Abstract
Prognostic factors for overall survival (OS), percutaneous endoscopic gastrostomy (PEG) dependency, and long-term speech rehabilitation via voice prosthesis (VP) after laryngectomy for laryngeal or hypopharyngeal cancer were investigated in a retrospective population-based study in Thuringia, Germany. A total of 617 patients (68.7% larynx; hypopharynx; 31.3%; 93.7% men; median age 62 years; 66.0% stage IV) from 2001 to 2020 were included. Kaplan–Meier and Cox multivariable regression analyses were performed. 23.7% of patients received a PEG. 74.7% received a VP. Median OS was 131 months. Independent factors for lower OS were stage IV (compared to stage II; hazard ratio [HR] = 3.455; confidence interval [CI] 1.395–8.556) and laryngectomy for a recurrent disease (HR = 1.550; CI 1.078–2.228). Median time to PEG removal was 7 months. Prior partial surgery before laryngectomy showed a tendency for independent association for later PEG removal (HR = 1.959; CI 0.921–4.167). Postoperative aspiration needing treatment was an independent risk factor (HR = 2.679; CI 1.001–7.167) for later definitive VP removal. Laryngectomy continuously plays an important role in a curative daily routine treatment setting of advanced laryngeal or hypopharyngeal cancer in Germany. Long-term dependency on nutrition via PEG is an important issue, whereas use of VP is a stable long-term measure for voice rehabilitation.
Similar content being viewed by others
Introduction
Total laryngectomy (in the following only laryngectomy) is one primary curative treatment option for advanced laryngeal and hypopharyngeal cancer1,2. There is an ongoing debate, if definitive radiochemotherapy is the better alternative3. When comparing both therapy strategies, it is important to regard both oncological results like overall survival (OS), but also the long-term functional outcome. Especially, voice and swallowing function can be disturbed after all curative, surgical or non-surgical approaches. Laryngectomy is followed by loss of the natural voice. High standard of speech rehabilitation is phonation through a tracheoesophageal puncture (TEP) with a voice prosthesis. It has clearly been shown that a TEP is leading to a significant increase in quality of life2. Chronic dysphagia due to narrowing of the neopharyngeal or proximal esophageal lumen rarely occurs due to stricture formation4. But chronic dysphagia and chronic voice disorder are also very common after radiochemotherapy for advanced laryngeal and hypopharyngeal cancer5,6.
The current knowledge on oncological and functional outcome on locally advanced laryngeal and hypopharyngeal cancer is mainly based on large multicenter prospective phase III clinical trial or on retrospective multicenter cohorts on specialized cancer centers3,7,8,9. The former have the disadvantage of patient selection in a highly standardized setting, are focused on non-surgical treatment, but mostly do not address long-term effects. In contrast, retrospective trials may be the subject to a selection bias. Population-based studies have the advantage to include all cancer patients within a given jurisdiction, are therefore less prone to selection and referral biases. Moreover, they reflect the routine cancer care10. The National Cancer Institute’s Surveillance, Epidemiology, and End Results (SEER) registry, the National Cancer Database (NCDB), or national cancer registries record patient and tumor characteristics as well as survival data, but typically not data on functional outcome11,12,13. To overcome this limitation, all eight Department of Otorhinolaryngology, Head and Neck Surgery in Thuringia, a federal state in Germany, have established the Thuringian Head And Neck Cancer Study (THANCS) Group allowing population-based analysis of all head and neck cancer patients treated in one German federal state with about two million inhabitants14,15. Furthermore, the network established a linkage between data from all five Thuringian cancer registries and the data of the patients’ charts in the eight hospitals, i.e. a linkage between registry-based and hospital-based data. This allows a linkage to data not recorded in cancer registries16,17.
Using the described population-based approach, data of patients with advanced laryngeal and hypopharyngeal cancer were linked to data related to voice and swallowing rehabilitation in all patients who received a laryngectomy between 2001 and 2020 in the entire population of Thuringia. This allowed a comprehensive analysis of the functional and oncological outcomes in a clinical-routine setting beyond clinical trials and specialized cancer centers.
Methods
Study design
This population-based, retrospective study included all patients with laryngeal or hypopharyngeal cancer undergoing laryngectomy between 2001 and 2020 (20 years) in Thuringia, Germany. All patients with the following operation and procedure classification system (OPS; version 20) codes were included: 5–303.0 to 5–303.07; 5–303.1 to 5–303.17; 5–303.2 to 5–303.27). As one hospital (Arnstadt) did not perform laryngectomies in this selected time period, this study included all 617 patients from the remaining seven department of otolaryngology in Thuringia (Bad Salzungen, Erfurt, Gera, Jena, Nordhausen, Suhl, Weimar). The patient data set was double-checked against the data sets of the five Thuringian cancer registries (Erfurt, Gera, Jena, Nordhausen, Suhl). So, no patient was overlooked. The patients’ charts were reviewed. All relevant parameters (patient characteristics, tumor-specific data, surgery, aftercare, voice rehabilitation, nutrition, and follow-up) were anonymously recorded in a data base.
All experimental procedures with human subjects followed the institutional research committee's ethical standards and the 1964 Helsinki Declaration and its later amendments. The Ethics Committee of the Jena University Hospital approved the retrospective study of clinical routine data (No. 2018-1075-Material; No. 2022-2526-BO). Informed consent of the patients was waived by the approving ethics committee, as this study had a non-interventional retrospective design and all data were analyzed anonymously.
Statistical analysis
The statistical analyses were performed with SPSS Statistics version 29.0.0.0 (IBM Deutschland GmbH, 71139 Ehningen, Germany). Nominal and ordinal data are presented as absolute number and percentage. Metric data are presented as mean, standard deviation, median, and range. Time-dependent statistics were needed for the three primary outcome parameters (OS, percutaneous endoscopic gastrostomy [PEG] dependency, maintenance of voice prosthesis use), because of the variable follow-up time of the patients. First, Kaplan–Meier statistics and the log-rank test were used to analyze to impact of variables on OS, duration of the use of a PEG, and maintenance of voice prosthesis use. The significance level was set to p ≤ 0.05. All significant factors from these univariate analyses were included in multivariate analyses using Cox proportional hazard ratio (HR) with corresponding 95% confidence intervals (CI). Furthermore, for the comparisons between patients with laryngeal versus hypopharyngeal cancer, Pearson’s chi-square test or Fisher’s exact test were needed for the nominal/ordinal data. The Mann–Whitney U-test was used to compare metric data. The significance level was again set to p ≤ 0.05.
The epidemiological calculations were performed using the absolute case numbers and data from the Thuringian State Office of Statistics (https://www.statistik.thueringen.de/) on the population of Thuringia during the observation period. The incidences of laryngectomy (surgical rate per 100,000 persons) per year and gender were calculated.
Ethics statement
The Ethics Committee of the Jena University Hospital approved the retrospective study of clinical routine data (No. 2018-1075-Material; No. 2022-2526-BO). Informed consent of the patients was waived, as this study had a non-interventional retrospective design and all data were analyzed anonymously.
Results
Patient’s and tumor characteristics, treatment and complications during long-term follow-up
The characteristics of the patients undergoing laryngectomy for laryngeal or hypopharyngeal cancer in Thuringia in 2001–2020 are shown in Table 1. A total of 617 patients were included in the study. Most patients (93.7%) were male. The median age was 62 years). Three quarters were smokers prior to laryngectomy (77.5%). Four out of five patients were drinking alcohol (82.2). About two third had the primary tumor in the larynx (68.7%) and about one third in the hypopharynx (31.3%). Most frequent tumors were pT4 (45.1%), pN0 (50.2%). A minority had distant metastasis at the time of diagnosis (M+; 2.4%). This resulted in a UICC stage IV in the majority of patients (66.0%).
The treatment characteristics are listed in Table 2. Three quarters received the laryngectomy as primary treatment (75.7%). One quarter was treated for a recurrent tumor (24.3%). Laryngectomy was performed without resection of parts of the pharynx in 57.5%, and with pharyngectomy in 42.5% of the cases. Only a minority of patients needed a reconstruction of the neopharynx with a flap (13.0%). Nearly all patients underwent bilateral neck dissection (96.8%). Three quarters (73.4%) needed a postoperative adjuvant treatment (radiotherapy, radiochemotherapy, or chemotherapy). Chemotherapy as single postoperative therapy was used in some M+ patients. The postoperative complications are summarized in Supplement Table 1. The two most frequent complications were: a pharyngocutaneous fistula (20.4%) and disturbed wound healing (19.0%).
Nutrition was ensured by a nasogastric feeding tube (NGT; median time of feeding: 10 days) in the majority of the patients (72.8%). 23.7% were fed via a PEG (median time of feeding: 3 months). Data on rehabilitation including speech rehabilitation are listed in Supplement Table 2. 461 patients (74.7%) received a tracheoesophageal puncture and a voice prosthesis for speech rehabilitation. Half of the patients learned esophageal speech (52.7%). Median time of inpatient speech therapy was four days.
Long-term complications are seen in Supplement Table 2. 12.5% of the patient developed a tumor recurrence during the follow-up time. 15.4% developed a secondary primary (mainly lung cancer, 7.8%). 22.9% of the patients died. Chronic or recurrent dysphagia (43.9%) and chronic neck pain (30.1%) were the two most frequent chronic complaints. Table 3 gives an overview about complications related to the voice prosthesis placement and related surgery. The most frequent problems were: leakage through the prosthesis (38.2%), recurrent dysphonia (33.8%), and obstruction of the prosthesis (24.9%). The three most frequent voice related treatments were: dilatation of the esophagus (13.0%), tracheostomy revision (12.6%), and botulinumtoxin injection into the parapharyngeal musculature (10.2%). During follow-up, the median number of voice prosthesis changes was two. The median time to the first change was 7 months. 6.5% of the patients needed definitive removal of the prosthesis and closure of the puncture. The median time to definitive removal in this subgroup of patients was 15.5 months.
Overall survival
The median follow-up time of all patients was 16 months. The median follow-up time of all patients still alive was 17 months. The 5-year OS rate of all patients was 67.2%. The results of the univariate analyses of factors associated to worse OS is presented in Supplement Table 4. Male patients, higher pT classification, higher pN classification, M+, higher UICC stage, and laryngectomy for tumor recurrence were related to worse OS (all p < 0.05; Fig. 1). The multivariate analyses are presented in Table 4. Model 1 included the TNM classification, model 2 the UICC stage. In model 1, pT4 had significantly lower OS than pT2 (HR = 1.947; CI 1.029–3.686; p = 0.041). pN2 had lower OS than pN0 (HR = 1.891; CI 1.304–2.741; p = 0.001). Laryngectomy of a recurrent tumor was associated with lower OS than primary laryngectomy (HR = 1.711; CI 1.167–2.508; p = 0.006). In model 2, stage IV showed lower OS than stage II (HR = 3.455; CI 1.395–8.556; p = 0.007). And again, laryngectomy for a recurrent tumor was followed by lower OS than primary laryngectomy (HR = 1.550; CI 1.078–2.228; p = 0.018).
Duration of nutrition via PEG
The results of the univariate analyses of factors associated to earlier removal of the PEG and return to oral nutrition over time are shown in Supplement Table 5. The overall 6-month and 12-month PEG removal rates were 42.7% and 84.7%, respectively. None smoking, laryngeal cancer, laryngectomy for primary tumor, and no prior partial resection were the factors associated to earlier removal (all p < 0.05; Fig. 2). The multivariate analyses are presented in Table 5. None of the factors revealed in univariate analysis was an independent factor for earlier removal of the PEG. At most, patients without prior partial surgery before laryngectomy compared to prior surgery showed a tendency for independent association for earlier PEG removal (HR = 1.959; CI 0.921–4.167; p = 0.081).
Long-term maintenance of the voice prosthesis use
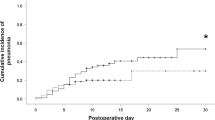
The voice prosthesis was removed during the follow-up in 6.5% of patients. The 5-year definitive voice prosthesis maintenance rate was 89.4%. The results of the univariate analyses of factors associated to long-term use of the voice prosthesis is presented in Supplement Table 6. Adjuvant postoperative chemotherapy, less than the median of two voice prosthesis changes, serious postoperative aspiration needing treatment, severe wound healing disorders after laryngectomy, and therapy-relevant neck swelling after surgery resulted in a higher risk for definitive voice prosthesis removal during follow-up (all p < 0.05; Fig. 3). The results of the multivariate analyses are presented in Table 6. Three models were calculated. The first model included all significant factors from the univariate analysis. Model 2 omitted the factor number of voice prosthesis changes as permanent removal might be directly related to a lower number of changes. Model 3 also omitted the factor postoperative adjuvant treatment as only 2.4% of the patients received a postoperative chemotherapy as monotherapy. In model 1, only less changes than the median of 2 was associated to higher risk of definitive removal (HR = 2.481; CI 1.063–5.786; p = 0.035). In model 2, postoperative aspiration needing treatment was an independent risk factor (HR = 2.679; CI 1.001–7.167; p = 0.050). The same result was seen in model 3 (HR = 3.126; CI 1.207–8.095; p = 0.019).
Maintenance of the tracheoesophageal puncture and of the voice prosthesis, Kaplan–Meier curves and influencing factors. (A) All patients with voice prosthesis. (B) <> median number of voice prosthesis changes (<> 2). (C) Postoperative aspiration needing treatment (no/yes). (D) Postoperative neck swelling needing treatment (no/yes).
Incidence of laryngectomy between 2001 and 2020
The Thuringian population varied between 2001 and 2020 between 2,120,237 and 2,431,255 people (female: 1,071,025–1,241,304; male: 1,049,212–1,189,951). The average incidence of laryngectomy in the observed 20 years was 1.32 ± 0.51 (Supplement Table 7). The laryngectomy rate was considerably higher in men (2.51 ± 1.01) than in women (0.17 ± 0.14). The laryngectomy rate was double as high in patients with advanced laryngeal cancer (1.85 ± 4.17) than in patients with hypopharyngeal cancer (0.85 ± 1.90). The incidence rate varied over the years in both genders and tumor locations, but a decrease over time was not seen (Fig. 4).
Comparison of patients with laryngeal cancer versus patients with hypopharyngeal cancer
A comparison of the two laryngectomy groups was not the primary objective of the present study. Explorative data on the comparisons between patients with laryngeal cancer and hypopharyngeal cancer are summarized in the Supplement Tables 8–14. The groups showed some differences. Briefly, patients with hypopharyngeal cancer had a higher N classification, a higher UICC staging, were younger, had less frequently a prior therapy, and a higher probability for postoperative adjuvant therapy than patients with laryngeal cancer (all p < 0.05). The occurrence of most voice prosthesis-related complications and treatments were not different, except for a lower frequency of tracheostomy revisions (p = 0.019) and need for botulinumtoxin injection (p = 0.025) in patients with hypopharyngeal cancer. Postoperative bleeding was more frequent after laryngectomy for hypopharyngeal cancer (p = 0.026). Some long-term complications were more frequent in patients with laryngeal cancer: more chronic/recurrent dysphagia (p = 0.020), more frequently tracheostomy dysfunction (p = 0.018), but less frequent chronic neck pain (p = 0.008).
Discussion
Laryngectomy with neck dissection followed by post-operative radiotherapy or radiochemotherapy is a standard treatment option for patients with locally advanced laryngeal or hypopharyngeal cancer. Despite the establishment of organ preservation strategies by radiochemotherapy, the use of laryngectomy is not declining18,19. Nevertheless, epidemiology studies of the general trend of advanced laryngeal and hypopharyngeal cancer undergoing laryngectomy in a population-based setting are sparse. The present study provides important population-based and therefore real world setting insights into the oncological and at the same time functional outcome after laryngectomy. Concerning the functional evaluation, we are not aware of any other comparable population-based study.
The incidence numbers over time showed that laryngectomy is still and consistently an important treatment option for advanced laryngeal and hypopharyngeal cancer in Germany. When laryngectomy is compared to non-surgical approaches in population-based studies, often only relative numbers in percentage are reported18,19,20. At least, the use of laryngectomy for advanced laryngeal cancer seems not to decrease in the United States19,20. In contrast, a relative decrease of the use of laryngectomy is reported for hypopharyngeal cancer in the Netherlands12.
The 5-year OS of all patients was 67.2%. This is comparable to results of other population-based studies on laryngectomy ± postoperative radio(chemo)therapy12,13,19,20,21,22, or even better than in some populations23. Of course, such comparisons of the results of different studies should be viewed with caution, because a relevant selection bias cannot be excluded. Anyhow, in accordance to other studies, higher tumor staging and treatment for a recurrent tumor were to most predictive factor for lower OS also in the present study19,20,24.
Prospective trials of post-laryngectomy complications are sparse18,25. This makes the population-based approach even more meaningful. The rates for the two most frequent complications, i.e. pharyngocutaneous fistula (20.4%) and disturbed wound healing (19.0%) were not higher than in other retrospective hospital-based studies2,26. Laryngectomy and postoperative radiotherapy have several effects on swallowing function because of the anatomic changes due to the neopharynx construction, and the intrapharyngeal pressure changes27. The altered swallowing dynamics and stenosis are the most important causes of dysphagia27,28. Post-laryngectomy dysphagia rates vary from 10 to 62%27. As it is standard, most patients (72.8%) in the present study only needed about 10 days of nutrition by a nasogastric tube to overcome the early postoperative phase. Nevertheless, about a quarter (23.7%) needed a PEG associated with a 12-month PEG removal rates of 84.7%. This rate was also seen in a recent population-based study from the United States18. Here, 14% and 7%, respectively, of patients who received a radiochemotherapy or a total laryngectomy were still PEG dependent after 12 months. Hence, the gastrostomy tube dependence seems to be lower than after radiochemotherapy for this type of advanced cancer18,29. Furthermore, chronic or recurrent dysphagia was reported by 36.8% of the patients, i.e. this was not only an issue for the patients needing a PEG. Whereas the univariate analysis showed some factors with influence on earlier removal (no smoking, primary tumor treatment, no prior partial resection), the multivariate analysis did not reveal an independent factor. Hence, longer dependency on the PEG seems to be multifactorial. Even, if not needed anymore, it could take time until the PEG is removed.
Tracheoesophageal speech through a tracheoesophageal fistula and voice prosthesis is the best speech rehabilitation method according to acoustic and perceptual outcomes30. We could not identify any population-based study on this topic. The voice prosthesis implantation rate was 74.7%. This number seems to be high but other population-based studies addressing this issue are missing. The rate is considerably higher than reported for the United States31. The clear majority received a primary TEP (97%). This is in contrast to other larger series, demonstrating a much more frequent use of secondary TEP31,32. Prospective trials typically investigate a new type of voice prosthesis33. Despite advances in voice prosthesis design, magnetized valves, and antimicrobial materials, device complications still are common28. The prostheses last only a limited period of time, and require repeated replacements on average every 2–3 months34. The average life time of the first voice prosthesis in the present study was 7 months. This is long compared to other studies32,35,36, and speaks for a good instruction of the laryngectomees how to handle the prosthesis28. Leakage through the prosthesis (38.2%) followed by obstruction of the prosthesis (24.9%) were the most frequent indications for replacement like showed also in other studies28,37,38. Data on long term use and maintenance of the TEP are sparse and we were not aware of any population-based study. The 5-year definitive voice prosthesis maintenance rate was 89.4%. During follow-up, the TEP was definitively closed in 6.5% of the patients. This seems to be in the range of the data of specialized centers37.
The present study was limited by its retrospective character. The follow-up evaluation was dependent on outpatient visits of the patients in the hospital. Voice prosthesis might have been changed elsewhere, too. Therefore, we probably underestimated the number of prosthesis changes (which should be taken into account, when interpreting the multivariate model 1 in Table 6). Hence, an analysis of the causal relationship between the detected predictive factors and outcome was not possible. Furthermore, several factors that might have influence on oncological and functional outcome, for instance like comorbidity, body mass index and socioeconomic factors, were not analyzed but would be of interest for future studies39,40,41. To directly compare laryngectomy ± postoperative radio(chemo)therapy to non-surgical treatment concepts is difficult in a retrospective population-based setting due to the selection bias19,20, but is planned as a next study in the Thuringian setting.
Conclusions
This retrospective, population-based study describes the oncological and the functional outcome of 617 patients with laryngeal or hypopharyngeal cancer undergoing total laryngectomy in Thuringia between 2001 and 2020. The study comprises a non-selected federal state-wide setting over a period of 20 years, and provides a comprehensive presentation of the trends in incidence, overall survival, and function of laryngeal or hypopharyngeal cancer undergoing laryngectomy in Germany. The surgical rates remained stable over the last 10 years reflecting the unchanged importance of the procedure. Speech rehabilitation with voice prosthesis is standard of care for most patients with a very high success rate. Transient nutrition via percutaneous endoscopic gastrostomy is important for about a quarter of the patients, but removal and return to oral nutrition can be seen within a few months after surgery.
Data availability
The original contributions presented in the study are included in the article and in the supplementary material. Further inquiries can be directed to the corresponding author.
References
Steuer, C. E., El-Deiry, M., Parks, J. R., Higgins, K. A. & Saba, N. F. An update on larynx cancer. CA Cancer J. Clin. 67, 31–50. https://doi.org/10.3322/caac.21386 (2017).
Hoffmann, T. K. Total laryngectomy-still cutting-edge?. Cancers. https://doi.org/10.3390/cancers13061405 (2021).
Argiris, A. & Lefebvre, J. L. Laryngeal preservation strategies in locally advanced laryngeal and hypopharyngeal cancers. Front. Oncol. 9, 419. https://doi.org/10.3389/fonc.2019.00419 (2019).
Cortina, L. E. et al. Predictors of multiple dilations and functional outcomes after total laryngectomy and laryngopharyngectomy. Head Neck 46, 138–144. https://doi.org/10.1002/hed.27545 (2024).
Machtay, M. et al. Hypopharyngeal dose is associated with severe late toxicity in locally advanced head-and-neck cancer: An RTOG analysis. Int. J. Radiat. Oncol. Biol. Phys. 84, 983–989. https://doi.org/10.1016/j.ijrobp.2012.03.005 (2012).
Cmelak, A. et al. ECOG-ACRIN 2399: Analysis of patient related outcomes after Chemoradiation for locally advanced head and neck Cancer. Cancers Head Neck 5, 12. https://doi.org/10.1186/s41199-020-00059-1 (2020).
Forastiere, A. A. et al. Long-term results of RTOG 91–11: A comparison of three nonsurgical treatment strategies to preserve the larynx in patients with locally advanced larynx cancer. J. Clin. Oncol. 31, 845–852. https://doi.org/10.1200/JCO.2012.43.6097 (2013).
Nutting, C. M. et al. Dose-escalated intensity-modulated radiotherapy in patients with locally advanced laryngeal and hypopharyngeal cancers: ART DECO, a phase III randomised controlled trial. Eur. J. Cancer 153, 242–256. https://doi.org/10.1016/j.ejca.2021.05.021 (2021).
Laccourreye, O. et al. Primary total laryngectomy for endolaryngeal cT3-4M0 squamous cell carcinoma: A STROBE analysis. Laryngoscope. https://doi.org/10.1002/lary.31129 (2023).
Booth, C. M. & Tannock, I. F. Randomised controlled trials and population-based observational research: Partners in the evolution of medical evidence. Br. J. Cancer 110, 551–555. https://doi.org/10.1038/bjc.2013.725 (2014).
Ruiter, L. N. et al. Association of histological features with laryngeal squamous cell carcinoma recurrences: A population-based study of 1502 patients in the Netherlands. BMC Cancer 22, 444. https://doi.org/10.1186/s12885-022-09533-0 (2022).
Petersen, J. F. et al. Trends in treatment, incidence and survival of hypopharynx cancer: A 20-year population-based study in the Netherlands. Eur. Arch. Oto-rhino-laryngol. 275, 181–189. https://doi.org/10.1007/s00405-017-4766-6 (2018).
Jakobsen, K. K. et al. Increasing incidence and survival of head and neck cancers in Denmark: A nation-wide study from 1980 to 2014. Acta Oncol. 57, 1143–1151. https://doi.org/10.1080/0284186X.2018.1438657 (2018).
Dittberner, A. et al. Gender disparities in epidemiology, treatment, and outcome for head and neck cancer in Germany: A population-based long-term analysis from 1996 to 2016 of the Thuringian Cancer Registry. Cancers. https://doi.org/10.3390/cancers12113418 (2020).
Kouka, M. et al. Early mortality among patients with head and neck cancer diagnosed in Thuringia, Germany, between 1996 and 2016—A population-based study. Cancers. https://doi.org/10.3390/cancers14133099 (2022).
Goellnitz, I. et al. Role of comorbidity on outcome of head and neck cancer: A population-based study in Thuringia, Germany. Cancer Med. 5, 3260–3271. https://doi.org/10.1002/cam4.882 (2016).
Morkramer, L. et al. Systemic therapy for recurrent and/or metastatic head and neck cancer: A population-based healthcare research study in Thuringia, Germany. J. Cancer Res. Clin. Oncol. https://doi.org/10.1007/s00432-021-03535-4 (2021).
O’Neill, C. B. et al. Treatment complications and survival in advanced laryngeal cancer: A population-based analysis. Laryngoscope 124, 2707–2713. https://doi.org/10.1002/lary.24658 (2014).
Ritter, A. R. et al. Factors associated with total laryngectomy utilization in patients with cT4a laryngeal cancer. Cancers. https://doi.org/10.3390/cancers15225447 (2023).
Hochfelder, C. G. et al. Treatment sequence and survival in locoregionally advanced hypopharyngeal cancer: A surveillance, epidemiology, and end results-based study. Laryngoscope 130, 2611–2621. https://doi.org/10.1002/lary.28452 (2020).
Timmermans, A. J. et al. Trends in treatment and survival for advanced laryngeal cancer: A 20-year population-based study in The Netherlands. Head neck 38(Suppl 1), E1247-1255. https://doi.org/10.1002/hed.24200 (2016).
Song, Z., Chen, Z., Sun, X. & Zou, L. Competing risk models versus traditional Cox models for prognostic factors’ prediction and care recommendation in patients with advanced laryngeal squamous carcinoma: A population-based study. Eur. Arch. Oto-rhino-laryngol. 280, 3745–3756. https://doi.org/10.1007/s00405-023-07983-1 (2023).
Blomkvist, R. et al. Treatment and outcome among patients with laryngeal squamous cell carcinoma in Stockholm—A population-based study. Laryngosc. Investig. Otolaryngol. 8, 441–449. https://doi.org/10.1002/lio2.1034 (2023).
Zhu, X. et al. Survival prediction and treatment strategies for patients with advanced laryngeal carcinoma: A population-based study. Int. J. Clin. Oncol. 25, 1483–1491. https://doi.org/10.1007/s10147-020-01688-9 (2020).
Gourin, C. G. et al. Impact of surgeon and hospital volume on short-term outcomes and cost of laryngeal cancer surgical care. Laryngoscope 121, 85–90. https://doi.org/10.1002/lary.21348 (2011).
Helman, S. N. et al. Factors associated with complications in total laryngectomy without microvascular reconstruction. Head Neck 40, 2409–2415. https://doi.org/10.1002/hed.25363 (2018).
Sullivan, P. A. & Hartig, G. K. Dysphagia after total laryngectomy. Curr. Opin. Otolaryngol. Head Neck Surg. 9, 139–146 (2001).
Zenga, J., Goldsmith, T., Bunting, G. & Deschler, D. G. State of the art: Rehabilitation of speech and swallowing after total laryngectomy. Oral Oncol. 86, 38–47. https://doi.org/10.1016/j.oraloncology.2018.08.023 (2018).
Lee, D. S. et al. Risk factors for functional outcomes in advanced laryngeal squamous cell carcinoma. Laryngoscope 133, 594–600. https://doi.org/10.1002/lary.30166 (2023).
van Sluis, K. E. et al. Objective and subjective voice outcomes after total laryngectomy: A systematic review. Eur. Arch. Oto-rhino-laryngol. 275, 11–26. https://doi.org/10.1007/s00405-017-4790-6 (2018).
Sethi, R. K. V. & Deschler, D. G. National trends in primary tracheoesophageal puncture after total laryngectomy. Laryngoscope 128, 2320–2325. https://doi.org/10.1002/lary.27066 (2018).
Lewin, J. S., Baumgart, L. M., Barrow, M. P. & Hutcheson, K. A. Device life of the tracheoesophageal voice prosthesis revisited. JAMA Otolaryngol. Head Neck Surg. 143, 65–71. https://doi.org/10.1001/jamaoto.2016.2771 (2017).
Hilgers, F. J. et al. Prospective clinical phase II study of two new indwelling voice prostheses (Provox Vega 22.5 and 20 Fr) and a novel anterograde insertion device (Provox Smart Inserter). Laryngoscope. 120, 1135–1143. https://doi.org/10.1002/lary.20925 (2010).
Brook, I. & Goodman, J. F. Tracheoesophageal voice prosthesis use and maintenance in laryngectomees. Int. Arch. Otorhinolaryngol. 24, e535–e538. https://doi.org/10.1055/s-0039-3402497 (2020).
Hancock, K. L., Lawson, N. R. & Ward, E. C. Device life of the Provox Vega voice prosthesis. Eur. Arch. Oto-rhino-laryngol. 270, 1447–1453. https://doi.org/10.1007/s00405-012-2154-9 (2013).
Heirman, A. N. et al. Does prophylactic replacement of voice prosthesis make sense? A study to predict prosthesis lifetime. Otolaryngol. Head Neck Surg. 168, 429–434. https://doi.org/10.1177/01945998221116815 (2023).
Op de Coul, B. M. et al. A decade of postlaryngectomy vocal rehabilitation in 318 patients: A single Institution’s experience with consistent application of provox indwelling voice prostheses. Arch. Otolaryngol. Head Neck Surg. 126, 1320–1328. https://doi.org/10.1001/archotol.126.11.1320 (2000).
Petersen, J. F. et al. Postlaryngectomy prosthetic voice rehabilitation outcomes in a consecutive cohort of 232 patients over a 13-year period. Head Neck 41, 623–631. https://doi.org/10.1002/hed.25364 (2019).
Helman, S. N. et al. Predictors of length of stay, reoperation, and readmission following total laryngectomy. Laryngoscope 127, 1339–1344. https://doi.org/10.1002/lary.26454 (2017).
Pires, R. C. et al. Analysis of durability and replacement of vocal prostheses in total laryngectomy patients at a head and neck surgery referral center. Eur. Arch. Oto-rhino-laryngol. https://doi.org/10.1007/s00405-023-08180-w (2023).
Sampieri, C. et al. Predicting the need for prophylactic gastrostomy in major head and neck cancer surgery: The PEG score. Head Neck 45, 3042–3052. https://doi.org/10.1002/hed.27532 (2023).
Acknowledgements
We would like to thank all departments and outpatient clinics that were involved in the treatment and data collection of the patients.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Author information
Authors and Affiliations
Contributions
OGL, AM: conceptualization. OGL: first draft preparation. MK and LB: Data acquisition. HK, DB, JB, AM, KH, JP, KP: Resources. HK, DB, JB, AM, KH, JP: data validation. MK, LB, and OGL: data analysis. OGL. TB and TE: supervision. All authors contributed to the article and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kouka, M., Beckmann, L., Bitter, T. et al. Oncological and functional outcome after laryngectomy for laryngeal and hypopharyngeal cancer: a population-based analysis in Germany from 2001 to 2020. Sci Rep 14, 7761 (2024). https://doi.org/10.1038/s41598-024-58423-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-58423-x
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.