Article
|
Open Access
Featured
-
-
Article
| Open Access Valve turning towards on-cycle in cobalt-catalyzed Negishi-type cross-coupling
Valve turning towards on-cycle in cobalt-catalyzed Negishi-type cross-coupling
Ligands and additives are often utilized to stabilize low-valent catalytic metal species experimentally, while their role in suppressing metal deposition has been less studied. Here, the authors report an on-cycle mechanism for CoCl2bpy2 catalyzed Negishi-type cross-coupling describing a full catalytic cycle for this reaction using multiple spectroscopic studies.
- Xu Luo
- , Dali Yang
- & Aiwen Lei
-
Article
| Open Access A photochemical method to evidence directional molecular motions
A photochemical method to evidence directional molecular motions
Light driven synthetic molecular motors represent crucial building blocks for advanced molecular machines and their applications but the development of very fast motors remains challenging. Here the authors restrict fast molecular motor rotations and force them to proceed in two separate rotation-photoequilibria enabling them to elucidate the directionality of the motion.
- Benjamin Lukas Regen-Pregizer
- , Ani Ozcelik
- & Henry Dube
-
Article
| Open Access Construction of C-B axial chirality via dynamic kinetic asymmetric cross-coupling mediated by tetracoordinate boron
Construction of C-B axial chirality via dynamic kinetic asymmetric cross-coupling mediated by tetracoordinate boron
The application of catalytic dynamic kinetic asymmetric transformation (DyKAT) in axial chirality is limited to fivemembered metalacyclic intermediate-directed equilibrium of substrate enantiomers. Here, the authors report a tetracoordinate boron-directed DyKAT of racemic, configurationally stable 3-bromo- 2,1-azaborines for the construction of C-B axial chirality.
- Kai Yang
- , Yanfei Mao
- & Qiuling Song
-
Article
| Open Access Unlocking full and fast conversion in photocatalytic carbon dioxide reduction for applications in radio-carbonylation
Unlocking full and fast conversion in photocatalytic carbon dioxide reduction for applications in radio-carbonylation
While the reduction of CO2 to CO is a very active research topic, use of the reduced CO in organic synthesis is not yet mature. Herein, the authors report a photocatalytic process that unlocks full CO2-to-CO conversion, opening opportunities for access to 11C- and 14C-labeled pharmaceuticals.
- Serena Monticelli
- , Alex Talbot
- & Davide Audisio
-
Article
| Open Access Carbene-catalyzed chemoselective reaction of unsymmetric enedials for access to Furo[2,3-b]pyrroles
Carbene-catalyzed chemoselective reaction of unsymmetric enedials for access to Furo[2,3-b]pyrroles
Chemoselective reactions of two or multiple functional groups with similar reactivities remain an ongoing challenge in chemistry. Here the authors disclosed a chemoselective reaction of enedial and observed that the high chemoselectivity is mainly controlled via redox (rather than steric) properties of the substrate/catalyst.
- Guodong Fan
- , Qingyun Wang
- & Yonggui Robin Chi
-
Article
| Open Access Photoswitchable dynamic conjugate addition-elimination reactions as a tool for light-mediated click and clip chemistry
Photoswitchable dynamic conjugate addition-elimination reactions as a tool for light-mediated click and clip chemistry
Photoinduced dynamic covalent bonds have the advantage of light-mediated bond making and breaking. Here, the authors report photoswitchable dynamic conjugate addition-elimination reactions with a broad scope and demonstrate diverse applications.
- Hanwei Lu
- , Hebo Ye
- & Lei You
-
Article
| Open Access Reaction performance prediction with an extrapolative and interpretable graph model based on chemical knowledge
Reaction performance prediction with an extrapolative and interpretable graph model based on chemical knowledge
Predictive modelling remains a key challenge for designing synthetic transformations. Here, the authors develop a knowledge-based graph model to predict reaction yield and stereoselectivity, offering an extrapolative and interpretable approach for evaluating reaction performance.
- Shu-Wen Li
- , Li-Cheng Xu
- & Xin Hong
-
Article
| Open Access Manipulating hydrogen bond dissociation rates and mechanisms in water dimer through vibrational strong coupling
Manipulating hydrogen bond dissociation rates and mechanisms in water dimer through vibrational strong coupling
Mechanistic understanding of how light-matter interaction alters chemical reactivities remains a challenge. Here, the authors show both hydrogen bond dissociation rate and pathways can be modified through cavity-modified molecular coupling patterns.
- Qi Yu
- & Joel M. Bowman
-
Article
| Open Access Migratory allylic arylation of 1,n-enols enabled by nickel catalysis
Migratory allylic arylation of 1,n-enols enabled by nickel catalysis
Allylic substitution reactions have emerged as a powerful tool for the construction of C − C/X bonds. Here, the authors develop a migratory strategy of allylic arylation under the catalysis of nickel and lanthanide triflate.
- Dan Zhao
- , Bing Xu
- & Can Zhu
-
Article
| Open Access Lanthanide-doped MoS2 with enhanced oxygen reduction activity and biperiodic chemical trends
Lanthanide-doped MoS2 with enhanced oxygen reduction activity and biperiodic chemical trends
Oxygen reduction reaction plays a key role in many applications of MoS2-based materials. Here, using first-principles simulations, the authors find the enhanced oxygen-reduction activity with a biperiodic chemical trend on the lanthanide-doped MoS2.
- Yu Hao
- , Liping Wang
- & Liang-Feng Huang
-
Article
| Open Access Direct allylic acylation via cross-coupling involving cooperative N‑heterocyclic carbene, hydrogen atom transfer, and photoredox catalysis
Direct allylic acylation via cross-coupling involving cooperative N‑heterocyclic carbene, hydrogen atom transfer, and photoredox catalysis
The versatility and importance of β,γ-unsaturated ketones has been appreciated for a long time in synthesis and medicinal chemistry. Here, the authors use carboxylic acids and readily available olefins as substrates for transformations involving N‑heterocyclic carbene catalysis, hydrogen atom transfer catalysis, and photoredox catalysis for the synthesis of β,γ-unsaturated ketones.
- Xiaochen Wang
- , Rongxin Yang
- & Qingmin Wang
-
Article
| Open Access Rehybridization dynamics into the pericyclic minimum of an electrocyclic reaction imaged in real-time
Rehybridization dynamics into the pericyclic minimum of an electrocyclic reaction imaged in real-time
Electrocyclic reactions proceed through critical geometries, which are known as pericyclic transition states in thermal reactions and pericyclic minima in photochemical reactions. Here, the authors image the structure of a pericyclic minimum in real time using a combination of ultrafast electron diffraction and ab initio dynamics simulations.
- Y. Liu
- , D. M. Sanchez
- & T. J. A. Wolf
-
Article
| Open Access Computational study on the catalytic control of endo/exo Diels-Alder reactions by cavity quantum vacuum fluctuations
Computational study on the catalytic control of endo/exo Diels-Alder reactions by cavity quantum vacuum fluctuations
Strong light-matter interactions are a pathway to chemical control at the molecular level. Here, authors theoretically show that an optical cavity allows to control catalysis, inhibition, and endo/exo stereoselectivity in Diels-Alder reactions.
- Fabijan Pavošević
- , Robert L. Smith
- & Angel Rubio
-
Article
| Open Access Switching imidazole reactivity by dynamic control of tautomer state in an allosteric foldamer
Switching imidazole reactivity by dynamic control of tautomer state in an allosteric foldamer
Molecular biology achieves control over complex reaction networks by means of molecular systems that translate a chemical input into an orthogonal chemical output but the application of dynamic foldamers has been limited by the restriction to stereochemical changes that produce spectroscopic signals. Here, the authors present an artificial molecular translation device that converts a chemical input into an unrelated chemical output.
- David P. Tilly
- , Jean-Paul Heeb
- & Jonathan Clayden
-
Article
| Open Access Photocatalyzed regioselective hydrosilylation for the divergent synthesis of geminal and vicinal borosilanes
Photocatalyzed regioselective hydrosilylation for the divergent synthesis of geminal and vicinal borosilanes
Geminal and vicinal borosilanes are useful building blocks in synthetic chemistry. But in contrast to the well-established transition-metal catalyzed methods, radical approaches are rarely explored. Here, the authors report the synthesis of geminal borosilanes from α-selective hydrosilylation of alkenyl boronates via photoinduced hydrogen atom transfer (HAT) catalysis
- Degong Kong
- , Muliang Zhang
- & Jie Wu
-
Article
| Open Access Insights into solvent and surface charge effects on Volmer step kinetics on Pt (111)
Insights into solvent and surface charge effects on Volmer step kinetics on Pt (111)
Excess free charges on electrode surfaces drive changes in hydrogen electrocatalysis kinetics. Here, the authors show how redox solvent reorganization energy is insensitive to interfacial electric field strength; instead, the charged surface directly modulates proton electrochemical potential.
- Jon C. Wilson
- , Stavros Caratzoulas
- & Yushan Yan
-
Article
| Open Access Transition-metal-free silylboronate-mediated cross-couplings of organic fluorides with amines
Transition-metal-free silylboronate-mediated cross-couplings of organic fluorides with amines
C–N bond cross-coupling is fundamental in organic chemistry, but the readily available organic fluorides are unsuitable as coupling substrates. Here, we realize defluorinative cross-coupling with amines via a silylboronate-mediated strategy.
- Jun Zhou
- , Zhengyu Zhao
- & Norio Shibata
-
Article
| Open Access Gas phase synthesis of the C40 nano bowl C40H10
Gas phase synthesis of the C40 nano bowl C40H10
Nanobowls represent building blocks of fullerenes and nanotubes as detected in combustion systems and deep space, but their formation mechanisms in these environments have remained elusive. Here, the authors explore the gas-phase formation of benzocorannulene and beyond to the C40 nanobowl.
- Lotefa B. Tuli
- , Shane J. Goettl
- & Ralf I. Kaiser
-
Article
| Open Access Electrochemical oxidative difunctionalization of diazo compounds with two different nucleophiles
Electrochemical oxidative difunctionalization of diazo compounds with two different nucleophiles
The difunctionalization of diazo compounds, traditionally with one nucleophile and one electrophile, is a powerful strategy. Here, the authors develop a path of electrochemical oxidative difunctionalization of diazo compounds with two different nucleophiles.
- Dongfeng Yang
- , Zhipeng Guan
- & Aiwen Lei
-
Article
| Open Access Prediction of transition state structures of gas-phase chemical reactions via machine learning
Prediction of transition state structures of gas-phase chemical reactions via machine learning
Obtaining good initial structures is the main challenge for the computational study of transition states. Here, fast and accurate predictions for transition state of gas phase reactions are achieved by machine learning based on interatomic distances.
- Sunghwan Choi
-
Article
| Open Access Polyfunctionalization of vicinal carbon centers and synthesis of unsymmetric 1,2,3,4-tetracarbonyl compounds
Polyfunctionalization of vicinal carbon centers and synthesis of unsymmetric 1,2,3,4-tetracarbonyl compounds
The synthesis of polycarbonyl compounds featuring four or more carbonyl groups is a synthetic challenge for organic chemists. Here, the authors describe how unsymmetric polycarbonyl compounds bearing up to 4 adjacent carbonyl groups can be accessed within a two-step synthesis from readily available starting materials and discuss their reactivity.
- Luca De Angelis
- , Chao Pei
- & Michael P. Doyle
-
Article
| Open Access Dearomative triple elementalization of quinolines driven by visible light
Dearomative triple elementalization of quinolines driven by visible light
Organoboron and organosilicon compounds have potential applications in various fields including materials science and medicinal chemistry. Here the authors report a protocol that enables the dearomative carbo-sila-boration of quinolines.
- Shiho Ishigaki
- , Yuki Nagashima
- & Ken Tanaka
-
Article
| Open Access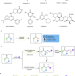 Azoarene activation for Schmidt-type reaction and mechanistic insights
Azoarene activation for Schmidt-type reaction and mechanistic insights
The Schmidt reaction enables nitrogen insertion across an aldehyde or ketone substrate by frequently using azide reagents. Here, the authors show a Schmidt reaction of aryldiazonium salts as a replacement of azide reagents to access skeletally diverse cyclic lactams.
- Fan-Tao Meng
- , Ya-Nan Wang
- & Bo Jiang
-
Article
| Open Access How mono- and diphosphine ligands alter regioselectivity of the Rh-catalyzed annulative cleavage of bicyclo[1.1.0]butanes
How mono- and diphosphine ligands alter regioselectivity of the Rh-catalyzed annulative cleavage of bicyclo[1.1.0]butanes
Ligands alter the regioselectivity of Rh(I)- catalyzed cycloisomerizations of bicyclobutanes. Here, mechanistic studies performed by the authors pinpoint a concerted C−C bond cleavage and carbenoid formation responsible for the divergence.
- Pan-Pan Chen
- , Peter Wipf
- & K. N. Houk
-
Article
| Open Access Synthesis of I(III)/S(VI) reagents and their reactivity in photochemical cycloaddition reactions with unsaturated bonds
Synthesis of I(III)/S(VI) reagents and their reactivity in photochemical cycloaddition reactions with unsaturated bonds
Novel methodologies for introducing sulfoxonium groups into organic molecules are desirable. Here the authors report the synthesis of an iodonium-sulfoxonium ylide reagent that enables the preparation of sulfoxonium-containing cyclic structures upon reaction with alkenes, alkynes, nitriles or allenes.
- Li Li
- , Kun Deng
- & Yong Huang
-
Article
| Open Access Dearomatization of aromatic asmic isocyanides to complex cyclohexadienes
Dearomatization of aromatic asmic isocyanides to complex cyclohexadienes
As aromatic compounds can be obtained easily, their functionalization is a cornerstone of organic synthesis. Here, the authors report a dearomatization of aromatic isocyanides, installing three carbon-carbon bonds, two quaternary stereocenters, and three orthogonal functionalities in one operation.
- Bilal Altundas
- , Embarek Alwedi
- & Fraser F. Fleming
-
Article
| Open Access Cascade cyclization of alkene-tethered acylsilanes and allylic sulfones enabled by unproductive energy transfer photocatalysis
Cascade cyclization of alkene-tethered acylsilanes and allylic sulfones enabled by unproductive energy transfer photocatalysis
Acylsilanes decompose under light irradiation, and this limits their use in light-induced organic transformations. Here the authors report a strategy to inhibit the light-induced decomposition of acylsilanes, enabling the photochemical synthesis of cyclopentanol derivatives from alkene-tethered acylsilanes and allylic sulfones.
- Yunxiao Zhang
- , Yizhi Zhang
- & Xiao Shen
-
Article
| Open Access Deep reaction network exploration at a heterogeneous catalytic interface
Deep reaction network exploration at a heterogeneous catalytic interface
This study demonstrates how reaction network characterization can be performed on heterogeneous catalytic surfaces predictively, rather than retrospectively, using automated exploration algorithms on an ethylene oligomerization exemplar reaction.
- Qiyuan Zhao
- , Yinan Xu
- & Brett M. Savoie
-
Article
| Open Access Dearomative di- and trifunctionalization of aryl sulfoxides via [5,5]-rearrangement
Dearomative di- and trifunctionalization of aryl sulfoxides via [5,5]-rearrangement
In contrast with well-established dearomatization protocols such as the Birch reduction, sigmatropic rearrangement-based dearomatizations are a less-explored but potentially effective route to saturated compounds. Here, the authors show a [5,5]-rearrangement-enabled dearomatization of aryl sulfoxides.
- Mengjie Hu
- , Yanping Liu
- & Bo Peng
-
Article
| Open Access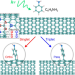 Photochemical spin-state control of binding configuration for tailoring organic color center emission in carbon nanotubes
Photochemical spin-state control of binding configuration for tailoring organic color center emission in carbon nanotubes
Chemical functionalization of the sidewalls of single-wall carbon nanotubes (SWCNTs) is an emerging route to introduce fluorescent quantum defects and tailor the emission properties. Here, authors demonstrate that spin-selective photochemistry diversifies SWCNT emission tunability by controlling the morphology of the emitting sites.
- Yu Zheng
- , Yulun Han
- & Sergei Tretiak
-
Article
| Open Access Unexpected steric hindrance failure in the gas phase F− + (CH3)3CI SN2 reaction
Unexpected steric hindrance failure in the gas phase F− + (CH3)3CI SN2 reaction
Base-induced elimination (E2) and bimolecular nucleophilic substitution (SN2) are of significant importance in physical organic chemistry. Here, the authors show that the competing factor of E2 as opposed to steric hindrance determines the low reactivity of SN2 in the F− + (CH3)3CI reaction.
- Xiaoxiao Lu
- , Chenyao Shang
- & Dong H. Zhang
-
Article
| Open Access Generation and direct observation of a triplet arylnitrenium ion
Generation and direct observation of a triplet arylnitrenium ion
Nitrenium ions are highly electrophilic reactive intermediates of formula R−N−R+, nitrogen analogue of carbenes. Here the authors report the detection of a triplet nitrenium ion using time-resolved spectroscopic methods and ab initio computations, allowing a glimpse at the properties and behavior of this important class of intermediates.
- Lili Du
- , Juanjuan Wang
- & Arthur H. Winter
-
Article
| Open Access The molecular mechanism of constructive remodeling of a mechanically-loaded polymer
The molecular mechanism of constructive remodeling of a mechanically-loaded polymer
Large mechanical loads usually degrade polymers by accelerating fragmentation of their backbones but rarely, they can cause new backbone bonds to form leading to arrested or reversed degradation when new bonds are formed faster than the original bonds break.Here, the authors report the molecular mechanism and analysis of constructive remodeling driven by the macroradical products of mechanochemical fragmentation of a hydrocarbon backbone.
- Chenxu Wang
- , Sergey Akbulatov
- & Roman Boulatov
-
Article
| Open Access Rhodium-catalyzed selective direct arylation of phosphines with aryl bromides
Rhodium-catalyzed selective direct arylation of phosphines with aryl bromides
The widespread use of biaryl monophosphines is frequently hampered by the challenges associated with their modular preparation. Here, the authors report a protocol that appends arenes to arylphosphines to access a series of these compounds via rhodium-catalysed P(III)- directed ortho C–H activation, enabling one-fold, two-fold, and threefold direct arylation.
- Dingyi Wang
- , Mingjie Li
- & Zhuangzhi Shi
-
Article
| Open Access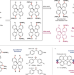 Ring walking as a regioselectivity control element in Pd-catalyzed C-N cross-coupling
Ring walking as a regioselectivity control element in Pd-catalyzed C-N cross-coupling
The phenomenon of „ring-walking‟, wherein a metal catalyst remains bound to a pi system as it migrates to another coupling site, is supported largely by circumstantial evidence. Here the authors perform an in-depth kinetic study of Buchwald- Hartwig animations with several catalytic systems delineating the phenomenon of ring walking from diffusion-controlled coupling.
- Madeleine C. Deem
- , Joshua S. Derasp
- & Jason E. Hein
-
Article
| Open Access Reactant-induced photoactivation of in situ generated organogold intermediates leading to alkynylated indoles via Csp2-Csp cross-coupling
Reactant-induced photoactivation of in situ generated organogold intermediates leading to alkynylated indoles via Csp2-Csp cross-coupling
Dual photo- and metal-catalysis is generally not well-understood when the metal catalyst is a gold complex. Here the authors show that a variation of a known metallaphotoredox annulation is possible without an added photocatalyst, proceeding putatively through a gold–intermediate photosensitization by the aggregate of a potassium amide reactant, and yielding indole derivatives.
- Fen Zhao
- , Mehdi Abdellaoui
- & Louis Fensterbank
-
Article
| Open Access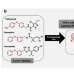 Skeletal Ni electrode-catalyzed C-O cleavage of diaryl ethers entails direct elimination via benzyne intermediates
Skeletal Ni electrode-catalyzed C-O cleavage of diaryl ethers entails direct elimination via benzyne intermediates
Biomass conversion holds promise as a more sustainable source of platform chemicals, but limitations in the ways in which lignin can be broken down is a current bottleneck. Here the authors report an electrocatalytic hydrogenolysis over skeletal Ni that cleaves diaryl ethers, chemically resistant moieties in both renewable carbon sources and persistent organic pollutants.
- Yuting Zhou
- , Grace E. Klinger
- & James E. Jackson
-
Article
| Open Access NiH-catalysed proximal-selective hydroalkylation of unactivated alkenes and the ligand effects on regioselectivity
NiH-catalysed proximal-selective hydroalkylation of unactivated alkenes and the ligand effects on regioselectivity
Difunctionalization of olefins is an ongoing and important focus of synthetic organic chemistry. Here the authors report a nickel-catalysed hydroalkylation of unactivated alkenes to obtain branched alkyl carboxylic acids or alkyl amines, using aminoquinoline and picolinamide as directing groups.
- Xiao-Xu Wang
- , Yuan-Tai Xu
- & Yao Fu
-
Article
| Open Access Dichotomy of platinum(II) and gold(III) carbene intermediates switching from N- to O-selectivity
Dichotomy of platinum(II) and gold(III) carbene intermediates switching from N- to O-selectivity
Benzofurazan, a cyclic heterocycle, can form open-chain metal carbene species in the presence of suitable catalysts. Here the authors show divergent reactivity when using gold(III) and platinum(II) catalysts, and perform computational and experimental mechanistic studies to explain the differing reactivity
- Hongming Jin
- , Wen-Yan Tong
- & A. Stephen K. Hashmi
-
Article
| Open Access Unconventional excited-state dynamics in the concerted benzyl (C7H7) radical self-reaction to anthracene (C14H10)
Unconventional excited-state dynamics in the concerted benzyl (C7H7) radical self-reaction to anthracene (C14H10)
The reaction of benzyl radical self-reaction to anthracene opens-up a previously overlooked avenue for a more efficient synthesis of aromatic, multi-ringed structures via excited state dynamics in the gas phase.
- Ralf. I. Kaiser
- , Long Zhao
- & Alexander M. Mebel
-
Article
| Open Access Insertion of ammonia into alkenes to build aromatic N-heterocycles
Insertion of ammonia into alkenes to build aromatic N-heterocycles
Aromatic heterocycles containing nitrogen are ubiquitous in biologically relevant small molecules. Here the authors show an unorthodox methodology for their synthesis, by inserting the nitrogen atom into a carbon ring, with ammonia in electrochemical conditions.
- Shuai Liu
- & Xu Cheng
-
Article
| Open Access Observation and rationalization of nitrogen oxidation enabled only by coupled plasma and catalyst
Observation and rationalization of nitrogen oxidation enabled only by coupled plasma and catalyst
Heterogeneous catalysts coupled with non-thermal plasma are known to achieve higher reaction yields, but disentangling the gain achieved by the individual components remains challenging. Here, NO is produced from N2 and O2 in a plasma-catalytic reactor at conditions at which neither catalyst nor plasma alone is productive, providing unambiguous evidence of plasma-catalyst synergy.
- Hanyu Ma
- , Rakesh K. Sharma
- & William F. Schneider
-
Article
| Open Access Catalytic atroposelective synthesis of axially chiral benzonitriles via chirality control during bond dissociation and CN group formation
Catalytic atroposelective synthesis of axially chiral benzonitriles via chirality control during bond dissociation and CN group formation
Nitriles have proven to be useful functional groups in pharmaceuticals and polymers, both as active moieties themselves or as easy precursors to further transformations, but synthesis of benzonitriles with axial chirality has often been difficult. Here the authors make axially chiral benzonitriles via N-heterocyclic carbene organocatalysis via a dynamic kinetic resolution process.
- Ya Lv
- , Guoyong Luo
- & Yonggui Robin Chi
-
Article
| Open Access SET activation of nitroarenes by 2-azaallyl anions as a straightforward access to 2,5-dihydro-1,2,4-oxadiazoles
SET activation of nitroarenes by 2-azaallyl anions as a straightforward access to 2,5-dihydro-1,2,4-oxadiazoles
Oxadiazoles are five-membered ring heterocycles comprising an oxygen and two nitrogens, and those with the 1,2,4-oxadiazole skeleton are of interest due to their prevalence in biologically active compounds. Here, the authors report a transition metal-free [3+2]-cycloaddition of nitroarenes with 2-azaallyl anions under basic conditions to access 2,5-dihydro-1,2,4-oxadiazoles.
- Dong Zou
- , Lishe Gan
- & Patrick J. Walsh
-
Article
| Open Access Proofreading experimentally assigned stereochemistry through Q2MM predictions in Pd-catalyzed allylic aminations
Proofreading experimentally assigned stereochemistry through Q2MM predictions in Pd-catalyzed allylic aminations
A predictive model has been created for a stereoselective palladium-catalysed allylic amination reaction. Derived only from quantum chemical data, the method is accurate enough to reveal multiple erroneous assignments in literature experiments.
- Jessica Wahlers
- , Jèssica Margalef
- & Per-Ola Norrby
-
Article
| Open Access Efficient synthesis of primary and secondary amides via reacting esters with alkali metal amidoboranes
Efficient synthesis of primary and secondary amides via reacting esters with alkali metal amidoboranes
Amides are important organic compounds that are widely applied in medicine, biochemistry, and materials science. Here, the authors report a method for synthesis of primary and secondary amides through a direct amidation of esters with sodium amidoboranes (NaNHRBH3, R = H, Me), at room temperature without the use of catalysts.
- Yu Guo
- , Ruo-Ya Wang
- & Xuenian Chen
-
Article
| Open Access Efficient access to aliphatic esters by photocatalyzed alkoxycarbonylation of alkenes with alkyloxalyl chlorides
Efficient access to aliphatic esters by photocatalyzed alkoxycarbonylation of alkenes with alkyloxalyl chlorides
Aliphatic esters are essential constituents of biologically active compounds but their preparation from readily available olefins remains challenging. Here the authors show a strategy to access aliphatic esters from olefins through a unique photocatalyzed alkoxycarbonylation reaction.
- Jian-Qiang Chen
- , Xiaodong Tu
- & Jie Wu
-
Article
| Open Access Facile generation of bridged medium-sized polycyclic systems by rhodium-catalysed intramolecular (3+2) dipolar cycloadditions
Facile generation of bridged medium-sized polycyclic systems by rhodium-catalysed intramolecular (3+2) dipolar cycloadditions
The bridged medium-sized ring bicyclo[m.n.2] family of natural products are commonly found but difficult to synthesize efficiently. Here the authors present a cascade reaction to form the carbon skeleton, via a [3+2] cycloaddition of a captured azavinyl carbene intermediate.
- Bao-Long Hou
- , Jonathan J. Wong
- & Chuang-Chuang Li
-
Article
| Open Access Identification of active catalysts for the acceptorless dehydrogenation of alcohols to carbonyls
Identification of active catalysts for the acceptorless dehydrogenation of alcohols to carbonyls
Identifying active catalysts for the conversion of alcohols into aldehydes or ketones and molecular hydrogen is highly desirable. Here the authors develop and validate against experiments a screening model based on DFT calculations and scaling relationships for identifying alcohol dehydrogenation catalysts.
- Tao Wang
- , Jin Sha
- & Carine Michel

 Modular assembly of indole alkaloids enabled by multicomponent reaction
Modular assembly of indole alkaloids enabled by multicomponent reaction