Featured
-
-
Article
| Open Access Dynamical barrier and isotope effects in the simplest substitution reaction via Walden inversion mechanism
Dynamical barrier and isotope effects in the simplest substitution reaction via Walden inversion mechanism
The H+CH4substitution reaction is the simplest reaction occurring via the Walden inversion mechanism. Here, the authors perform a theoretical study of the reaction and uncover the important effect of the umbrella motion of the non-reacting methyl group on the reaction dynamics.
- Zhiqiang Zhao
- , Zhaojun Zhang
- & Dong H Zhang
-
Article
| Open Access Near-infrared uncaging or photosensitizing dictated by oxygen tension
Near-infrared uncaging or photosensitizing dictated by oxygen tension
The generation of reactive oxygen species (ROS) in photodynamic cancer treatments is limited by low intraturmoural oxygen availability. Here the authors show that irradiation of a silicon phthalocyanine leads to uncaging of a biologically active molecule or to ROS formation in an oxygen-dependent manner.
- Erin D. Anderson
- , Alexander P. Gorka
- & Martin J. Schnermann
-
Article
| Open Access Deoxyfluorination of alcohols with 3,3-difluoro-1,2-diarylcyclopropenes
Deoxyfluorination of alcohols with 3,3-difluoro-1,2-diarylcyclopropenes
Fluorination of organic molecules can dramatically change the properties and is commonly used for the synthesis of bioactive molecules. Here, the authors report the deoxyfluorination of alcohols and the selective monofluorination of diols using bench stable, tunable difluorocyclopropene reagents.
- Lingchun Li
- , Chuanfa Ni
- & Jinbo Hu
-
Article
| Open Access Product lambda-doublet ratios as an imprint of chemical reaction mechanism
Product lambda-doublet ratios as an imprint of chemical reaction mechanism
Propensity for a given Λ-doublet level is a common feature in many chemical reactions, but has so far remained unexplained. Here, the authors show how to predict computationally those propensities and relate them to the reaction mechanism on concurrent potential energy surfaces.
- P. G. Jambrina
- , A. Zanchet
- & F. J. Aoiz
-
Article
| Open Access Metal-free photochemical silylations and transfer hydrogenations of benzenoid hydrocarbons and graphene
Metal-free photochemical silylations and transfer hydrogenations of benzenoid hydrocarbons and graphene
Baird’s rules say that the first triplet state of benzene displays antiaromatic character. Here, the authors exploit this to show that aromatic molecules can undergo rapid transfer hydrogenation or silylations without the need for metal catalysts when photochemcially excited into this state.
- Raffaello Papadakis
- , Hu Li
- & Henrik Ottosson
-
Article
| Open Access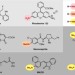 Stille coupling via C–N bond cleavage
Stille coupling via C–N bond cleavage
The Stille reaction is a long-standing method for the construction of carbon-carbon bonds by coupling aryl halides with organotin compounds. Here the authors report a Stille coupling between aryl ammonium salts and arylstannanes catalysed by nickel.
- Dong-Yu Wang
- , Masatoshi Kawahata
- & Masanobu Uchiyama
-
Article
| Open Access Asymmetric cyanation of imines via dipeptide-derived organophosphine dual-reagent catalysis
Asymmetric cyanation of imines via dipeptide-derived organophosphine dual-reagent catalysis
The Strecker reaction is a power method for the synthesis of cyano-substituted compounds. Here the authors report a dual-reagent system for asymmetric Strecker reactions, where an in situformed organocatalyst/methyl acrylate zwitterionic adduct activates both the cyanide source and the electrophile.
- Hong-Yu Wang
- , Chang-Wu Zheng
- & Gang Zhao
-
Article
| Open Access Solvent-driven electron trapping and mass transport in reduced graphites to access perfect graphene
Solvent-driven electron trapping and mass transport in reduced graphites to access perfect graphene
Graphite intercalation compounds are promising precursors for the reductive exfoliation of graphene. Here, the authors unveil the discharging mechanism of reduced graphitic compounds in a solid/liquid phase reaction, and further demonstrate its practical use for graphene production.
- Philipp Vecera
- , Johannes Holzwarth
- & Andreas Hirsch
-
Article
| Open Access Selective catalytic two-step process for ethylene glycol from carbon monoxide
Selective catalytic two-step process for ethylene glycol from carbon monoxide
Conversion of one-carbon feedstocks to more complex structures is vital for the production of bulk chemicals. Here, the authors report a highly selective method for the conversion of carbon monoxide to ethylene glycol by means of an oxamide intermediate.
- Kaiwu Dong
- , Saravanakumar Elangovan
- & Matthias Beller
-
Article
| Open Access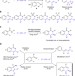 Merging rhodium-catalysed C–H activation and hydroamination in a highly selective [4+2] imine/alkyne annulation
Merging rhodium-catalysed C–H activation and hydroamination in a highly selective [4+2] imine/alkyne annulation
C–H activations and hydroaminations are useful synthetic procedures for building up complex, functional molecules. Here, the authors report a single catalyst capable of performing tandem C–H activation and hydroamination, giving access to N-heterocycles via the [4+2] annulation of aromatic imines and alkynes.
- Rajith S. Manan
- & Pinjing Zhao
-
Article
| Open Access Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism
Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism
Palladium catalyzed allylic substitution reactions typically proceed via an outer sphere mechanism, yielding predominately linear products. Here, the authors report an inner sphere process for the allylic substitution of ketone enolates, giving branched products with up to three contiguous stereocentres.
- Da-Chang Bai
- , Fei-Le Yu
- & Xue-Long Hou
-
Article
| Open Access Efficient hydrogen evolution in transition metal dichalcogenides via a simple one-step hydrazine reaction
Efficient hydrogen evolution in transition metal dichalcogenides via a simple one-step hydrazine reaction
Transition metal dichalcogenides are promising hydrogen evolution catalysts however they can require expensive processing steps to enhance their activity. Here, the authors report a one-step activation step in which room temperature hydrazine treatment results in much enhanced electrocatalytic performance.
- Dustin R. Cummins
- , Ulises Martinez
- & Gautam Gupta
-
Article
| Open Access Evidence for a chemical clock in oscillatory formation of UiO-66
Evidence for a chemical clock in oscillatory formation of UiO-66
Reactions with non-linear kinetics, such as chemical clocks, are reasonably common but only well understood in the liquid phase. Here, the authors report and rationalize a chemical clock reaction taking place in a solidifying metal-organic framework.
- M. G. Goesten
- , M. F. de Lange
- & Jorge Gascon
-
Article
| Open Access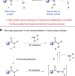 A two-step approach to achieve secondary amide transamidation enabled by nickel catalysis
A two-step approach to achieve secondary amide transamidation enabled by nickel catalysis
Transamidation reactions are kinetically and thermodynamically challenging because of the stability of the amide starting materials. Here, the authors show a two-step process—activation of a secondary amide, followed by nickel-catalysed C–N bond cleavage—that allows mild and high yielding transamidation.
- Emma L. Baker
- , Michael M. Yamano
- & Neil K. Garg
-
Article
| Open Access Synthesis of acetic acid via methanol hydrocarboxylation with CO2 and H2
Synthesis of acetic acid via methanol hydrocarboxylation with CO2 and H2
Industrial routes to acetic acid use carbon monoxide for the carbonylation of methanol. Here, the authors report a hydrocarboxylation method that instead uses carbon dioxide and hydrogen for the conversion of methanol into acetic acid.
- Qingli Qian
- , Jingjing Zhang
- & Buxing Han
-
Article
| Open Access Visualizing non-equilibrium lithiation of spinel oxide via in situ transmission electron microscopy
Visualizing non-equilibrium lithiation of spinel oxide via in situ transmission electron microscopy
Non-equilibrium intercalation reactions may determine the performance of lithium-ion battery materials undergoing lithiation, but it is difficult to probe in real time. Here, the authors use in situelectron microscopy to identify kinetically-driven phase evolution in magnetite single nanoparticles.
- Kai He
- , Sen Zhang
- & Dong Su
-
Article
| Open Access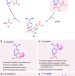 Enantioselective acyl transfer catalysis by a combination of common catalytic motifs and electrostatic interactions
Enantioselective acyl transfer catalysis by a combination of common catalytic motifs and electrostatic interactions
Nucleophilic catalysts are widely used for acyl transfer reactions, but chiral variants can be difficult to design or synthesise. Here, the authors report catalysts with chirality imparted from a binaphtyl moiety with tert-alcohol unit that show both high activity and enantioselectivity for a range of acyl transfer reactions.
- Hiroki Mandai
- , Kazuki Fujii
- & Seiji Suga
-
Article
| Open Access Practical carbon–carbon bond formation from olefins through nickel-catalyzed reductive olefin hydrocarbonation
Practical carbon–carbon bond formation from olefins through nickel-catalyzed reductive olefin hydrocarbonation
Olefins are employed in many coupling procedures but direct hydrocarbonations of unactivated olefins remain to be developed. Here, the authors report the nickel-catalyzed reductive coupling of olefins with aryl and alky electrophiles under mild conditions and with a broad substrate scope.
- Xi Lu
- , Bin Xiao
- & Lei Liu
-
Article
| Open Access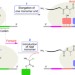 A strategy for sequence control in vinyl polymers via iterative controlled radical cyclization
A strategy for sequence control in vinyl polymers via iterative controlled radical cyclization
The ultimate control in polymer synthesis, precise monomer sequencing, is a highly sought after but complex problem. Here, the authors employ a repetitive and iterative intramolecular cyclization reaction, via a radical intermediate, to generate sequence-controlled vinyl oligomers.
- Yusuke Hibi
- , Makoto Ouchi
- & Mitsuo Sawamoto
-
Article
| Open Access Metal-free intermolecular formal cycloadditions enable an orthogonal access to nitrogen heterocycles
Metal-free intermolecular formal cycloadditions enable an orthogonal access to nitrogen heterocycles
Nitrogen containing heterocycles play key roles as both bioactive compounds and synthetic building blocks in organic chemistry. Here, the authors report the synthesis of a range of heterocyclic structures via metal free cycloadditions and provide mechanistic analyses for the transformations.
- Lan-Gui Xie
- , Supaporn Niyomchon
- & Nuno Maulide
-
Article
| Open Access A competitive and reversible deactivation approach to catalysis-based quantitative assays
A competitive and reversible deactivation approach to catalysis-based quantitative assays
Assays for catalytic systems—particularly ones with simple colorimetric readouts—are useful for the rapid evaluation of performance. Here, the authors report an assay based on a concurrent colour-forming reaction working across a wide range that can be stopped to allow measurements and subsequently restarted.
- Kazunori Koide
- , Matthew P. Tracey
- & Christopher J. Welch
-
Article
| Open Access Iron-catalysed cross-coupling of organolithium compounds with organic halides
Iron-catalysed cross-coupling of organolithium compounds with organic halides
Cross coupling reactions of organolithiums are challenging due to their high reactivity, though palladium catalysed versions have been reported. Here, the authors report the cross coupling of organolithiums with aryl halides with inexpensive and widely available iron catalysts.
- Zhenhua Jia
- , Qiang Liu
- & Henry N. C. Wong
-
Article
| Open Access Acceptorless dehydrogenation of small molecules through cooperative base metal catalysis
Acceptorless dehydrogenation of small molecules through cooperative base metal catalysis
Acceptorless dehydrogenation of simple alkanes and alcohols provides useful functionality and typically requires precious metal catalysts. Here, the authors dehydrogenate unactivated alkanes and alcohols at room temperature using earth-abundant metals with hydrogen as the sole by-product.
- Julian G. West
- , David Huang
- & Erik J. Sorensen
-
Article
| Open Access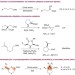 Asymmetric intramolecular α-cyclopropanation of aldehydes using a donor/acceptor carbene mimetic
Asymmetric intramolecular α-cyclopropanation of aldehydes using a donor/acceptor carbene mimetic
The formation of chiral centres adjacent to carbonyl groups is a highly challenging task in asymmetric catalysis. Here, the authors report the asymmetric diakylation of carbonyls via an intramolceular α-cyclopropanation procedure.
- Chaosheng Luo
- , Zhen Wang
- & Yong Huang
-
Article
| Open Access Reversed reactivity of anilines with alkynes in the rhodium-catalysed C–H activation/carbonylation tandem
Reversed reactivity of anilines with alkynes in the rhodium-catalysed C–H activation/carbonylation tandem
C-H activation reactions allow transformations on relatively unfunctionalised organic molecules. Here, the authors describe rhodium-catalysed C-H bond activation of unprotected anilines with alkynes leading to C-C bond formation (rather than the more usual C-N), and subsequent annulation to form quinolines.
- Siba P. Midya
- , Manoj K. Sahoo
- & Ekambaram Balaraman
-
Article
| Open Access High catalytic activity of oriented 2.0.0 copper(I) oxide grown on graphene film
High catalytic activity of oriented 2.0.0 copper(I) oxide grown on graphene film
Supported metal nanoparticles have been widely used as heterogeneous catalysts. Here, the authors report the synthesis of (1.1.1) copper on few layer graphene which oxidize to orientated (2.0.0) copper(I) oxide nanoplatelets which display high catalytic activity for a number of organic coupling reactions.
- Ana Primo
- , Ivan Esteve-Adell
- & Hermenegildo García
-
Article
| Open Access Formation of oligopeptides in high yield under simple programmable conditions
Formation of oligopeptides in high yield under simple programmable conditions
Typically, in order to form large peptides, complex procedures or activating agents are required. Here, the authors show that simple aqueous conditions with dehydration-hydration cycles are sufficient for the oligomerization of amino acids into peptides (up to 20 amino acids long) in yields of up to 50%.
- Marc Rodriguez-Garcia
- , Andrew J. Surman
- & Leroy Cronin
-
Article
| Open Access Coordinating activation strategy for C(sp3)–H/C(sp3)–H cross-coupling to access β-aromatic α-amino acids
Coordinating activation strategy for C(sp3)–H/C(sp3)–H cross-coupling to access β-aromatic α-amino acids
Oxidative coupling of two C(sp3)-H groups is an attractive approach for the formation of carbon-carbon bonds, building structural complexity while avoiding the formation of waste salts. Here, the authors report such a transformation, using a nickel catalyst to functionalise amino acids with benzylic radicals.
- Kaizhi Li
- , Qian Wu
- & Jingsong You
-
Article
| Open Access Coordination templated [2+2+2] cyclotrimerization in a porous coordination framework
Coordination templated [2+2+2] cyclotrimerization in a porous coordination framework
Nanospace defined by unsaturated metal coordination sites opens up the possibility of controlling and templating chemical reactivity. Here, the authors show that a Fe(III) dicarboxylate framework with triangularly arranged metal sites can direct the [2+2+2] cyclisation of nitriles, alkynes and alkenes.
- Yong-Sheng Wei
- , Mei Zhang
- & Xiao-Ming Chen
-
Article
| Open Access Non-covalent synthesis of supermicelles with complex architectures using spatially confined hydrogen-bonding interactions
Non-covalent synthesis of supermicelles with complex architectures using spatially confined hydrogen-bonding interactions
Ubiquitous in nature, hierarchical architectures are less commonly achieved in synthetic functional materials. Here, the authors design and carefully assemble block copolymer micelles into complex supermicelles using hydrogen bonding in orthogonal combination with other non-covalent interactions.
- Xiaoyu Li
- , Yang Gao
- & Ian Manners
-
Article
| Open Access Cationic mononuclear ruthenium carboxylates as catalyst prototypes for self-induced hydrogenation of carboxylic acids
Cationic mononuclear ruthenium carboxylates as catalyst prototypes for self-induced hydrogenation of carboxylic acids
Many structurally diverse carboxylic acids are available from renewable and petrochemical sources. Here, the authors report a method to reduce carboxylic acids to alcohols using hydrogen, with high selectivity, wherein the carboxylates form active and hydrogen-accepting parts of the catalyst system.
- Masayuki Naruto
- & Susumu Saito
-
Article
| Open Access A scalable and operationally simple radical trifluoromethylation
A scalable and operationally simple radical trifluoromethylation
Trifluoromethylation is a key transformation, particularly for pharmaceuticals, but many reagents are expensive and difficult to scale up. Here, the authors show that trifluoroacetic anhydride can act as a CF3source, allowing the radical reactions to be easily and inexpensively carried out at scale.
- Joel W. Beatty
- , James J. Douglas
- & Corey R. J. Stephenson
-
Article
| Open Access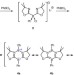 1,3,2,5-Diazadiborinine featuring nucleophilic and electrophilic boron centres
1,3,2,5-Diazadiborinine featuring nucleophilic and electrophilic boron centres
Boron atoms in aromatic heterocycles typically behave simply as electron pair acceptors. Here, the authors report an aromatic system containing both electrophilic and nucleophilic boron atoms, and demonstrate how this imparts frustrated Lewis pair type reactivity, including binding of carbon dioxide.
- Di Wu
- , Lingbing Kong
- & Rei Kinjo
-
Article
| Open Access Chemical reaction mechanisms in solution from brute force computational Arrhenius plots
Chemical reaction mechanisms in solution from brute force computational Arrhenius plots
Obtaining activation entropies and enthalpies of a reaction is important for distinguishing between alternative reaction mechanisms. Here the authors use computational methods to accurately obtain the enthalpic/entropic components of the activation free energy for hydrolytic deamination reactions.
- Masoud Kazemi
- & Johan Åqvist
-
Article
| Open Access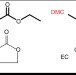 Radiolysis as a solution for accelerated ageing studies of electrolytes in Lithium-ion batteries
Radiolysis as a solution for accelerated ageing studies of electrolytes in Lithium-ion batteries
The degradation of organic solvents used in lithium-ion batteries reduces battery performance. Here, the authors present a radiolysis technique which is not only more efficient than conventional thermally activated ageing methods, but also allows mechanistic analysis of the degradation process.
- Daniel Ortiz
- , Vincent Steinmetz
- & Sophie Le Caër
-
Article
| Open Access Radical cascade reaction of alkynes with N-fluoroarylsulfonimides and alcohols
Radical cascade reaction of alkynes with N-fluoroarylsulfonimides and alcohols
Cascade and multi-component reactions allow highly functionalized molecules to be built from simpler precursors. Here, the authors report a three-component coupling giving α-amino-α-aryl ketones, initiated by the regioselective addition of nitrogen radicals to alkynes.
- Guangfan Zheng
- , Yan Li
- & Qian Zhang
-
Article
| Open Access Cobalt-catalysed site-selective intra- and intermolecular dehydrogenative amination of unactivated sp3 carbons
Cobalt-catalysed site-selective intra- and intermolecular dehydrogenative amination of unactivated sp3 carbons
Functionalizing unactivated carbon–hydrogen bonds is challenging, especially when using non-precious metals and dealing with sp3 hybridized carbons. Here, the authors report an intramolecular cobalt catalysed amination of C–H bonds of sp3carbons, giving access to β- and γ-lactams.
- Xuesong Wu
- , Ke Yang
- & Haibo Ge
-
Article |
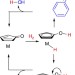 Direct and selective hydrogenolysis of arenols and aryl methyl ethers
Direct and selective hydrogenolysis of arenols and aryl methyl ethers
Deoxygenating phenols is a difficult task, made more complex by the tendency of hydrogenation techniques to also reduce the aromatic ring. Here, the authors show an iridium catalyst that can selectively cleave the C–O bond in phenols and related compounds, as well as cleaving aryl methyl ethers.
- Shuhei Kusumoto
- & Kyoko Nozaki
-
Article |
 Revealing a double-inversion mechanism for the F−+CH3Cl SN2 reaction
Revealing a double-inversion mechanism for the F−+CH3Cl SN2 reaction
The SN2 reaction is a stereospecific process, normally occurring via back-side inversion or front-side retention. Here, the authors report a global potential energy surface for the reaction between fluoride and CH3Cl and identify an additional double inversion mechanism.
- István Szabó
- & Gábor Czakó
-
Article
| Open Access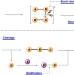 Bond cleavage, fragment modification and reassembly in enantioselective three-component reactions
Bond cleavage, fragment modification and reassembly in enantioselective three-component reactions
Multi-component reactions allow complex structures to be rapidly built from simple starting materials. Here, the authors report an enantioselective three-component coupling of imines, enamines and aryldiazoacetates catalysed by a phosphinic acid and palladium(II).
- Dan Zhang
- , Jun Zhou
- & Wenhao Hu
-
Article |
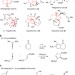 Asymmetric total synthesis of (−)-lingzhiol via a Rh-catalysed [3+2] cycloaddition
Asymmetric total synthesis of (−)-lingzhiol via a Rh-catalysed [3+2] cycloaddition
Bicyclic rings with quaternary bridgehead positions are difficult targets, yet are frequently observed in nature. Here, the authors report a cycloaddition reaction giving access to these structures in a stereoselective manner and employ it as the key step in a natural product total synthesis.
- Rong Long
- , Jun Huang
- & Zhen Yang
-
Article |
 Asymmetric trapping of zwitterionic intermediates by sulphur ylides in a palladium-catalysed decarboxylation-cycloaddition sequence
Asymmetric trapping of zwitterionic intermediates by sulphur ylides in a palladium-catalysed decarboxylation-cycloaddition sequence
Sulphur ylides are useful reagents in organic synthesis, typically reacting with highly polar reagents. Here, the authors show a catalytic asymmetric process for the addition of sulphur ylides to cyclic allylic esters with loss of CO2, giving access to chiral, highly functionalised indolines.
- Tian-Ren Li
- , Fen Tan
- & Wen-Jing Xiao
-
Article |
 Synthesis of chiral heterocycles by ligand-controlled regiodivergent and enantiospecific Suzuki Miyaura cross-coupling
Synthesis of chiral heterocycles by ligand-controlled regiodivergent and enantiospecific Suzuki Miyaura cross-coupling
Chiral heterocyclic compounds are highly valuable due to their biological and synthetic utility. Here the authors report a stereospecific cross-coupling methodology for the synthesis of non-aromatic heterocycles, giving products in high enantiomeric excess with control over the regioselectivity of bond formation.
- Jinyue Ding
- , Taras Rybak
- & Dennis G. Hall
-
Article |
 The intriguing dual-directing effect of 2-cyanobenzyl ether for a highly stereospecific glycosylation reaction
The intriguing dual-directing effect of 2-cyanobenzyl ether for a highly stereospecific glycosylation reaction
Glycosylation reactions can be hampered by a lack of selectivity, giving mixtures of α- and β-products. Here, the authors demonstrate the use of 2-cyanobenzyl functionalization as a directing group to give α- or β-selectivity from a single starting material.
- Kim Le Mai Hoang
- & Xue-Wei Liu
-
Article |
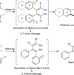 Intramolecular C–F and C–H bond cleavage promoted by butadienyl heavy Grignard reagents
Intramolecular C–F and C–H bond cleavage promoted by butadienyl heavy Grignard reagents
Grignard reagents are one of the mainstays of organic synthesis, but analogues with other Group 2 metals (heavy Grignards) are less easy to control. Here, the authors generate butadienyl calcium and barium heavy Grignards and control their reactivity in C–F and C–H cleavage reactions.
- Heng Li
- , Xiao-Ye Wang
- & Zhenfeng Xi
-
Article |
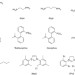 The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water
The Barbier–Grignard reaction is a commonly used method for forming carbon–carbon bonds, but for many substrates it is carried out in organic solvents. Here, the authors show a procedure for carrying out the reaction with unactivated aryl halides in water.
- Feng Zhou
- & Chao-Jun Li
-
Article |
 Roaming dynamics in radical addition–elimination reactions
Roaming dynamics in radical addition–elimination reactions
Roaming dynamics have been shown to be important in unimolecular decompositions, but the relevance to bimolecular reactions has been less clear. Here, the authors study radical addition/elimination reactions and implicate a roaming transition state in a bimolecular reaction.
- Baptiste Joalland
- , Yuanyuan Shi
- & Alexander M. Mebel
-
Article
| Open Access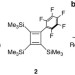 A Diels–Alder super diene breaking benzene into C2H2 and C4H4 units
A Diels–Alder super diene breaking benzene into C2H2 and C4H4 units
The stability of the benzene molecule reduces its tendency to react in ways that perturb the aromaticity—for example, in Diels–Alder reactions. Here the authors report a diene capable of not only inducing benzene to react under mild conditions but also of breaking apart the ring system itself.
- Yusuke Inagaki
- , Masaaki Nakamoto
- & Akira Sekiguchi
-
Article |
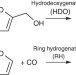 Directing reaction pathways by catalyst active-site selection using self-assembled monolayers
Directing reaction pathways by catalyst active-site selection using self-assembled monolayers
Control of active sites of heterogeneous catalysts can lead to enhanced product selectivity. Here, the authors use self-assembled alkanethiolate monolayers with varying surface densities to tune the selectivity of hydrogenation and hydrodeoxygenation of furfural on supported palladium catalysts.
- Simon H. Pang
- , Carolyn A. Schoenbaum
- & J. Will Medlin

 Synthesis and structures of gold and copper carbene intermediates in catalytic amination of alkynes
Synthesis and structures of gold and copper carbene intermediates in catalytic amination of alkynes