Featured
-
-
Article
| Open Access Iterative SuFEx approach for sequence-regulated oligosulfates and its extension to periodic copolymers
Iterative SuFEx approach for sequence-regulated oligosulfates and its extension to periodic copolymers
The synthesis of sequence-regulated oligosulfates has not yet been established due to the difficulties in precise reactivity control. Here, the authors report a multi-directional divergent iterative method to furnish oligosulfates based on a chain homologation approach, in which the fluorosulfate unit is regenerated.
- Min Pyeong Kim
- , Swatilekha Kayal
- & Sung You Hong
-
Article
| Open Access Palladium-catalyzed asymmetric carbene coupling en route to inherently chiral heptagon-containing polyarenes
Palladium-catalyzed asymmetric carbene coupling en route to inherently chiral heptagon-containing polyarenes
Developing facile and direct synthesis routes for enantioselective construction of cyclic π-conjugated molecules is crucial but the chirality orginiating from the distorted structure around heptagon-containing polyarenes is largely overlooked. Herein the authors present a highly enantioselective synthesis for fabrication of all carbon heptagon-containing polyarenes via palladium-catalyzed carbene-based cross–coupling of benzyl bromides and N-arylsulfonylhydrazones.
- Huan Zhang
- , Chuan-Jun Lu
- & Ren-Rong Liu
-
Article
| Open Access Selenium catalysis enables negative feedback organic oscillators
Selenium catalysis enables negative feedback organic oscillators
Multiple autocatalytic reactions producing thiols are known, but negative feedback loop motifs are unavailable for thiol chemistry. Here, the authors develop a negative feedback loop based on the selenocarbonates, in which thiols induce the release of aromatic selenols that catalyze the oxidation of thiols by organic peroxides.
- Xiuxiu Li
- , Polina Fomitskaya
- & Sergey N. Semenov
-
Article
| Open Access Nickel-catalyzed switchable arylative/endo-cyclization of 1,6-enynes
Nickel-catalyzed switchable arylative/endo-cyclization of 1,6-enynes
Divergent functionalizations of pi bonds allow for synthetic chemists to move from simplicity to complexity. Here, the authors report a nickel-catalyzed switchable arylation/cyclization of 1,6-enynes in which the nature of the ligand dictates the regioselectivity of alkyne arylation, while the electrophilic trapping reagents determine the selectivity of the cyclization mode.
- Wenfeng Liu
- , Wei Li
- & Wangqing Kong
-
Article
| Open Access Synthesis of meta-carbonyl phenols and anilines
Synthesis of meta-carbonyl phenols and anilines
Phenols and anilines are of extreme importance for medicinal chemistry and material science but the selective preparation of meta-substituted phenols and anilines remains challenging. Here the authors report an efficient copper-catalyzed dehydrogenation strategy to exclusively synthesize meta-carbonyl phenols and anilines from carbonyl substituted cyclohexanes.
- Bao-Yin Zhao
- , Qiong Jia
- & Yong-Qiang Wang
-
Article
| Open Access Enantioselective functionalization of unactivated C(sp3)–H bonds through copper-catalyzed diyne cyclization by kinetic resolution
Enantioselective functionalization of unactivated C(sp3)–H bonds through copper-catalyzed diyne cyclization by kinetic resolution
Site- and stereoselective C–H functionalization is challenging in the synthetic chemistry, and the catalytic enantioselective C(sp3)–H functionalization based on vinyl cations has scarcely explored. Here, the authors report an asymmetric copper-catalyzed tandem diyne cyclization/unactivated C(sp3)–H insertion reaction via a kinetic resolution.
- Yang-Bo Chen
- , Li-Gao Liu
- & Long-Wu Ye
-
Article
| Open Access Silver-catalyzed direct conversion of epoxides into cyclopropanes using N-triftosylhydrazones
Silver-catalyzed direct conversion of epoxides into cyclopropanes using N-triftosylhydrazones
Epoxides are prominent small-ring O-heterocycles found in a variety of bioactive natural products and pharmaceuticals. Here, the authors report a silver carbene strategy to achieve O-to-C atom exchange of epoxides in a single step, affording diverse fluoroalkylcyclopropanes.
- Linxuan Li
- , Paramasivam Sivaguru
- & Xihe Bi
-
Article
| Open Access Dimethyl sulfate and diisopropyl sulfate as practical and versatile O-sulfation reagents
Dimethyl sulfate and diisopropyl sulfate as practical and versatile O-sulfation reagents
O-Sulfation is a vital post-translational modification in bioactive molecules, yet there are significant challenges for the synthesis. Here, the authors report a general and robust approach to O-sulfation by harnessing the tunable reactivity of dimethyl sulfate or diisopropyl sulfate under tetrabutylammonium bisulfate activation.
- Shuaishuai Yue
- , Guoping Ding
- & Jiakun Li
-
Article
| Open Access Remote-carbonyl-directed sequential Heck/isomerization/C(sp2)–H arylation of alkenes for modular synthesis of stereodefined tetrasubstituted olefins
Remote-carbonyl-directed sequential Heck/isomerization/C(sp2)–H arylation of alkenes for modular synthesis of stereodefined tetrasubstituted olefins
The synthesis of highly substituted olefins has been pursued for decades. Here, the authors show a carbonyl-directed sequential Heck/isomerization/C–H arylation of alkenes for modular synthesis of stereodefined all-carbon tetrasubstituted olefins.
- Runze Luan
- , Ping Lin
- & Weiping Su
-
Article
| Open Access Observation of geometric phase effect through backward angular oscillations in the H + HD → H2 + D reaction
Observation of geometric phase effect through backward angular oscillations in the H + HD → H2 + D reaction
In a combined experimental and theoretical study of the H + HD → H2 + D reaction at low collision energy (1.72 eV), the authors obtain detailed information on the quantum reaction dynamics surrounding a conical intersection.
- Shihao Li
- , Jiayu Huang
- & Xueming Yang
-
Article
| Open Access C-SuFEx linkage of sulfonimidoyl fluorides and organotrifluoroborates
C-SuFEx linkage of sulfonimidoyl fluorides and organotrifluoroborates
Sulfur fluoride exchange (SuFEx), a new type of linkage reaction, has great potential for application in functional molecule linkage to prepare pharmaceuticals, biomolecules, and polymers. Herein, the authors report the synthesis of a range of sulfoximines through SuFEx reaction between sulfonimidoyl fluorides and aryl/alkyl organotrifluoroborates.
- Suqin Zhao
- , Daming Zeng
- & Xuefeng Jiang
-
Article
| Open Access Unified metal-free intermolecular Heck-type sulfonylation, cyanation, amination, amidation of alkenes by thianthrenation
Unified metal-free intermolecular Heck-type sulfonylation, cyanation, amination, amidation of alkenes by thianthrenation
Direct and site-selective C–H functionalization of alkenes under environmentally benign conditions represents a useful and attractive yet challenging transformation to access value-added molecules. Here, the authors report a protocol for a variety of intermolecular Heck-type functionalization of C(sp2)–H bond of alkenes by thianthrenation.
- Ming-Shang Liu
- , Hai-Wu Du
- & Wei Shu
-
Article
| Open Access Regio- and enantioselective synthesis of acyclic quaternary carbons via organocatalytic addition of organoborates to (Z)-Enediketones
Regio- and enantioselective synthesis of acyclic quaternary carbons via organocatalytic addition of organoborates to (Z)-Enediketones
All-carbon quaternary stereocenters are an important synthetic motif but are especially difficult to synthesize enantioselectively. Here, the authors demonstrate the organocatalytic regio- and enantioselective synthesis of valuable acyclic 1,4-dicarbonyl products with vinylated and arylated quaternary centers.
- Po-Kai Peng
- , Andrew Isho
- & Jeremy A. May
-
Article
| Open Access C−F bond activation enables synthesis of aryl difluoromethyl bicyclopentanes as benzophenone-type bioisosteres
C−F bond activation enables synthesis of aryl difluoromethyl bicyclopentanes as benzophenone-type bioisosteres
Bioisosteric design has become an essential approach during drug molecule discovery process. Here, the authors describe a strategy for defluorinative couplings of propellanes, enabling the synthesis of aryl difluoromethyl bicyclopentanes as benzophenone-type bioisosteres.
- Mingshuo Chen
- , Yuang Cui
- & Xiaheng Zhang
-
Article
| Open Access Unlocking regioselective meta-alkylation with epoxides and oxetanes via dynamic kinetic catalyst control
Unlocking regioselective meta-alkylation with epoxides and oxetanes via dynamic kinetic catalyst control
Regioselective arene C−H bond alkylation is a powerful tool in synthetic chemistry, yet subject to many challenges. Here, the authors report the meta-C−H bond alkylation of aromatics bearing N-directing groups using (hetero)aromatic epoxides as alkylating agents.
- Peng-Bo Bai
- , Alastair Durie
- & Igor Larrosa
-
Article
| Open Access Atroposelective hydroarylation of biaryl phosphines directed by phosphorus centres
Atroposelective hydroarylation of biaryl phosphines directed by phosphorus centres
Directing group strategies have been used extensively in C–H activation reactions but current methods rely heavily on coordination with nitrogen or oxygen atoms in molecules and have therefore been found to exhibit limited generality in asymmetric syntheses. Here, the authors report enantioselective C–H activation with unsaturated hydrocarbons directed by phosphorus centres to rapidly construct libraries of axially chiral phosphines through dynamic kinetic resolution.
- Zexian Li
- , Minyan Wang
- & Zhuangzhi Shi
-
Article
| Open Access Alkali metal reduction of alkali metal cations
Alkali metal reduction of alkali metal cations
Here the authors demonstate that counter to expectation provided by the relevant standard reduction potentials, a chloroberyllate, [{SiNDipp}BeClLi]2, reacts with the group 1 elements (M = Na, K, Rb, Cs) to provide the respective heavier alkali metal analogues, [{SiNDipp}BeClM]2.
- Kyle G. Pearce
- , Han-Ying Liu
- & Michael S. Hill
-
Article
| Open Access Highly scalable photoinduced synthesis of silanols via untraversed pathway for chlorine radical (Cl•) generation
Highly scalable photoinduced synthesis of silanols via untraversed pathway for chlorine radical (Cl•) generation
Contrary to photocatalysis, the use of photochemical energy solely to initiate reactions is underexplored. Here, the authors demonstrate light initiated synthesis of functionalized organosilanols, silanediols, and polymeric siloxanol under ambient reaction conditions.
- Argha Saha
- , Wajid Ali
- & Debabrata Maiti
-
Article
| Open Access Near-infrared light-triggered prodrug photolysis by one-step energy transfer
Near-infrared light-triggered prodrug photolysis by one-step energy transfer
Prodrug photolysis enables spatiotemporal control of drug release at the desired lesions, but most of the photocleavable groups cannot be directly activated by near-infrared (NIR) light that features deep penetration and low phototoxicity. Here, the authors report an upconversion-like process via only one step of energy transfer for NIR light-triggered prodrug photolysis.
- Kaiqi Long
- , Wen Lv
- & Weiping Wang
-
Article
| Open Access Selective ring expansion and C−H functionalization of azulenes
Selective ring expansion and C−H functionalization of azulenes
Fused carbocycles are key structural elements of molecules in nature and they are often found in drugs and organic materials, but bicyclic systems containing six and seven-membered rings are difficult to prepare. Here, the authors functionalize an azulene skeleton that consists of fused five and seven-membered rings and carry out a ring expansion reaction to afford the desired bicycles.
- Sangjune Park
- , Cheol-Eui Kim
- & Phil Ho Lee
-
Article
| Open Access Ligand-modulated nickel-catalyzed regioselective silylalkylation of alkenes
Ligand-modulated nickel-catalyzed regioselective silylalkylation of alkenes
Multicomponent cross-coupling of alkenes with silicon reagents is used to yield complex silicon-containing compounds from readily accessible feedstock chemicals but the reaction with simple alkenes remains challenging. Here, the authors report a regioselective silylalkylation of simple alkenes, which is enabled by using a stable Ni(II) salt and an inexpensive trans-1,2-diaminocyclohexane ligand as a catalyst.
- Chao Ding
- , Yaoyu Ren
- & Guoyin Yin
-
Article
| Open Access Cation-induced changes in the inner- and outer-sphere mechanisms of electrocatalytic CO2 reduction
Cation-induced changes in the inner- and outer-sphere mechanisms of electrocatalytic CO2 reduction
The underlying mechanism of cation effects on CO2RR remains debated. Combining constrained density function theory, Marcus theory, and slow-growth sampling approaches, we resolve how cations modulate the inner- and outer-sphere pathways of CO2RR at solid-liquid interfaces.
- Xueping Qin
- , Heine A. Hansen
- & Marko M. Melander
-
Article
| Open Access Mechanistic study on the side arm effect in a palladium/Xu-Phos-catalyzed enantioselective alkoxyalkenylation of γ-hydroxyalkenes
Mechanistic study on the side arm effect in a palladium/Xu-Phos-catalyzed enantioselective alkoxyalkenylation of γ-hydroxyalkenes
Recently, the asymmetric bifunctionalization of alkenes has received much attention but the development of enantioselective alkoxyalkenylation has posed a considerable challenge and has lagged largely behind. Here, the authors report a palladium-catalyzed enantioselective alkoxyalkenylation reaction, using a range of primary, secondary, and tertiary γ-hydroxyalkenes with alkenyl halides.
- Shuai Zhu
- , Zihao Ye
- & Junliang Zhang
-
Article
| Open Access Stereoselectivity control in Rh-catalyzed β-OH elimination for chiral allene formation
Stereoselectivity control in Rh-catalyzed β-OH elimination for chiral allene formation
The chiral allene formation reactions catalyzed by palladium occur via a syn elimination mechanism, but there is a lack in understanding of the stereoselectivity in rhodium catalyzed reactions, specifically the β-OH elimination step. Here, the authors report the sterically controlled Rh-catalyzed SN2’-type substitution reactions of tertiary propargylic alcohols with arylmetallic species affording the non-readily available enantioenriched tetrasubstituted allenes via either exclusive syn- or anti-β-OH elimination.
- Jie Wang
- , Wei-Feng Zheng
- & Shengming Ma
-
Article
| Open Access Selection of isomerization pathways of multistep photoswitches by chalcogen bonding
Selection of isomerization pathways of multistep photoswitches by chalcogen bonding
Multistep photoswitches can engage in different photoisomerization pathways, but are challenging to control. Here, the authors report the development of multistep photoswitches by manipulation of the strength and mechanism of chalcogen bonding interactions.
- Shuaipeng Jia
- , Hebo Ye
- & Lei You
-
Article
| Open Access Nucleophilic aromatization of monoterpenes from isoprene under nickel/iodine cascade catalysis
Nucleophilic aromatization of monoterpenes from isoprene under nickel/iodine cascade catalysis
Organic compounds possessing two isoprene units play important roles in chemical industry. Herein, the authors use bulk C5 chemical—isoprene to synthesise various monoterpenoids via a nucleophilic aromatization of monoterpenes under cascade catalysis of nickel and iodine
- Wei-Song Zhang
- , Ding-Wei Ji
- & Qing-An Chen
-
Article
| Open Access Asymmetric formal C–C bond insertion into aldehydes via copper-catalyzed diyne cyclization
Asymmetric formal C–C bond insertion into aldehydes via copper-catalyzed diyne cyclization
The formal C–C bond insertion into aldehydes is an attractive methodology for the assembly of homologated carbonyl compounds but the homologation of aldehydes has been limited to diazo approach and the enantioselective reaction was rarely developed. Here, the authors report an asymmetric formal C–C bond insertion into aldehydes through diyne cyclization strategy
- Cui-Ting Li
- , Lin-Jun Qi
- & Bo Zhou
-
Article
| Open Access Synthesis of tertiary alkylphosphonate oligonucleotides through light-driven radical-polar crossover reactions
Synthesis of tertiary alkylphosphonate oligonucleotides through light-driven radical-polar crossover reactions
Alkylphosphonates are used as chargeneutral replacements for naturally-occurring phosphodiester backbones in nucleotides but the choice of alkyl moieties that can be attached to phosphorus atoms in these compounds is limited to methyl groups or primary/secondary alkyls. Here, the authors demonstrate the tertiary alkylation of the phosphorus atoms of phosphites bearing two 2’-deoxynuclosides using a carbocation generated via a light-driven radical-polar crossover mechanism
- Kenji Ota
- , Kazunori Nagao
- & Hirohisa Ohmiya
-
Article
| Open Access Electrochemical halogen-atom transfer alkylation via α-aminoalkyl radical activation of alkyl iodides
Electrochemical halogen-atom transfer alkylation via α-aminoalkyl radical activation of alkyl iodides
Alkyl halides, widely recognized as important building blocks and reagents in organic synthesisbut generating alkyl radicals directly from unactivated alkyl halides under mild conditions remains a challenge. Here the authors report an effective electrooxidation strategy for generating alkyl radicals from unactivated alkyl iodides via an electrochemical halogen-atom transfer process under mild conditions
- Xiang Sun
- & Ke Zheng
-
Article
| Open Access Mechanistic insights into excited-state palladium catalysis for C–S bond formations and dehydrogenative sulfonylation of amines
Mechanistic insights into excited-state palladium catalysis for C–S bond formations and dehydrogenative sulfonylation of amines
Photocatalytic selective C(sp3)–H activation/cross-coupling reactions are appealing in organic synthesis. Here the authors describe the development of excited-state Pd-catalyzed dehydrogenative β-sulfonylation reactions using amines and aryl sulfonyl chlorides via intermolecular hydrogen atom transfer and C−S cross-coupling processes at room temperature.
- Krishnamoorthy Muralirajan
- , Rajesh Kancherla
- & Magnus Rueping
-
Article
| Open Access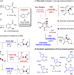 Catalytic 4-exo-dig carbocyclization for the construction of furan-fused cyclobutanones and synthetic applications
Catalytic 4-exo-dig carbocyclization for the construction of furan-fused cyclobutanones and synthetic applications
Aromatic ring fused cyclobutanone is a strained motif with broad applications. Here, the authors report a catalytic 4- exo-dig process, which proved successful to access furan-fused cyclobutanones that can serve as versatile synthetic blocks.
- Kemiao Hong
- , Yi Zhou
- & Xinfang Xu
-
Article
| Open Access Stereospecific synthesis of silicon-stereogenic optically active silylboranes and general synthesis of chiral silyl Anions
Stereospecific synthesis of silicon-stereogenic optically active silylboranes and general synthesis of chiral silyl Anions
Silicon-stereogenic optically active silylboranes could potentially allow the formation of chiral silyl nucleophiles as well as the synthesis of various chiral silicon compounds but the synthesis of such silicon-stereogenic silylboranes remains underdeveloped. Here, the authors report the synthesis of silicon-stereogenic optically active silylboranes via a stereospecific Si–H borylation of chiral hydrosilanes in high yield enantiospecificity
- Xihong Wang
- , Chi Feng
- & Hajime Ito
-
Article
| Open Access Kinetic resolution of substituted amido[2.2]paracyclophanes via asymmetric electrophilic amination
Kinetic resolution of substituted amido[2.2]paracyclophanes via asymmetric electrophilic amination
Planar chiral [2.2]paracyclophanes have found significant applications in various research fields. Here, Yang and coworkers report an efficient kinetic resolution method for various substituted amido[2.2]paracyclophanes via asymmetric C-H amination.
- Shaoze Yu
- , Hanyang Bao
- & Xiaoyu Yang
-
Article
| Open Access Enantioselective organocatalytic synthesis of axially chiral aldehyde-containing styrenes via SNAr reaction-guided dynamic kinetic resolution
Enantioselective organocatalytic synthesis of axially chiral aldehyde-containing styrenes via SNAr reaction-guided dynamic kinetic resolution
The precise and efficient construction of axially chiral scaffolds, particularly toward the aryl-alkene atropoisomers remains challenging. Here, the authors discuss an organocatalytic asymmetric nucleophilic aromatic substitution reaction of aldehyde-substituted styrenes involving a dynamic kinetic resolution process via a hemiacetal intermediate, offering a pathway to axial styrene scaffolds.
- Fengyuan Guo
- , Siqiang Fang
- & Tianli Wang
-
Article
| Open Access Regio- and enantioselective CuH-catalyzed 1,2- and 1,4-hydrosilylation of 1,3-enynes
Regio- and enantioselective CuH-catalyzed 1,2- and 1,4-hydrosilylation of 1,3-enynes
Functionalization of 1,3-enyne represent a powerful method to construct allenyl- and propargylderivatives, but hydrosilylation reaction of 1,3-enynes remains underdeveloped. Here, the authors report a copper-catalyzed ligand-controlled selective 1,2- and 1,4- hydrosilylation of 1,3-enynes which proceeds enantioselectively with high yields.
- Zi-Lu Wang
- , Qi Li
- & Yun-He Xu
-
Article
| Open Access Diastereo- and atroposelective synthesis of N-arylpyrroles enabled by light-induced phosphoric acid catalysis
Diastereo- and atroposelective synthesis of N-arylpyrroles enabled by light-induced phosphoric acid catalysis
Axially chiral N-arylpyrrole motifs are privileged scaffolds in numerous biologically active compounds but asymmetric synthesis of N-arylpyrroles is challenging. Here, the authors develop a diastereo- and atroposelective synthesis of N-arylpyrroles enabled by light-induced phosphoric acid catalysis.
- Lei Dai
- , Xueting Zhou
- & Yixin Lu
-
Article
| Open Access Modular assembly of indole alkaloids enabled by multicomponent reaction
Modular assembly of indole alkaloids enabled by multicomponent reaction
Indole alkaloids are one of the largest alkaloid classes, proving valuable structural moiety in pharmaceuticals but the direct single-step synthesis with broad structural diversity remains challenging. Herein, the authors report a modular assembly of tetrahydrocarboline type of indole alkaloids from simple building blocks in a single step while showing broad compatibility with medicinally relevant functionality
- Jiaming Li
- , Zhencheng Lai
- & Sunliang Cui
-
Article
| Open Access Valve turning towards on-cycle in cobalt-catalyzed Negishi-type cross-coupling
Valve turning towards on-cycle in cobalt-catalyzed Negishi-type cross-coupling
Ligands and additives are often utilized to stabilize low-valent catalytic metal species experimentally, while their role in suppressing metal deposition has been less studied. Here, the authors report an on-cycle mechanism for CoCl2bpy2 catalyzed Negishi-type cross-coupling describing a full catalytic cycle for this reaction using multiple spectroscopic studies.
- Xu Luo
- , Dali Yang
- & Aiwen Lei
-
Article
| Open Access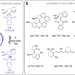 A photochemical method to evidence directional molecular motions
A photochemical method to evidence directional molecular motions
Light driven synthetic molecular motors represent crucial building blocks for advanced molecular machines and their applications but the development of very fast motors remains challenging. Here the authors restrict fast molecular motor rotations and force them to proceed in two separate rotation-photoequilibria enabling them to elucidate the directionality of the motion.
- Benjamin Lukas Regen-Pregizer
- , Ani Ozcelik
- & Henry Dube
-
Article
| Open Access Construction of C-B axial chirality via dynamic kinetic asymmetric cross-coupling mediated by tetracoordinate boron
Construction of C-B axial chirality via dynamic kinetic asymmetric cross-coupling mediated by tetracoordinate boron
The application of catalytic dynamic kinetic asymmetric transformation (DyKAT) in axial chirality is limited to fivemembered metalacyclic intermediate-directed equilibrium of substrate enantiomers. Here, the authors report a tetracoordinate boron-directed DyKAT of racemic, configurationally stable 3-bromo- 2,1-azaborines for the construction of C-B axial chirality.
- Kai Yang
- , Yanfei Mao
- & Qiuling Song
-
Article
| Open Access Unlocking full and fast conversion in photocatalytic carbon dioxide reduction for applications in radio-carbonylation
Unlocking full and fast conversion in photocatalytic carbon dioxide reduction for applications in radio-carbonylation
While the reduction of CO2 to CO is a very active research topic, use of the reduced CO in organic synthesis is not yet mature. Herein, the authors report a photocatalytic process that unlocks full CO2-to-CO conversion, opening opportunities for access to 11C- and 14C-labeled pharmaceuticals.
- Serena Monticelli
- , Alex Talbot
- & Davide Audisio
-
Article
| Open Access Carbene-catalyzed chemoselective reaction of unsymmetric enedials for access to Furo[2,3-b]pyrroles
Carbene-catalyzed chemoselective reaction of unsymmetric enedials for access to Furo[2,3-b]pyrroles
Chemoselective reactions of two or multiple functional groups with similar reactivities remain an ongoing challenge in chemistry. Here the authors disclosed a chemoselective reaction of enedial and observed that the high chemoselectivity is mainly controlled via redox (rather than steric) properties of the substrate/catalyst.
- Guodong Fan
- , Qingyun Wang
- & Yonggui Robin Chi
-
Article
| Open Access Photoswitchable dynamic conjugate addition-elimination reactions as a tool for light-mediated click and clip chemistry
Photoswitchable dynamic conjugate addition-elimination reactions as a tool for light-mediated click and clip chemistry
Photoinduced dynamic covalent bonds have the advantage of light-mediated bond making and breaking. Here, the authors report photoswitchable dynamic conjugate addition-elimination reactions with a broad scope and demonstrate diverse applications.
- Hanwei Lu
- , Hebo Ye
- & Lei You
-
Article
| Open Access Reaction performance prediction with an extrapolative and interpretable graph model based on chemical knowledge
Reaction performance prediction with an extrapolative and interpretable graph model based on chemical knowledge
Predictive modelling remains a key challenge for designing synthetic transformations. Here, the authors develop a knowledge-based graph model to predict reaction yield and stereoselectivity, offering an extrapolative and interpretable approach for evaluating reaction performance.
- Shu-Wen Li
- , Li-Cheng Xu
- & Xin Hong
-
Article
| Open Access Manipulating hydrogen bond dissociation rates and mechanisms in water dimer through vibrational strong coupling
Manipulating hydrogen bond dissociation rates and mechanisms in water dimer through vibrational strong coupling
Mechanistic understanding of how light-matter interaction alters chemical reactivities remains a challenge. Here, the authors show both hydrogen bond dissociation rate and pathways can be modified through cavity-modified molecular coupling patterns.
- Qi Yu
- & Joel M. Bowman
-
Article
| Open Access Migratory allylic arylation of 1,n-enols enabled by nickel catalysis
Migratory allylic arylation of 1,n-enols enabled by nickel catalysis
Allylic substitution reactions have emerged as a powerful tool for the construction of C − C/X bonds. Here, the authors develop a migratory strategy of allylic arylation under the catalysis of nickel and lanthanide triflate.
- Dan Zhao
- , Bing Xu
- & Can Zhu
-
Article
| Open Access Lanthanide-doped MoS2 with enhanced oxygen reduction activity and biperiodic chemical trends
Lanthanide-doped MoS2 with enhanced oxygen reduction activity and biperiodic chemical trends
Oxygen reduction reaction plays a key role in many applications of MoS2-based materials. Here, using first-principles simulations, the authors find the enhanced oxygen-reduction activity with a biperiodic chemical trend on the lanthanide-doped MoS2.
- Yu Hao
- , Liping Wang
- & Liang-Feng Huang
-
Article
| Open Access Direct allylic acylation via cross-coupling involving cooperative N‑heterocyclic carbene, hydrogen atom transfer, and photoredox catalysis
Direct allylic acylation via cross-coupling involving cooperative N‑heterocyclic carbene, hydrogen atom transfer, and photoredox catalysis
The versatility and importance of β,γ-unsaturated ketones has been appreciated for a long time in synthesis and medicinal chemistry. Here, the authors use carboxylic acids and readily available olefins as substrates for transformations involving N‑heterocyclic carbene catalysis, hydrogen atom transfer catalysis, and photoredox catalysis for the synthesis of β,γ-unsaturated ketones.
- Xiaochen Wang
- , Rongxin Yang
- & Qingmin Wang
-
Article
| Open Access Rehybridization dynamics into the pericyclic minimum of an electrocyclic reaction imaged in real-time
Rehybridization dynamics into the pericyclic minimum of an electrocyclic reaction imaged in real-time
Electrocyclic reactions proceed through critical geometries, which are known as pericyclic transition states in thermal reactions and pericyclic minima in photochemical reactions. Here, the authors image the structure of a pericyclic minimum in real time using a combination of ultrafast electron diffraction and ab initio dynamics simulations.
- Y. Liu
- , D. M. Sanchez
- & T. J. A. Wolf

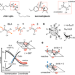 Comparative quantum-classical dynamics of natural and synthetic molecular rotors show how vibrational synchronization modulates the photoisomerization quantum efficiency
Comparative quantum-classical dynamics of natural and synthetic molecular rotors show how vibrational synchronization modulates the photoisomerization quantum efficiency