Key Points
-
The development of biologic DMARDs has revolutionized the therapeutic management of juvenile idiopathic arthritis (JIA)
-
A treat-to-target approach combined with tight control, in which patients are frequently reassessed, can help improve disease outcomes and achieve disease remission off medication
-
Initiating intensive therapy early in the course of JIA (during the window of opportunity) is likely to improve long-term disease outcomes, and is a focus of ongoing clinical trials
-
Guidelines and consensus protocols have been pivotal in improving the management of children with JIA, but further development and investigator-led clinical trials are required, including by multinational collaborations
-
Optimal management strategies for patients with JIA can be achieved by combining guideline-led care and personalized treatment approaches, and by focusing efforts on targeted treatment
Abstract
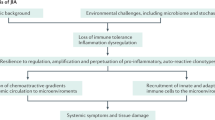
The treatment of juvenile idiopathic arthritis (JIA) is evolving. The growing number of effective drugs has led to successful treatment and prevention of long-term sequelae in most patients. Although patients with JIA frequently achieve lasting clinical remission, sustained remission off medication is still elusive for most. Treatment approaches vary substantially among paediatric rheumatologists owing to the inherent heterogeneity of JIA and, until recently, to the lack of accepted and well-evidenced guidelines. Furthermore, many pertinent questions related to patient management remain unanswered, in particular regarding treatment targets, and selection, intensity and sequence of initiation or withdrawal of therapy. Existing JIA guidelines and recommendations do not specify treat-to-target or tight control strategies, in contrast to adult rheumatology in which these approaches have been successful. The concepts of window of opportunity (early treatment to improve long-term outcomes) and immunological remission (abrogation of subclinical disease activity) are also fundamental when defining treatment methodologies. This Review explores the application of these concepts to JIA and their possible contribution to the development of future clinical guidelines or consensus treatment protocols. The article also discusses how diverse forms of standardized, guideline-led care and personalized treatment can be combined into a targeted, patient-centred approach to optimize management strategies for patients with JIA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Prakken, B., Albani, S. & Martini, A. Juvenile idiopathic arthritis. Lancet 377, 2138–2149 (2011).
Barnes, M. G. et al. Subtype-specific peripheral blood gene expression profiles in recent-onset juvenile idiopathic arthritis. Arthritis Rheum. 60, 2102–2112 (2009).
van den Ham, H. J., de Jager, W., Bijlsma, J. W., Prakken, B. J. & de Boer, R. J. Differential cytokine profiles in juvenile idiopathic arthritis subtypes revealed by cluster analysis. Rheumatology (Oxford) 48, 899–905 (2009).
Lehman, T. J. The future of pediatric rheumatology: many questions remain. Arthritis Rheum. 56, 2815–2816 (2007).
Guzman, J. et al. The outcomes of juvenile idiopathic arthritis in children managed with contemporary treatments: results from the ReACCh-Out cohort. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2014-205372.
Nordal, E. et al. Ongoing disease activity and changing categories in a long-term Nordic cohort study of juvenile idiopathic arthritis. Arthritis Rheum. 63, 2809–2818 (2011).
Oen, K. et al. Disease course and outcome of juvenile rheumatoid arthritis in a multicenter cohort. J. Rheumatol. 29, 1989–1999 (2002).
Selvaag, A. M. et al. Early disease course and predictors of disability in juvenile rheumatoid arthritis and juvenile spondyloarthropathy: a 3 year prospective study. J. Rheumatol. 32, 1122–1130 (2005).
Wallace, C. A., Huang, B., Bandeira, M., Ravelli, A. & Giannini, E. H. Patterns of clinical remission in select categories of juvenile idiopathic arthritis. Arthritis Rheum. 52, 3554–3562 (2005).
Ansell, B. M. & Swann, M. The management of chronic arthritis of children. J. Bone Joint Surg. Br. 65, 536–543 (1983).
Fries, J. F. Current treatment paradigms in rheumatoid arthritis. Rheumatology (Oxford) 39 (Suppl 1), 30–35 (2000).
Giannini, E. H. et al. Methotrexate in resistant juvenile rheumatoid arthritis. Results of the U.S.A.—U.S.S.R. double-blind, placebo-controlled trial. The Pediatric Rheumatology Collaborative Study Group and The Cooperative Children's Study Group. N. Engl. J. Med. 326, 1043–1049 (1992).
European Medicines Agency. Successes of the Paediatric Regulation after 5 years [online], (2013).
Stoll, M. L. & Cron, R. Q. Treatment of juvenile idiopathic arthritis: a revolution in care. Pediatr. Rheumatol. Online J. 12, 13 (2014).
Ruperto, N., Vesely, R., Saint-Raymond, A., Martini, A. & Paediatric Rheumatology International Trials Organization (PRINTO). Impact of the European paediatric legislation in paediatric rheumatology: past, present and future. Ann. Rheum. Dis. 72, 1893–1896 (2013).
Levinson, J. E. & Wallace, C. A. Dismantling the pyramid. J. Rheumatol. Suppl. 33, 6–10 (1992).
Recommendations of the German Society of Rheumatology for therapy with tumor necrosis factor inhibitors. Pharmacotherapy Committee of the German Society of Rheumatology [German]. Z. Rheumatol. 59, 291–292 (2000).
Tynjala, P. et al. Aggressive combination drug therapy in very early polyarticular juvenile idiopathic arthritis (ACUTE–JIA): a multicentre randomised open-label clinical trial. Ann. Rheum. Dis. 70, 1605–1612 (2011).
Wallace, C. A. et al. Trial of early aggressive therapy in polyarticular juvenile idiopathic arthritis. Arthritis Rheum. 64, 2012–2021 (2012).
Smolen, J. S. et al. Treating rheumatoid arthritis to target: recommendations of an international task force. Ann. Rheum. Dis. 69, 631–637 (2010).
Consolaro, A. et al. Toward a treat-to-target approach in the management of juvenile idiopathic arthritis. Clin. Exp. Rheumatol. 30, S157–S162 (2012).
Smolen, J. S. Treat-to-target: rationale and strategies. Clin. Exp. Rheumatol. 30, S2–S6 (2012).
Schoels, M. et al. Evidence for treating rheumatoid arthritis to target: results of a systematic literature search. Ann. Rheum. Dis. 69, 638–643 (2010).
Grigor, C. et al. Effect of a treatment strategy of tight control for rheumatoid arthritis (the TICORA study): a single-blind randomised controlled trial. Lancet 364, 263–269 (2004).
Felson, D. T. et al. American College of Rheumatology/European League against Rheumatism provisional definition of remission in rheumatoid arthritis for clinical trials. Ann. Rheum. Dis. 70, 404–413 (2011).
Smolen, J. S. et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs. Ann. Rheum. Dis. 69, 964–975 (2010).
Smolen, J. S. et al. EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs: 2013 update. Ann. Rheum. Dis. 73, 492–509 (2014).
Consolaro, A. et al. Remission, minimal disease activity, and acceptable symptom state in juvenile idiopathic arthritis: defining criteria based on the juvenile arthritis disease activity score. Arthritis Rheum. 64, 2366–2374 (2012).
Wallace, C. A. et al. American College of Rheumatology provisional criteria for defining clinical inactive disease in select categories of juvenile idiopathic arthritis. Arthritis Care Res. (Hoboken) 63, 929–936 (2011).
Wallace, C. A., Ruperto, N. & Giannini, E. Preliminary criteria for clinical remission for select categories of juvenile idiopathic arthritis. J. Rheumatol. 31, 2290–2294 (2004).
Foell, D. et al. Methotrexate withdrawal at 6 vs 12 months in juvenile idiopathic arthritis in remission: a randomized clinical trial. JAMA 303, 1266–1273 (2010).
Hinze, C. et al. The role of serum S100A12 protein levels in maintaining inactive disease on anti-tumor necrosis factor therapy in polyarticular forms of juvenile idiopathic arthritis [abstract A68]. Arthritis Rheumatol. 66 (Suppl. 11), S99–S100 (2014).
Hinze, C. et al. The role of serum S100A12 protein levels in disease flare after withdrawal of anti-tumor necrosis factor therapy in polyarticular forms of juvenile idiopathic arthritis [abstract A12]. Arthritis Rheumatol. 66 (Suppl. 11), S19–S20 (2014).
Prevoo, M. L. et al. Modified disease activity scores that include twenty-eight-joint counts. Development and validation in a prospective longitudinal study of patients with rheumatoid arthritis. Arthritis Rheum. 38, 44–48 (1995).
Consolaro, A. et al. Development and validation of a composite disease activity score for juvenile idiopathic arthritis. Arthritis Rheum. 61, 658–666 (2009).
Bulatovic Calasan, M., de Vries, L. D., Vastert, S. J., Heijstek, M. W. & Wulffraat, N. M. Interpretation of the Juvenile Arthritis Disease Activity Score: responsiveness, clinically important differences and levels of disease activity in prospective cohorts of patients with juvenile idiopathic arthritis. Rheumatology (Oxford) 53, 307–312 (2014).
McErlane, F. et al. Validity of a three-variable Juvenile Arthritis Disease Activity Score in children with new-onset juvenile idiopathic arthritis. Ann. Rheum. Dis. 72, 1983–1988 (2013).
Wittkowski, H. et al. S100A12 is a novel molecular marker differentiating systemic-onset juvenile idiopathic arthritis from other causes of fever of unknown origin. Arthritis Rheum. 58, 3924–3931 (2008).
Gerss, J. et al. Phagocyte-specific S100 proteins and high-sensitivity C reactive protein as biomarkers for a risk-adapted treatment to maintain remission in juvenile idiopathic arthritis: a comparative study. Ann. Rheum. Dis. 71, 1991–1997 (2012).
Miossec, P. et al. Biomarkers and personalised medicine in rheumatoid arthritis: a proposal for interactions between academia, industry and regulatory bodies. Ann. Rheum. Dis. 70, 1713–1718 (2011).
Hunter, P. J. & Wedderburn, L. R. Pediatric rheumatic disease: can molecular profiling predict the future in JIA? Nat. Rev. Rheumatol. 5, 593–594 (2009).
Kessel, C., Holzinger, D. & Foell, D. Phagocyte-derived S100 proteins in autoinflammation: putative role in pathogenesis and usefulness as biomarkers. Clin. Immunol. 147, 229–241 (2013).
Rothmund, F. et al. Validation of relapse risk biomarkers for routine use in patients with juvenile idiopathic arthritis. Arthritis Care Res. (Hoboken) 66, 949–955 (2014).
Collado, P. et al. Detection of synovitis by ultrasonography in clinically inactive juvenile idiopathic arthritis on and off medication. Clin. Exp. Rheumatol. 32, 597–603 (2014).
Javadi, S., Kan, J. H., Orth, R. C. & DeGuzman, M. Wrist and ankle MRI of patients with juvenile idiopathic arthritis: identification of unsuspected multicompartmental tenosynovitis and arthritis. AJR Am. J. Roentgenol. 202, 413–417 (2014).
Vastert, S. J. et al. Effectiveness of first line use of recombinant IL-1RA treatment in steroid naive systemic juvenile idiopathic arthritis: results of a prospective cohort study. Arthritis Rheumatol. http://dx.doi.org/10.1002/art.38296.
Nigrovic, P. A. Review: is there a window of opportunity for treatment of systemic juvenile idiopathic arthritis? Arthritis Rheumatol. 66, 1405–1413 (2014).
van Rossum, M. A. et al. Long-term outcome of juvenile idiopathic arthritis following a placebo-controlled trial: sustained benefits of early sulfasalazine treatment. Ann. Rheum. Dis. 66, 1518–1524 (2007).
Ting, T. V. & Lovell, D. J. Does early sulfasalazine treatment provide long-term benefits to patients with juvenile idiopathic arthritis? Nat. Clin. Pract. Rheumatol. 4, 344–345 (2008).
Hissink Muller, P. C. E. et al. A comparison of three treatment strategies in recent onset DMARD naïve juvenile idiopathic arthritis: 3-months results of the BeSt for Kids-Study. Late breaking abstracts presented at the ACR Annual Meeting 2014 [online], (2014).
Institute of Medicine (US) Committee on Standards for Developing Trustworthy Clinical Practice Guidelines (eds Graham, R. et al.). Clinical Practice Guidelines we can Trust (National Academies Press, 2011).
McMaster, P., Rogers, D., Kerr, M. & Spencer, A. Getting guidelines to work in practice. Arch. Dis. Child. 92, 104–106 (2007).
Beukelman, T. et al. 2011 American College of Rheumatology recommendations for the treatment of juvenile idiopathic arthritis: initiation and safety monitoring of therapeutic agents for the treatment of arthritis and systemic features. Arthritis Care Res. (Hoboken) 63, 465–482 (2011).
Dueckers, G. et al. Evidence and consensus based GKJR guidelines for the treatment of juvenile idiopathic arthritis. Clin. Immunol. 142, 176–193 (2012).
Martini, A. JIA in 2011: New takes on categorization and treatment. Nat. Rev. Rheumatol. 8, 67–68 (2012).
Huppertz, H. I. Recommendations for juvenile idiopathic arthritis by the American College of Rheumatology: comment on the article by Beukelman. et al. Arthritis Care Res. (Hoboken) 63, 1354–1355 (2011).
Ringold, S. et al. Childhood arthritis and rheumatology research alliance consensus treatment plans for new onset polyarticular juvenile idiopathic arthritis. Arthritis Care Res. (Hoboken) 66, 1063–1072 (2013).
Davies, K. et al. BSPAR Standards of Care for children and young people with juvenile idiopathic arthritis. Rheumatology (Oxford) 49, 1406–1408 (2010).
Wallace, C. A. Developing standards of care for patients with juvenile idiopathic arthritis. Rheumatology (Oxford) 49, 1213–1214 (2010).
Hull, R. G. & British Paediatric Rheumatology Group. Guidelines for management of childhood arthritis. Rheumatology (Oxford) 40, 1309–1312 (2001).
Ringold, S. et al. 2013 update of the 2011 American College of Rheumatology recommendations for the treatment of juvenile idiopathic arthritis: recommendations for the medical therapy of children with systemic juvenile idiopathic arthritis and tuberculosis screening among children receiving biologic medications. Arthritis Rheum. 65, 2499–2512 (2013).
Eccleston, C. et al. Psychological therapies for the management of chronic and recurrent pain in children and adolescents. Cochrane Database of Systematic Reviews, Issue 12, Art. No.: CD003968. http://dx.doi.org/10.1002/14651858.CD003968.pub3.
Hawke, F., Burns, J., Radford, J. A. & du Toit, V. Custom-made foot orthoses for the treatment of foot pain. Cochrane Database of Systematic Reviews, Issue 3. Art. No.: CD006801. http://dx.doi.org/10.1002/14651858.CD006801.pub2.
Rome, K., Ashford, R. L. & Evans, A. Non-surgical interventions for paediatric pes planus. Cochrane Database of Systematic Reviews, Issue 7. Art. No.: CD006311. http://dx.doi.org/10.1002/14651858.CD006311.pub2.
Takken, T. et al. Exercise therapy in juvenile idiopathic arthritis. Cochrane Database of Systematic Reviews, Issue 16, Art. No.: CD005954. http://dx.doi.org/10.1002/14651858.CD005954.pub2.
Takken, T., Van Der Net, J. & Helders, P. J. Methotrexate for treating juvenile idiopathic arthritis. Cochrane Database of Systematic Reviews, Issue 4. Art. No.: CD003129. http://dx.doi.org/10.1002/14651858.CD003129.
Wallen, M. & Gillies, D. Intra-articular steroids and splints/rest for children with juvenile idiopathic arthritis and adults with rheumatoid arthritis. Cochrane Database of Systematic Reviews, Issue 1. Art. No.: CD002824. http://dx.doi.org/10.1002/14651858.CD002824.pub2.
Institute of Medicine (US) Committee on Quality of Health Care in America. Crossing the Quality Chasm: a New Health System for the 21st Century (National Academies Press, 2001).
Goldberger, J. J. & Buxton, A. E. Personalized medicine vs guideline-based medicine. JAMA 309, 2559–2560 (2013).
Krieckaert, C. L. et al. Personalised treatment using serum drug levels of adalimumab in patients with rheumatoid arthritis: an evaluation of costs and effects. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2013-204101.
Malik, N. N. Biologics: personalized drugs should cut care costs. Nature 485, 582 (2012).
Burnett, H. F., Regier, D. A., Feldman, B. M., Miller, F. A. & Ungar, W. J. Parents' preferences for drug treatments in juvenile idiopathic arthritis: a discrete choice experiment. Arthritis Care Res. (Hoboken) 64, 1382–1391 (2012).
Becker, M. L. Pharmacogenomics in pediatric rheumatology. Curr. Opin. Rheumatol. 24, 541–547 (2012).
Ellis, J. A. et al. CLARITY—Childhood Arthritis Risk factor Identification Study. Pediatr. Rheumatol. Online J. 10, 37 (2012).
Oen, K. et al. Predictors of early inactive disease in a juvenile idiopathic arthritis cohort: results of a Canadian multicenter, prospective inception cohort study. Arthritis Rheum. 61, 1077–1086 (2009).
Cobb, J. E., Hinks, A. & Thomson, W. The genetics of juvenile idiopathic arthritis: current understanding and future prospects. Rheumatology (Oxford) 53, 592–599 (2014).
Hawtree, S., Muthana, M. & Wilson, A. G. The role of histone deacetylases in rheumatoid arthritis fibroblast-like synoviocytes. Biochem. Soc. Trans. 41, 783–788 (2013).
Wellcome Trust Sanger Institute. yourgenome.org [online], (2014).
Szekanecz, Z. et al. Pharmacogenetics and pharmacogenomics in rheumatology. Immunol. Res. 56, 325–333 (2013).
Smith, S. L., Plant, D., Eyre, S. & Barton, A. The potential use of expression profiling: implications for predicting treatment response in rheumatoid arthritis. Ann. Rheum. Dis. 72, 1118–1124 (2013).
Smolen, J. S. & Aletaha, D. Forget personalised medicine and focus on abating disease activity. Ann. Rheum. Dis. 72, 3–6 (2013).
Wilke, W. S. Reply to: forget personalized medicine and focus on abating disease activity. Ann. Rheum. Dis. 72, e8 (2013).
Dunn, G., Emsley, R., Liu, H. & Landau, S. Integrating biomarker information within trials to evaluate treatment mechanisms and efficacy for personalised medicine. Clin. Trials 10, 709–719 (2013).
Tak, P. P. A personalized medicine approach to biologic treatment of rheumatoid arthritis: a preliminary treatment algorithm. Rheumatology (Oxford) 51, 600–609 (2012).
Bendtzen, K. Personalized medicine: theranostics (therapeutics diagnostics) essential for rational use of tumor necrosis factor-α antagonists. Discov. Med. 15, 201–211 (2013).
Spamer, M. et al. Physiotherapy for juvenile idiopathic arthritis [German]. Z. Rheumatol. 71, 387–395 (2012).
Lien, G. et al. Frequency of osteopenia in adolescents with early-onset juvenile idiopathic arthritis: a long-term outcome study of one hundred five patients. Arthritis Rheum. 48, 2214–2223 (2003).
Consolaro, A. & Ravelli, A. Paediatric rheumatology: juvenile idiopathic arthritis—are biologic agents effective for pain? Nat. Rev. Rheumatol. 9, 447–448 (2013).
La Hausse de Lalouviere, L., Ioannou, Y. & Fitzgerald, M. Neural mechanisms underlying the pain of juvenile idiopathic arthritis. Nat. Rev. Rheumatol. 10, 205–211 (2014).
Taylor, M. J. et al. Systematic review of the application of the plan-do-study-act method to improve quality in healthcare. BMJ Qual. Saf. 23, 290–298 (2014).
Acknowledgements
F.G. is supported by the European Union Seventh Framework Programme for Research and Technological Development (FP7) grant EUTRAIN (European Translational Training for Autoimmunity & Immune Manipulation Network, ref. 289903). D.F. is supported by the FP7 grant MIAMI (Monitoring Innate Immunity in Arthritis and Mucosal Inflammation project, ref. 305266) and by a Clinical Research Award (CRA-04) from the Interdisciplinary Centre of Clinical Research (IZKF), Münster, Germany.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, made substantial contributions to the discussion of content, wrote the article and reviewed/edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
C.H. declares that he has received lecture fees from Novartis. D.F. declares that he has received lecture fees from Chugai, Novartis, Pfizer, Roche and Swedish Orphan Biovitrum. F.G. declares no competing interests.
Rights and permissions
About this article
Cite this article
Hinze, C., Gohar, F. & Foell, D. Management of juvenile idiopathic arthritis: hitting the target. Nat Rev Rheumatol 11, 290–300 (2015). https://doi.org/10.1038/nrrheum.2014.212
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2014.212
This article is cited by
-
Significant pain decrease in children with non-systemic Juvenile Idiopathic Arthritis treated to target: results over 24 months of follow up
Pediatric Rheumatology (2023)
-
Treat-to-Target in Pediatric Rheumatic Diseases
Current Rheumatology Reports (2023)
-
Enthesitis-related arthritis: the clinical characteristics and factors related to MRI remission of sacroiliitis
BMC Musculoskeletal Disorders (2022)
-
Looking for the best strategy to treat children with new onset juvenile idiopathic arthritis: presentation of the “comparison of STep-up and step-down therapeutic strategies in childhood ARthritiS” (STARS) trial
Pediatric Rheumatology (2022)
-
Secondary consequences of juvenile idiopathic arthritis in children and adolescents with knee involvement: physical activity, adiposity, fitness, and functional performance
Rheumatology International (2022)