Featured
-
-
Article |
 Stem cell conversion to the cardiac lineage requires nucleotide signalling from apoptosing cells
Stem cell conversion to the cardiac lineage requires nucleotide signalling from apoptosing cells
Fort et al. implicate apoptosis and an epithelial-to-mesenchymal transition in the cardiomyocyte conversion of human stem cells. Nucleotides released from dying cells act through P2Y2 receptors on surviving cells to license WNT-dependent mesoderm specification.
- Loic Fort
- , Vivian Gama
- & Ian G. Macara
-
Letter |
 A role for Flower and cell death in controlling morphogen gradient scaling
A role for Flower and cell death in controlling morphogen gradient scaling
Merino et al. report that the cell-death factor Flower can control responses to morphogen gradients and thus has a role in gradient scaling.
- Marisa M. Merino
- , Carole Seum
- & Marcos Gonzalez-Gaitan
-
Article |
 Proteotoxic stress is a driver of the loser status and cell competition
Proteotoxic stress is a driver of the loser status and cell competition
Baumgartner et al. identify proteotoxic stress as the underlying cause of the loser status in a cell competition model caused by reduced autophagic, proteasomal flux and accumulation of protein aggregates.
- Michael E. Baumgartner
- , Michael P. Dinan
- & Eugenia Piddini
-
News & Views |
 IRE1α maintains HSC stemness under ER-stress
IRE1α maintains HSC stemness under ER-stress
Healthy and malignant haematopoietic stem cells (HSCs) must overcome a variety of cell intrinsic and extrinsic stresses to maintain their functionality. Now, IRE1α –XBP1 signalling is shown to protect HSCs and to promote survival of, and confer competitive advantages to, NRAS-mutated pre-leukaemic cells.
- Marina Scheller-Wendorff
- & Carsten Müller-Tidow
-
Article |
 Adaptive endoplasmic reticulum stress signalling via IRE1α–XBP1 preserves self-renewal of haematopoietic and pre-leukaemic stem cells
Adaptive endoplasmic reticulum stress signalling via IRE1α–XBP1 preserves self-renewal of haematopoietic and pre-leukaemic stem cells
Liu et al. show that the adaptive branch of unfolded protein response signalling, IRE1α–XBP1, protects haematopoietic stem cells and N-Ras pre-leukaemic stem cells from endoplasmic reticulum stress-induced apoptosis and supports their self-renewal.
- Lu Liu
- , Meiling Zhao
- & Qing Li
-
News & Views |
 TBK1 and IKKε restrain cell death
TBK1 and IKKε restrain cell death
RIPK1 plays a key role in several inflammatory and cell death signalling pathways. Understanding its regulation is pivotal for identifying diseases that might therapeutically benefit from RIPK1 inhibition. Recent studies now show that TBK1 and IKKε constitute a cell death checkpoint that restrains RIPK1 activation.
- Klaus Heger
- & Vishva M. Dixit
-
Article |
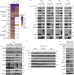 TBK1 and IKKε prevent TNF-induced cell death by RIPK1 phosphorylation
TBK1 and IKKε prevent TNF-induced cell death by RIPK1 phosphorylation
Lafont et al. uncover a checkpoint mediated by TBK1 and IKKε, which phosphorylate RIPK1 in the TNFR1-SC. TBK1 and IKKε recruitment depends on M1 ubiquitylation and NEMO to restrict TNF-induced cell death.
- Elodie Lafont
- , Peter Draber
- & Henning Walczak
-
Review Article |
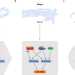 Life, death and autophagy
Life, death and autophagy
In this Review, Doherty and Baehrecke discuss the multiple roles of autophagy during cell survival and cell death. They cover the interplay between autophagy, apoptosis and necrosis, as well as engulfment and inflammation.
- Johnna Doherty
- & Eric H. Baehrecke
-
Article |
 Diminished apoptotic priming and ATM signalling confer a survival advantage onto aged haematopoietic stem cells in response to DNA damage
Diminished apoptotic priming and ATM signalling confer a survival advantage onto aged haematopoietic stem cells in response to DNA damage
Gutierrez-Martinez et al. show that an impaired DNA damage response and reduced apoptotic priming in old haematopoietic stem cells (HSCs) contribute to the survival and expansion of damaged HSCs in the bone marrow of aged mice.
- Paula Gutierrez-Martinez
- , Leah Hogdal
- & Derrick J. Rossi
-
News & Views |
 MK2 balances inflammation and cell death
MK2 balances inflammation and cell death
The cytokine tumour necrosis factor (TNF) and the toll-like receptors (TLRs) coordinate immune responses by activating inflammatory transcriptional programs, but these signals can also trigger cell death. Recent studies identify the MAP kinase substrate MK2 as a key player in determining whether cells live or die in response to TNF and TLR signalling.
- Andrew Oberst
-
Article |
 MK2 phosphorylation of RIPK1 regulates TNF-mediated cell death
MK2 phosphorylation of RIPK1 regulates TNF-mediated cell death
Dondelinger et al. and Menon et al. show that MAPKAP kinase-2 (MK2) phosphorylates RIPK1 to regulate TNF-mediated cell death as well as RIPK1 signalling in inflammation and bacterial infection.
- Yves Dondelinger
- , Tom Delanghe
- & Mathieu J. M. Bertrand
-
Article |
 p38MAPK/MK2-dependent phosphorylation controls cytotoxic RIPK1 signalling in inflammation and infection
p38MAPK/MK2-dependent phosphorylation controls cytotoxic RIPK1 signalling in inflammation and infection
Dondelinger et al. and Menon et al. show that MAPKAP kinase-2 (MK2) phosphorylates RIPK1 to regulate TNF-mediated cell death as well as RIPK1 signalling in inflammation and bacterial infection.
- Manoj B. Menon
- , Julia Gropengießer
- & Klaus Ruckdeschel
-
News & Views |
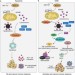 A caspase-independent way to kill cancer cells
A caspase-independent way to kill cancer cells
Cancer treatments often focus on killing tumour cells through apoptosis, which is thought to typically require mitochondrial outer membrane permeabilization (MOMP) and subsequent caspase activation. A study now shows that MOMP can trigger TNF-dependent, but caspase-independent cell death, suggesting a different approach to improve cancer therapy.
- Brent E. Fitzwalter
- & Andrew Thorburn
-
Article |
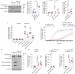 Mitochondrial permeabilization engages NF-κB-dependent anti-tumour activity under caspase deficiency
Mitochondrial permeabilization engages NF-κB-dependent anti-tumour activity under caspase deficiency
Tait and colleagues show that caspase-independent cell death induced by mitochondrial permeabilization stimulates NF-κB activity through downregulation of inhibitor of apoptosis, and enhances anti-tumour effects.
- Evangelos Giampazolias
- , Barbara Zunino
- & Stephen W. G. Tait
-
News & Views |
 PARL paves the way to apoptosis
PARL paves the way to apoptosis
Although the mitochondrial inner membrane rhomboid peptidase PARL is known to participate in critical signalling cascades, its role in apoptosis has remained unclear. PARL is now shown to process the mitochondrial pro-apoptotic protein Smac (also known as DIABLO) for its subsequent release into the cytosol where it antagonizes XIAP-mediated caspase inhibition to promote apoptosis.
- Naotada Ishihara
- & Katsuyoshi Mihara
-
Article |
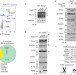 PARL mediates Smac proteolytic maturation in mitochondria to promote apoptosis
PARL mediates Smac proteolytic maturation in mitochondria to promote apoptosis
Saita et al. show that PARL cleaves Smac (also known as DIABLO) to generate an IAP-binding motif required for its apoptotic activity, identifying PARL-mediated Smac processing as a pro-apoptotic mitochondrial pathway.
- Shotaro Saita
- , Hendrik Nolte
- & Thomas Langer
-
Article |
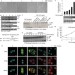 Cathepsin-B-mediated cleavage of Disabled-2 regulates TGF-β-induced autophagy
Cathepsin-B-mediated cleavage of Disabled-2 regulates TGF-β-induced autophagy
Jiang et al. show that Disabled-2 (Dab2) regulates the switch between autophagy and apoptosis in TGB-β-treated cells, through regulation of the Beclin-1–Vps34 interaction.
- Yong Jiang
- , Alec N. Woosley
- & Philip H. Howe
-
Article |
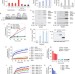 An interconnected hierarchical model of cell death regulation by the BCL-2 family
An interconnected hierarchical model of cell death regulation by the BCL-2 family
Chen et al. produced a quadruple knockout of all the known BH3-only proteins in order to characterize their pro-apoptotic functions. They establish that NOXA is a direct activator of BAX/BAK and not merely a sensitizer as previously suggested.
- Hui-Chen Chen
- , Masayuki Kanai
- & Emily H. Cheng
-
Article |
 AMPK and PFKFB3 mediate glycolysis and survival in response to mitophagy during mitotic arrest
AMPK and PFKFB3 mediate glycolysis and survival in response to mitophagy during mitotic arrest
Malumbres and colleagues reveal that mitotic arrest is accompanied by reduced mitochondrial mass and oxidative respiration resulting in activation of AMPK and induction of glycolysis to promote cell survival.
- Elena Doménech
- , Carolina Maestre
- & Marcos Malumbres
-
Review Article |
 Proteostasis control by the unfolded protein response
Proteostasis control by the unfolded protein response
- Claudio Hetz
- , Eric Chevet
- & Scott A. Oakes
-
Article |
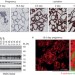 EGF-mediated induction of Mcl-1 at the switch to lactation is essential for alveolar cell survival
EGF-mediated induction of Mcl-1 at the switch to lactation is essential for alveolar cell survival
Visvader and colleagues report that in the mouse mammary gland, EGF and mTOR signalling induce expression of the anti-apoptotic Bcl2 family member Mcl-1 and show that this is required for the survival of milk-producing mammary epithelial cells.
- Nai Yang Fu
- , Anne C. Rios
- & Jane E. Visvader
-
News & Views |
 Anti-tubulins DEPendably induce apoptosis
Anti-tubulins DEPendably induce apoptosis
Anti-tubulin drugs are life-saving chemotherapeutics that kill cancer cells by stabilizing or disrupting microtubules. Despite their clinical utility, the molecular mechanisms by which anti-tubulins cause apoptotic cell death remain poorly understood. It is now shown that microtubule disruption can inhibit anti-apoptotic Bcl-2 family proteins through an evolutionarily conserved signalling axis involving DEPDC1 (LET-99 in Caenorhabditis elegans) and JNK.
- Daniel P. Denning
- & Takashi Hirose
-
Review Article |
 Organelle-specific initiation of cell death
Organelle-specific initiation of cell death
- Lorenzo Galluzzi
- , José Manuel Bravo-San Pedro
- & Guido Kroemer
-
Letter |
 DEPDC1/LET-99 participates in an evolutionarily conserved pathway for anti-tubulin drug-induced apoptosis
DEPDC1/LET-99 participates in an evolutionarily conserved pathway for anti-tubulin drug-induced apoptosis
Hengartner and colleagues use an RNAi-based screening approach in C. elegans to identify effectors of the chemotherapy drug vincristine. They report that the drug induces apoptosis by targeting a conserved LET-99–GPA-11–JNK1 pathway.
- Ataman Sendoel
- , Simona Maida
- & Michael O. Hengartner
-
Article |
 Protein interaction switches coordinate Raf-1 and MST2/Hippo signalling
Protein interaction switches coordinate Raf-1 and MST2/Hippo signalling
The Raf-1 kinase is important for mitogenic MAPK signalling but also inhibits pro-apoptotic Hippo signalling. Kolch and colleagues have found that the Hippo kinase LATS1 phosphorylates Raf-1 in a feedback regulatory loop to inhibit both pathways, and demonstrate by experiments and modelling that competing protein interaction can form the basis for a switch-like transition between signalling pathways.
- David Romano
- , Lan K. Nguyen
- & Walter Kolch
-
-
-
-
News & Views |
 Mcl-1 rescues a glitch in the matrix
Mcl-1 rescues a glitch in the matrix
Bcl-2 family proteins are known to control cell death and influence mitochondrial function. The function of Mcl-1, an anti-apoptotic Bcl-2 protein, is now shown to depend on its subcellular localization. Mcl-1 at the mitochondrial outer membrane inhibits mitochondrial permeabilization to block apoptosis. However, a cleaved form of Mcl-1 localizes to the mitochondrial matrix and controls inner mitochondrial morphology and oxidative phosphorylation, without directly modulating apoptosis.
- Joshua L. Andersen
- & Sally Kornbluth
-
Article |
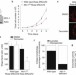 Anti-apoptotic MCL-1 localizes to the mitochondrial matrix and couples mitochondrial fusion to respiration
Anti-apoptotic MCL-1 localizes to the mitochondrial matrix and couples mitochondrial fusion to respiration
MCL-1 is an anti-apoptotic BCL-2 family member and is frequently upregulated in cancer, but the mechanism by which it promotes cell survival has been elusive. Opferman and colleagues provide insight into this process by showing that MCL-1 exists in different forms with discrete localizations and functions. MCL-1 variants targeted to the outer mitochondrial membrane antagonize BAX and BAK activation, whereas an N-terminally truncated isoform localizes to the mitochondrial matrix and regulates mitochondrial metabolism.
- Rhonda M. Perciavalle
- , Daniel P. Stewart
- & Joseph T. Opferman
-
Letter |
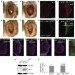 CDK5 and MEKK1 mediate pro-apoptotic signalling following endoplasmic reticulum stress in an autosomal dominant retinitis pigmentosa model
CDK5 and MEKK1 mediate pro-apoptotic signalling following endoplasmic reticulum stress in an autosomal dominant retinitis pigmentosa model
ER stress activates the unfolded protein response (UPR), which can ultimately result in apoptosis. Using a Drosophila model of ER stress, Ryoo and colleagues find that apoptosis is induced independently of the known UPR branches. They show that ER stress induces CDK5-mediated phosphorylation of MEKK1, which activates JNK and induces apoptosis.
- Min-Ji Kang
- , Jaehoon Chung
- & Hyung Don Ryoo
-
Letter |
 Toll-like receptor activation suppresses ER stress factor CHOP and translation inhibition through activation of eIF2B
Toll-like receptor activation suppresses ER stress factor CHOP and translation inhibition through activation of eIF2B
Endoplasmic reticulum (ER) stress and activation of the unfolded protein response inhibits de novo protein translation and activates CHOP. However, the long-term induction of these pathways in response to prolonged ER stress would be detrimental. Tabas and colleagues now reveal a mechanism through which Toll-like receptor signalling suppresses CHOP activation and promotes protein translation, thus allowing cells to adapt to persistent ER stress.
- Connie W. Woo
- , Lydia Kutzler
- & Ira Tabas
-
Article |
 Bcl-xL regulates metabolic efficiency of neurons through interaction with the mitochondrial F1FO ATP synthase
Bcl-xL regulates metabolic efficiency of neurons through interaction with the mitochondrial F1FO ATP synthase
ATP production by mitochondria requires the efficient flow of protons through the F1FO ATP-synthase complex. Jonas and colleagues show that Bcl-xL interacts with the F1FO complex in the mitochondrial matrix and increases the efficiency of this enzyme by decreasing proton leak.
- Kambiz N. Alavian
- , Hongmei Li
- & Elizabeth A. Jonas
-
Review Article |
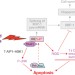 Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress
Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress
- Ira Tabas
- & David Ron
-
Letter |
 Loss of the RhoGAP SRGP-1 promotes the clearance of dead and injured cells in Caenorhabditis elegans
Loss of the RhoGAP SRGP-1 promotes the clearance of dead and injured cells in Caenorhabditis elegans
Apoptotic cell corpses are engulfed and removed through an evolutionarily conserved pathway. In Caenorhabditis elegans, inhibition of the Rac GAP SRGP-1 permits sick or dying cells to escape clearance by this pathway.
- Lukas J. Neukomm
- , Andreas P. Frei
- & Michael O. Hengartner
-
Article |
 Cytoskeletal keratin glycosylation protects epithelial tissue from injury
Cytoskeletal keratin glycosylation protects epithelial tissue from injury
Keratin 8 and 18 protect hepatocytes from apoptosis. Inhibiting keratin 18 glycosylation is shown to sensitize cells to liver and pancreatic injury and apoptosis, through a pathway involving Akt and PKCθ.
- Nam-On Ku
- , Diana M. Toivola
- & M. Bishr Omary
-
News & Views |
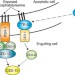 Tagging the dead: a bridging factor for Caenorhabditis elegans phagocyte receptors
Tagging the dead: a bridging factor for Caenorhabditis elegans phagocyte receptors
Recognition of apoptotic cells by phagocytic cells in Caenorhabditis elegans has been something of a mystery. A secreted transthyretin-like protein, TTR-52, has been identified as a bridging molecule between apoptotic cells and CED-1 on the phagocytic cells that engulf them.
- Rachael Rutkowski
- & Anton Gartner
-
Article |
 Caenorhabditis elegans transthyretin-like protein TTR-52 mediates recognition of apoptotic cells by the CED-1 phagocyte receptor
Caenorhabditis elegans transthyretin-like protein TTR-52 mediates recognition of apoptotic cells by the CED-1 phagocyte receptor
Recognition of apoptotic cells by phagocytes is not fully understood. In C. elegans, the transthyretin-like protein, TTR-52, is secreted by the endoderm and clusters around apoptotic cells, inducing their engulfment through the CED-1/6/7 pathway.
- Xiaochen Wang
- , Weida Li
- & Ding Xue
-
Article |
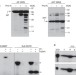 MTCH2/MIMP is a major facilitator of tBID recruitment to mitochondria
MTCH2/MIMP is a major facilitator of tBID recruitment to mitochondria
BH3-only protein tBID induces mitochondrial outer membrane permeabilization in response to death receptor activation. The mitochondrial carrier homologue 2 (MTCH2) protein acts as the tBID receptor on the mitochondria and is required for fas-induced cell death in mouse liver.
- Yehudit Zaltsman
- , Liat Shachnai
- & Atan Gross
-
Turning Points |
Signalling lessons from death receptors: the importance of cleavage
- Vishva M. Dixit

 NRF2 mediates melanoma addiction to GCDH by modulating apoptotic signalling
NRF2 mediates melanoma addiction to GCDH by modulating apoptotic signalling