESCRT-associated protein Alix and regulate late endosome localization and exosome biogenesis.
- Lyndsay Murrow
- , Ritu Malhotra
- & Jayanta Debnath
-
Article |
 Huntingtin functions as a scaffold for selective macroautophagy
Huntingtin functions as a scaffold for selective macroautophagy
Zhang, Cuervo and colleagues find that Huntingtin (Htt), which is commonly mutated in Huntington disease, regulates selective autophagy. Htt enhances interactions between p62, LC3 and ubiquitylated cargo and derepresses ULK1 kinase activity.
- Yan-Ning Rui
- , Zhen Xu
- & Sheng Zhang
-
Article |
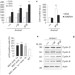 AMBRA1 links autophagy to cell proliferation and tumorigenesis by promoting c-Myc dephosphorylation and degradation
AMBRA1 links autophagy to cell proliferation and tumorigenesis by promoting c-Myc dephosphorylation and degradation
mTOR signalling both inhibits autophagy and promotes cell proliferation. Cecconi and colleagues report that AMBRA1 links these two processes by facilitating dephosphorylation and degradation of the proto-oncogene c-Myc.
- Valentina Cianfanelli
- , Claudia Fuoco
- & Francesco Cecconi
-
Article |
 Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice
Inactivation of the ferroptosis regulator Gpx4 triggers acute renal failure in mice
Ferroptosis is a form of non-apoptotic cell death with unclear physiological relevance. Conrad and colleagues now report that unrestrained ferroptosis can lead to renal failure. They also identify a small molecule that limits ferroptosis in vivo.
- Jose Pedro Friedmann Angeli
- , Manuela Schneider
- & Marcus Conrad
-
News & Views |
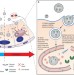 Milk makes lysosomes lethal
Milk makes lysosomes lethal
Stat3 has been shown to regulate lysosome membrane permeabilization and cell death in vivo during post-lactation mammary gland involution. It is now revealed that Stat3 induces lysosome membrane permeabilization by causing phagocytosis of milk fat globules, which destabilize the lysosome membrane leading to leakage of cathepsin proteases.
- Shefali Krishna
- & Michael Overholtzer
-
Article |
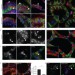 Stat3 controls cell death during mammary gland involution by regulating uptake of milk fat globules and lysosomal membrane permeabilization
Stat3 controls cell death during mammary gland involution by regulating uptake of milk fat globules and lysosomal membrane permeabilization
Lysosomal membrane permeabilization releases cathepsins to promote cell death and mammary gland involution. Sargeant et al. report that Stat3-driven phagocytic uptake of fatty acids in milk triglycerides permeabilizes lysosomes to induce cell death.
- Timothy J. Sargeant
- , Bethan Lloyd-Lewis
- & Christine J. Watson
-
News & Views |
 Anti-tubulins DEPendably induce apoptosis
Anti-tubulins DEPendably induce apoptosis
Anti-tubulin drugs are life-saving chemotherapeutics that kill cancer cells by stabilizing or disrupting microtubules. Despite their clinical utility, the molecular mechanisms by which anti-tubulins cause apoptotic cell death remain poorly understood. It is now shown that microtubule disruption can inhibit anti-apoptotic Bcl-2 family proteins through an evolutionarily conserved signalling axis involving DEPDC1 (LET-99 in Caenorhabditis elegans) and JNK.
- Daniel P. Denning
- & Takashi Hirose
-
Review Article |
 Organelle-specific initiation of cell death
Organelle-specific initiation of cell death
- Lorenzo Galluzzi
- , José Manuel Bravo-San Pedro
- & Guido Kroemer
-
Letter |
 DEPDC1/LET-99 participates in an evolutionarily conserved pathway for anti-tubulin drug-induced apoptosis
DEPDC1/LET-99 participates in an evolutionarily conserved pathway for anti-tubulin drug-induced apoptosis
Hengartner and colleagues use an RNAi-based screening approach in C. elegans to identify effectors of the chemotherapy drug vincristine. They report that the drug induces apoptosis by targeting a conserved LET-99–GPA-11–JNK1 pathway.
- Ataman Sendoel
- , Simona Maida
- & Michael O. Hengartner
-
Article |
 Protein interaction switches coordinate Raf-1 and MST2/Hippo signalling
Protein interaction switches coordinate Raf-1 and MST2/Hippo signalling
The Raf-1 kinase is important for mitogenic MAPK signalling but also inhibits pro-apoptotic Hippo signalling. Kolch and colleagues have found that the Hippo kinase LATS1 phosphorylates Raf-1 in a feedback regulatory loop to inhibit both pathways, and demonstrate by experiments and modelling that competing protein interaction can form the basis for a switch-like transition between signalling pathways.
- David Romano
- , Lan K. Nguyen
- & Walter Kolch
-
-
News & Views |
 Autophagy chews Fap to promote apoptosis
Autophagy chews Fap to promote apoptosis
Macroautophagy is a key regulator of cellular integrity and viability, but how the process facilitates apoptosis has remained poorly defined. It has now become clear that autophagy degrades the Fap-1 protein phosphatase, a critical negative regulator of apoptotic cell death signalled by the TNF receptor family member, Fas.
- Sanket Joshi
- & Kevin M. Ryan
-
Article |
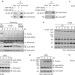 Plasma membrane translocation of trimerized MLKL protein is required for TNF-induced necroptosis
Plasma membrane translocation of trimerized MLKL protein is required for TNF-induced necroptosis
Liu and colleagues find that MLKL translocates to the plasma membrane to induce TNF-induced necroptosis, possibly through an effect on calcium influx and the action of the cation channel TRPM7.
- Zhenyu Cai
- , Siriporn Jitkaew
- & Zheng-Gang Liu
-
Article |
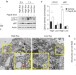 Autophagy variation within a cell population determines cell fate through selective degradation of Fap-1
Autophagy variation within a cell population determines cell fate through selective degradation of Fap-1
Data suggest that autophagy, a process normally associated with cell survival, also promotes cell death, depending on the stimulus or cell type. Thorburn and colleagues find that differences in basal autophagy levels in cells determine survival or death in response to death receptor activation, through modulation of Fap-1 degradation.
- Jacob M. Gump
- , Leah Staskiewicz
- & Andrew Thorburn
-
News & Views |
 Life without the mitochondrial calcium uniporter
Life without the mitochondrial calcium uniporter
Calcium enters mitochondria through a dedicated channel referred to as the mitochondrial calcium uniporter (MCU), whose molecular identity has long remained elusive. Since the discovery of the gene encoding the MCU protein two years ago, researchers have awaited the generation of a mouse lacking the MCU. These mice are fully viable and show defects limited to performance of high-energy-demanding exercises. Strikingly, no protection against necrosis is observed following ischaemia-reperfusion in the heart.
- Sébastien Herzig
- , Kinsey Maundrell
- & Jean-Claude Martinou
-
Article |
 The physiological role of mitochondrial calcium revealed by mice lacking the mitochondrial calcium uniporter
The physiological role of mitochondrial calcium revealed by mice lacking the mitochondrial calcium uniporter
Until the recent discovery of the mitochondrial calcium uniporter (MCU), the effect of increases in mitochondrial calcium levels could not be tested in vivo. Finkel and colleagues have knocked out the gene coding for MCU in adult mice, and show that MCU is required for transport of calcium into the mitochondria. They also show that, in its absence, the function of skeletal muscle is altered; however, surprisingly, no effects are observed on the sensitivity to cell-death-inducing agents.
- Xin Pan
- , Jie Liu
- & Toren Finkel
-
-
News & Views |
 Preventing lysosomal fat indigestion
Preventing lysosomal fat indigestion
Autophagy contributes to lipid catabolism through direct mobilization and breakdown of cellular lipid stores. Two recent studies reveal the regulatory mechanisms activated by cells during starvation to ensure that the cellular compartments involved in autophagic lipid catabolism are ready to receive, process and use these lipids. The regulators represent attractive therapeutic targets to help fight lipid-excess-associated diseases.
- Ana Maria Cuervo
-
-
Article |
 ER-stress-induced transcriptional regulation increases protein synthesis leading to cell death
ER-stress-induced transcriptional regulation increases protein synthesis leading to cell death
In the presence of stress stimuli, the endoplasmic reticulum either adapts the protein synthesis or triggers an apoptotic response, but the mechanisms underlying this decision are unknown. Kaufman and colleagues show that the ER stress response factors ATF4 and CHOP increase protein synthesis, which in turn induces oxidative stress and increased ATP consumption, leading to cell death during chronic ER stress.
- Jaeseok Han
- , Sung Hoon Back
- & Randal J. Kaufman
-
-
Letter |
 Selective autophagy degrades DICER and AGO2 and regulates miRNA activity
Selective autophagy degrades DICER and AGO2 and regulates miRNA activity
Voinnet and colleagues show that autophagy targets RNAi components DICER and AGO2 for degradation when they are not bound to miRNA. The autophagy receptor NDP52 is required for this homeostatic regulation of the RNAi machinery. The authors also found that autophagy influences the post-translational regulation of DICER mRNA.
- Derrick Gibbings
- , Serge Mostowy
- & Olivier Voinnet
-

 TBK1 and IKKε restrain cell death
TBK1 and IKKε restrain cell death