Abstract
Long non-coding RNAs (lncRNAs) have important biological functions, but their involvement in ovarian cancer remains elusive. We analyzed high-throughput data to identify lncRNAs associated with ovarian cancer outcomes. Our search led to the discovery of lncRNA TOPORS Antisense RNA 1 (TOPORS-AS1). Patients with high TOPORS-AS1 expression had favorable overall survival compared to low expression. This association was replicated in our study and confirmed by meta-analysis. In vitro experiments demonstrated that overexpressing TOPORS-AS1 in ovarian cancer cells suppressed cell proliferation and inhibited aggressive cell behaviors, including migration, invasion, and colony formation. Analysis of tumor cell transcriptomes indicated TOPORS-AS1′s influence on the Wnt/β-catenin signaling. Additional experiments revealed that TOPORS-AS1 increased the phosphorylation of β-catenin and suppressed the expression of CTNNB1, disrupting the Wnt/β-catenin pathway. Our experiments further discovered that vitamin D receptor (VDR) upregulated TOPORS-AS1 expression and that inhibition of β-catenin by TOPORS-AS1 required a RNA binding protein, hnRNPA2B1 (heterogeneous nuclear ribonucleoprotein A2B1). Taken together, these findings suggest that TOPORS-AS1 may behave like a tumor suppressor in ovarian cancer through interrupting the Wnt/β-catenin signaling and that VDR upregulates the expression of TOPORS-AS1. Assessing TOPORS-AS1 expression in ovarian cancer may help predict disease prognosis and develop treatment strategy
Similar content being viewed by others
Introduction
Ovarian cancer is a lethal gynecological disease, and the 5-year survival rate is dismal, only 47%1. As most patients are diagnosed with an advanced disease, few effective therapies are available for ovarian cancer treatment. Currently, there are no reliable tests available to detect ovarian cancer early. Biomarkers for accurate prediction of disease outcome and treatment response are also limited. Studies of ovarian cancer mechanisms have largely focused on proteins and their coding genes. Since protein-coding genes account for only 2% of the human genome2, our understanding of ovarian cancer development and progression from the genome perspective remains inadequate. Lately, long non-coding RNAs (lncRNAs), which contain 200 nucleotides or more with no or limited protein-coding potential, have been recognized to have crucial roles to play in regulation of biological functions and cellular activities. Emerging evidence also suggests that lncRNAs play a role in cancer, including ovarian cancer3,4,5,6,7,8,9. Understanding the involvement of lncRNAs in ovarian cancer may provide new insights and offer novel opportunities for diagnosis, prognosis, and treatment. In search for lncRNAs which might be implicated in ovarian cancer, we analyzed ovarian cancer survival in association with many lncRNAs whose expression data are available online in public databases. Our search led us to find the expression of TOPORS Antisense RNA 1 (TOPORS-AS1) significantly associated with overall and progression-free survival of ovarian cancer.
Vitamin D receptor (VDR) is a nuclear receptor which binds to its ligand, 1, 25-dihydroxyvitamin D3, and acts as a transcription factor in regulating important cellular activities and functions, such as immunity, cell proliferation, differentiation and apoptosis10. VDR is known to have strong anti-tumor properties, including suppression of proliferation and inflammation, promotion of differentiation and apoptosis, as well as inhibition of angiogenesis and tumor cell invasion and metastasis11. VDR is expressed in the ovaries and is involved in the regulation of estrogen biosynthesis and aromatase activity12,13,14. Increasing VDR expression in ovarian cancer cells inhibits cell proliferation and tumor growth15. Evidence also suggests that VDR may interact with lncRNAs to exert its functions16. VDR was found to increase the expression of MEG3 (maternally expressed gene 3), a tumor suppressor in ovarian17 and colorectal cancers18. In this report, we discussed our discovery of lncRNA TOPORS-AS1 whose expression was regulated by VDR, associated with ovarian cancer survival, and able to inhibit the Wnt/β-catenin signaling.
Results
Association of TOPORS-AS1 expression with disease stage and patient survival
Data from our study (Table 1) showed that TOPORS-AS1 expression was higher in early (Stage I or II) than late stages (Stage III or IV) of ovarian cancer (p = 0.0046). TOPORS-AS1 expression was not different between serous and non-serous tumors, nor changed by tumor grade or patient age at surgery. High expression of TOPORS-AS1 was found to be associated with favorable overall survival, and the association remained significant after adjusting for patient age, disease stage, tumor grade and histology (Table 2). In comparison to those with low TOPORS-AS1, patients with high expression had over 40% reduction in risk of death (HR = 0.58, 95% CI 0.34–0.99, p = 0.046). Risks for disease progression, however, were not significantly different between patients with high and low TOPORS-AS1 expression regardless of their clinicopathological features.
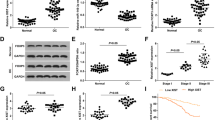
Using the Kaplan–Meier Plotter to analyze an online dataset of 1566 patients, we found that TOPORS-AS1 expression was associated with both overall and progression-free survivals. Patients with high TOPORS-AS1 had lower risk of death and disease progression compared to those with low expression (Fig. 1A,B), although our study showed a significant association only with overall survival, not progression-free survival (Fig. 1C,D).
Kaplan–Meier survival curves in ovarian cancer patients with high and low TOPORS-AS1 expression. (A) Kaplan–Meier overall survival curves by high (upper tertile) and low (lower tertile) expression of TOPORS-AS1 from online Kaplan–Meier Plotter. (B) Kaplan–Meier progression-free survival curves by high (upper tertile) and low (lower tertile) expression of TOPORS-AS1from online Kaplan–Meier Plotter. (C) Kaplan–Meier overall survival curves by high (upper tertile) and low (lower tertile) expression of TOPORS-AS1 from our study. (D) Kaplan–Meier progression-free survival curves by high (upper tertile) and low (lower tertile) expression of TOPORS-AS1 from our study. (E) Forest plots for associations between TOPORS-AS1 expression and overall survival. (F) Forest plots for associations between TOPORS-AS1 expression and progression-free survival.
Combining our study with 5 GEO (GSE14764, GSE30161, GSE18520, GSE26193, and GSE26712) and one TCGA datasets which contained information on survival outcomes and TOPORS-AS1 expression in ovarian cancer, we performed a meta-analysis on the association between ovarian cancer survival and TOPORS-AS1 expression in a total of 1,157 patients. The results showed that patients with high expression of TOPORS-AS1 had a significantly reduced risk of death, Hazard Ratio (HR) = 0.73, 95% CI 0.55–0.91 (Fig. 1E). Among the 7 datasets, 4 had information on progression-free survival, including GSE30161, GSE26193, TCGA and our study, and the meta-analysis showed no significant association between TOPORS-AS1 expression and progression-free survival, HR = 0.84, 95% CI 0.58–1.21 (Fig. 1F).
TOPORS-AS1 expression in ovarian cancer cells
TOPORS-AS1 expression was measured in 6 ovarian cancer cell lines (IGROV1, SKOV3, OVCAR3, OVCAR4, OVCAR5, OVCAR8) with RT-qPCR, and none of the tested cell lines showed high expression of this lncRNA (Fig. 2A). To assess its effect on ovarian cancer cells, we constructed a TOPORS-AS1 plasmid and transfected two cell lines (IGROV1, SKOV3) to make stable overexpression of the lncRNA. Compared to the mock cells, TOPORS-AS1 expression was about 60-fold higher in the transfected IGROV1 and 150-fold higher in transfected SKOV3 (Fig. 2B,C). We evaluated cell proliferation, migration, invasion, and colony formation in these transfected cells. The experiments showed that TOPORS-AS1 overexpression led to reduced cell proliferation, migration, invasion, and colony formation (Fig. 2D–K). These results were consistent in both cell lines (Supplemental Figure S1A,B).
Inhibition of ovarian cancer cell proliferation, invasion, migration, and colony formation by TOPORS-AS1. (A) RT-qPCR results showing TOPORS-AS1 expression in 6 ovarian cancer cell lines. (B,C) RT-qPCR results showing increased TOPORS-AS1 expression in IGROV1 and SKOV3 after TOPORS-AS1 transfection. (D,E) Cell proliferation assay showing slower proliferation of IGROV1 and SKOV3 after TOPORS-AS1 transfection. (F,G) Cell transwell migration assay showing reduced cell migration in IGROV1 and SKOV3 after TOPORS-AS1 transfection. (H,I) Cell transwell invasion assay showing reduced cell invasion in IGROV1 and SKOV3 after TOPORS-AS1 transfection. (J,K) Colony formation assay showing fewer colonies formed in IGROV1 and SKOV3 after TOPORS-AS1 transfection. (L,M) RT-qPCR results showing reduced TOPORS-AS1 expression after si-TOPORS-AS1 knockdown in IGROV1 and SKOV3 transfected with the TOPORS-AS1 plasmid. (N,O) Cell proliferation assay showing increased cell proliferation after si-TOPORS-AS1 knockdown in IGROV1 and SKOV3 transfected with the TOPORS-AS1 plasmid. (P,Q) Cell transwell migration assay showing increased cell migration after si-TOPORS-AS1 knockdown in IGROV1 and SKOV3 transfected with the TOPORS-AS1 plasmid. (R,S) Cell transwell invasion assay showing increased cell invasion after si-TOPORS-AS1 knockdown in IGROV1 and SKOV3 transfected with the TOPORS-AS1 plasmid. ***P < 0.0001; **P < 0.001.
To confirm the above effects from TOPORS-AS1, we treated the TOPORS-AS1 overexpressing cells (SKOV3, IGROV1) with siRNAs targeting the lncRNA. After the treatment, TOPORS-AS1 expression was significantly decreased in the cells (Fig. 2L,M). Compared to the control cells (treated with scrambled siRNAs), cells with TOPORS-AS1 knockdown had increased proliferation, migration, and invasion (Fig. 2N,S), and the results were consistent in both cell lines (Supplemental Figure S1C).
Interaction between TOPORS-AS1 and the Wnt/β-catenin pathway
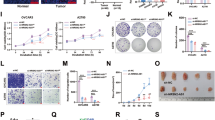
To investigate the effect of TOPORS-AS1 in ovarian cancer, we compared the transcriptomes of SKOV3 and IGROV1 with and without TOPORS-AS1 overexpression using the Human Transcriptome Array 2.0 (Affymetrix). Figure 3A,B are heatmaps of the differentially expressed genes (DEGs) related to TOPORS-AS1 overexpression in IGROV1 and SKOV3, respectively. There were 51 up-regulated and 6 down-regulated genes (fold change ≥ 1.5 and p < 0.05) which were shared by both cell lines. This list of DEGs was uploaded to IPA for interrogation of the biological networks involving TOPORS-AS1 overexpression. As shown in Fig. 3C, the involvement of Wnt/β-catenin was suggested in both cell lines overexpressing TOPORS-AS1.
Transcriptomic analysis of ovarian cancer cells associated with TOPORS-AS1 expression and discovery of β-catenin involvement. (A,B) Heatmap of differentially expressed genes due to TOPORS-AS1 overexpression in IGROV1 and SKOV3. (C) IPA results of common pathways associated with TOPORS-AS1 overexpression in both IGROV1 and SKOV3. (D) Western blot results showing reduced β-catenin, LEF1, TCF1/TCF7 and c-Myc, increased phosphorylation of β-catenin, and no changes in GSK3β (either total or phosphorylated) in both IGROV1 and SKOV3 due to TOPORS-AS1 overexpression. (E,G) Western blot results showing the pulldown of hnRNPA2B1 in mock and TOPORS-AS1 transfected IGROV1 and SKOV3. (F,H) RIP results showing marked enrichment of TOPORS-AS1 in cell extracts processed with anti-hnRNPA2B1 antibody pulldown, but not in IgG pulldown, from IGROV1 and SKOV3. (I,J) Co-immunoprecipitation (IP) results suggesting β-catenin binding to hnRNPA2B1 in IGROV1 and SKOV3. (K) Western blot results showing that β-catenin suppression by TOPORS-AS1 could be reversed by knockdown hnRNPA2B1 through si-hnRNPA2B1 in IGROV1 and SKOV3. ***P < 0.0001; **P < 0.001.
To determine if the Wnt/β-catenin signaling was involved, we analyzed several major components of the pathway in the transfected cells. Figure 3D shows the results of our western blot analysis in which we found increased phosphorylation of β-catenin and decreased levels of β-catenin, LEF1, TCF1/TCF7, and c-Myc in TOPORS-AS1 overexpressing cells. GSK3β, both total and phosphorylated form (Y216), showed no changes. As phosphorylation of β-catenin leads to its degradation which results in suppression of the Wnt/β-catenin signaling, our results support the IPA prediction that TOPORS-AS1 may interrupt the Wnt/β-catenin pathway in ovarian cancer.
To further assess the lncRNA’s effect on β-catenin, we analyzed the β-catenin gene (CTNNB1) expression in cells with and without TOPORS-AS1 overexpression. Our qPCR results showed that levels of β-catenin mRNA were significantly reduced in IGROV1 and SKOV3 overexpressing TOPORS-AS1, compared with the mock cells (Supplemental Figure S2), suggesting that TOPORS-AS1 overexpression may suppress both β-catenin transcription and phosphorylation.
RIP assay was performed to determine if TOPORS-AS1 interacts with β-catenin in IGROV1 and SKOV3. The analysis showed no direct interaction between these molecules (data not shown), suggesting that the effect of TOPORS-AS1 on β-catenin may involve or act through other molecules.
Interaction between TOPORS-AS1 and hnRNPA2B1
In search for molecules which may interact with TOPORS-AS1, we used an online program NPInter19 that makes in silico prediction between RNAs and proteins. The software predicted several candidate molecules which may contain putative regions interactive with TOPORS-AS1. Among them, hnRNPA2B1 (heterogeneous nuclear ribonucleoproteins A2/B1) was shown to have a binding site in the exon region of TOPORS-AS1. This hypothetic interaction was supported by our RIP assay in which hnRNPA2B1 was shown to interact with TOPORS-AS1 both in IGROV1 and SKOV3 (Fig. 3E–H). Using co-immunoprecipitation, we also found that hnRNPA2B1 could interact with β-catenin, and the interaction was observed in both cell lines (Fig. 3I,J). As mentioned earlier, TOPORS-AS1 overexpression could reduce the level of β-catenin. When we suppressed the expression of hnRNPA2B1 with siRNA, β-catenin was increased in the cells overexpressing TOPORS-AS1 (Fig. 3K), suggesting that hnRNPA2B1 may be required in the inhibition of β-catenin by TOPORS-AS1.
Interaction between TOPORS-AS1 and VDR
PROMO predicted that vitamin D receptor (VDR) was a transcription factor of TOPORS-AS1, and a VDR binding site in the lncRNA’s promoter, from − 1470 to − 1478, was suggested (Fig. 4A). To validate the VDR binding site, we made a VDR vector, pCMV_VDR, which was transfected to 293 T cells. The transfection increased VDR expression in the cells (Fig. 4B), and the increase in VDR expression led to elevated luciferase activities in the cells co-transfected with a wild-type TOPORS-AS1 promoter, but not in those with a mutant promoter (Fig. 4C). The binding between VDR and the TOPORS-AS1 promoter was confirmed by chromatin immunoprecipitation (Fig. 4D).
Upregulation of TOPORS-AS1 expression by VDR and its effect on tumor cell proliferation, migration, and invasion. (A) PROMO prediction of a VDR binding site in the TOPORS-AS1 promoter. (B) Western blot results showing increased VDR expression in 293 T after transfecting the cells with a pCMV_VDR plasmid. (C) Luciferase reporter assay results showing increased luciferase activity in 293 T cells co-transfected with the VDR plasmid (pCMV_VDR) and luciferase reporter gene linked to TOPORS-AS1-wt (wild type) and no increase in TOPORS-AS1-mut (mutated). (D) Chromatin immunoprecipitation (ChIP) results suggesting VDR binding to the TOPORS-AS1 promoter. (E) Western blot results showing increased VDR and decreased β-catenin in IGROV1 and SKOV3 after VDR transfection. (F,G) RT-qPCR results showing increased TOPORS-AS1 expression in IGROV1 and SKOV3 after VDR transfection. (H,I) Cell proliferation assay showing slower IGROV1 and SKOV3 proliferation after VDR transfection. (J,K) Cell transwell migration assay showing fewer migrated IGROV1 and SKOV3 after VDR transfection. (L,M) Cell transwell invasion assay showing fewer invaded IGROV1and SKOV3 after VDR transfection. ***P < 0.0001; **P < 0.001.
After confirming the interaction between VDR and TOPORS-AS1 in 293 T cell, we evaluated the VDR effects on tumor cells by transfecting IGROV1 and SKOV3 with the VDR plasmid. Our experiments showed that the VDR-transfected tumor cells had reduced β-catenin (Fig. 4E) and increased TOPORS-AS1 expression (Fig. 4F,G). VDR transfection also led to decreased cell proliferation (Fig. 4H,I), migration (Fig. 4J,K), and invasion (Fig. 4L,M). The results were consistent in both cell lines (Supplemental Figure S3). To examine if the VDR effect on β-catenin was mediated through TOPORS-AS1, we transfected IGROV1 and SKOV3 first with VDR (Fig. 5A,B) and then TOPORS-AS1 siRNAs (Fig. 5C,D). After siRNA knockdown, we observed no β-catenin suppression (Fig. 5E,F), and cell proliferation was increased (Fig. 5G,H), suggesting that TOPORS-AS1may mediate the VDR effect on tumor cells.
TOPORS-AS1 mediates the effect of VDR on ovarian cancer cells. (A,B) Western blot results showing reduced β-catenin in IGROV1 and SKOV3 after VDR transfection. (C,D) RT-qPCR results showing TOPORS-AS1 knockdown by si-TOPORS-AS1 in VDR-transfected IGROV1 and SKOV3. (E,F) Western blot results showing increased β-catenin after TOPORS-AS1 knockdown by si-TOPORS-AS1 in VDR-transfected IGROV1 and SKOV3. (G,H) Increased cell proliferation in VDR-transfected IGROV1 and SKOV3 after TOPORS-AS1 knockdown by si-TOPORS-AS1. (I,K) Decreased cell proliferation in IGROV1 and SKOV3 after transfection with TOPORS-AS1. (J,L) Western blot results showing no change in VDR levels over time in IGROV1 and SKOV3 after transfection with TOPORS-AS1. ***P < 0.0001; **P < 0.001.
We also evaluated if TOPORS-AS1 overexpression in ovarian tumor cells could increase the expression of VDR which subsequently suppressed tumor proliferation. Our experiments showed that VDR expression did not change over time in IGROV1 and SKOV3 after TOPORS-AS1 transfection (Fig. 5J,L), but cell proliferation was suppressed in the transfected cells (Fig. 5I,K), indicating that TOPORS-AS1 does not affect the expression of VDR and the lncRNA suppresses cell proliferation, not VDR.
Discussion
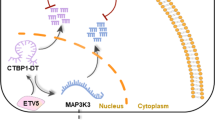
In this study, we found that ovarian cancer patients with early-stage disease tended to have high TOPORS-AS1 in the tumor and that high expression was associated with favorable overall survival compared to low expression. This survival association was not affected by disease stage and tumor histology and replicated in several independent datasets. A meta-analysis of 1157 patients confirmed the association between TOPORS-AS1 and overall survival. Results of our in vitro experiments supported the finding in patients, demonstrating that TOPORS-AS1 expression in ovarian cancer cells inhibited tumor cell proliferation, migration, invasion, and colony formation. Further investigation of biological function and molecular regulation revealed that TOPORS-AS1 interacted with hnRNPA2B1 and increased the phosphorylation of β-catenin which led to its degradation, resulting in suppression of the Wnt/β-catenin signaling pathway. Our experiments also discovered that VDR was a transcription factor for TOPORS-AS1 and that the suppression of ovarian cancer cells by VDR could be mediated through TOPORS-AS1. The relationships among TOPORS-AS1, VDR, β-catenin, and hnRNPA2B1 are depicted in Fig. 6.
TOPORS-AS1 is encoded by a gene in chromosome 9p21, and the gene has a 554 bp transcript (NR_033992.2). The function and regulation of this transcript was not known before. We found in the study that TOPORS-AS1 expression was different by disease stage, but not by tumor histology or grade, suggesting that the lncRNA’s expression may be affected by the size or time of tumor growth, but not by the type or differentiation of tumor cells. Our finding of no difference in expression between serous and non-serous tumors also indicates that the biomarker may have broader clinical implications in ovarian cancer, not only to those high-grade serous tumors. Our study showed that TOPORS-AS1′s association with survival was only observed consistently in overall survival, but not in progression-free survival. It is unclear what the reasons for this discrepancy are, but we acknowledge the fact that progression-free survival of ovarian cancer is difficult to assess consistently across studies compared to overall survival given its high mortality.
The finding of high TOPORS-AS1 in association with favorable ovarian cancer survival is in agreement with the observations of two previous studies by Su et al.20 and Sorenson et al.21 in breast cancer. In addition, our finding of VDR in regulation of TOPORS-AS1 expression is also in line with the understanding of VDR’s role in cancer22. VDR belongs to the superfamily of steroid hormone receptors23, and the protein is expressed in many organs including the ovaries where it regulates important cellular activities and biological functions15,24. VDR is known to be a strong tumor suppressor and is able to inhibit ovarian cancer cell growth15. We found in the study that VDR was able to bind to the TOPORS-AS1 promoter, upregulating its expression. Our experiments also indicated that TOPORS-AS1 could mediate the inhibitory effects of VDR on cell proliferation, migration, and invasion.
Our study further suggests that TOPORS-AS1 may play a role in connection to the reciprocal suppression between VDR and the Wnt/β-catenin pathway in ovarian cancer25. It is known that VDR deficiency leads to increased Wnt/β-catenin signaling in colon cancer26. When the pathway is activated, β-catenin is translocated from cytoplasm to nucleus where it binds to LEF/TCF to increase the expression of Myc and Cyclin D1, promoting cell migration and tumor progression. When deactivated, β-catenin is released from the APC/Axin/GSK3β complex in cytoplasm, undergoing phosphorylation and degradation27. In various human malignancies including ovarian cancer, the Wnt/β-catenin signaling is highly activated28,29. Defects in APC or β-catenin were found in colorectal cancer, which stabilized β-catenin, resulting in constitutive activation of the Wnt/β-catenin pathway30,31,32. Evidence suggests that the Wnt/β-catenin pathway is also important in ovarian cancer28,33,34,35.
A number of lncRNAs have been identified to influence or regulate the Wnt/β-catenin pathway36,37,38,39,40,41. In our experiments, we found increased TOPORS-AS1 expression in ovarian cancer cells after VDR transfection, and these increases in expression of TOPORS-AS1 and VDR resulted in decreased β-catenin expression and increased phosphorylation of β-catenin, leading to suppression of the Wnt/β-catenin pathway. To evaluate which one, VDR or TOPORS-AS1, was responsible for the change in β-catenin, we added si-TOPORS-AS1 in cell culture and found that the decline in β-catenin was abolished after TOPORS-AS1 knockdown, suggesting that the lncRNA may mediate the effect of VDR on β-catenin. Our experiments further indicated no direct interaction between TOPORS-AS1 and β-catenin, and the effect of TOPORS-AS1 on β-catenin involved an RNA-binding protein. We found that hnRNPA2B1 was able to interact with both TOPORS-AS1 and β-catenin. This protein is a member of the heterogeneous nuclear ribonucleoprotein family which functions as a splicing factor to process and stabilize pre-mRNAs. MYU (c-Myc-upregulated lncRNA) associates with the RNA binding protein hnRNP-K to stabilize CDK6 expression, and the lncRNA is a direct target of the Wnt/c-Myc pathway5. It was found that hnRNPA2 could interact with the 3′-UTR of CTNNB1, affecting the expression of CTNNB1 and β-catenin42. In our study, β-catenin levels were decreased by TOPORS-AS1 through its interaction with hnRNPA2B1. Without the RNA-binding protein, the effect of TOPORS-AS1 on β-catenin was not observed, suggesting that both TOPORS-AS1 and hnRNPA2B1 are required in their action on β-catenin.
In summary, our study showed that high expression of lncRNA TOPORS-AS1 was associated with favorable prognosis of ovarian cancer. In vitro experiments demonstrated that TOPORS-AS1 behaved like a tumor suppressor in ovarian cancer as the lncRNA could suppress cell proliferation, migration, invasion, and colony formation. Furthermore, TOPORS-AS1 increased β-catenin degradation and inhibited the expression of CTNNB1, suppressing the Wnt/β-catenin signaling. Our experiments also found that VDR was able to bind to the TOPORS-AS1 promoter, upregulating its expression, and that TOPORS-AS1 could mediate the inhibitory effects of VDR on cell proliferation, migration, and invasion. The effect of TOPORS-AS1 on β-catenin in ovarian cancer relied on the presence of hnRNPA2B1. Taken together, these findings suggest that TOPORS-AS1 is a tumor suppressor in ovarian cancer and that assessing the lncRNA level in tumor tissue may help to predict the disease prognosis and design treatment strategy.
Materials and methods
Study patients
To study epithelial ovarian cancer, we recruited 266 patients who were operated in two hospitals affiliated with the University of Turin in Italy. The recruitment was approved by the AOU Citta della Salute’s ethic review committee and the Mauriziano hospital’s eithic review committee, and informed consents were obtained from the patients. All the study protocols were performed in accordance with relevant guidelines and regulations. Disease stage was determined based on the International Federation of Gynecology and Obstetrics (FIGO) Classification, and tumor histology and grade were characterized according to the WHO Guidelines43,44. Detailed description of the study patients is provided in Supplemental Materials and Methods S1.
Tumor samples and RNA extraction
Fresh tumor samples were collected from the patients during surgery, and the samples were evaluated independently by two pathologists to confirm tumor cell contents (> 80% tumor cells). The tissue specimens were pulverized using a tissue homogenizer. Approximately 30 mg of pulverized tissue powder was used for total RNA extraction, using the AllPrep DNA/RNA Mini kit (Qiagen). After extraction, total RNAs were treated with RNase-free DNase to remove DNA contamination. The purity and quantity of the RNA samples were evaluated with specific light absorbances and RNA Integrity Number (RIN). The same methods were also used for RNA extraction and quantification from cell lines.
RT-qPCR
A high capacity cDNA reverse transcription kit was used to convert total RNA to cDNA (Applied Biosystems). The cDNA samples were analyzed for expression of TOPORS-AS1, CTNNB1 and GAPDH, using the SYBR Green-based quantitative PCR. PCR primer sequences are shown in Supplemental Table S1. Details of the analysis are provided in Supplemental Materials and Methods S1.
Cell culture
The NCI-60 DTP Human Tumor Cell Screening Panel was purchased from NIH for research by the High-throughput Drug Screening Core at University of Hawaii Cancer Center (UHCC). From the panel we selected six ovarian cancer cell lines for testing, including OVCAR3, OVCAR4, OVCAR5, OVCAR8, IGROV1, and SKOV3. The 293 T cell line was purchased from the American Type Culture Collection (ATCC). All the cells were cultured in RPMI 1640 medium containing 10% FBS and 100 units/ml of penicillin/streptomycin (Pen/Strep) at 37 °C under a humidified atmosphere containing 5% CO2.
Plasmid construction and amplification
Based on the transcript NR_033992.2 (554 bp), a TOPORS-AS1 template was synthesized by GENEWIZ and inserted into a lentiviral vector, pCDH-EF1-MCS-pA-PGK-copGFP-T2A-Puro (System Biosciences), with two restriction sites, Nhe1 and BamH1. The plasmid contains two promoters, EF1 which controls the expression of TOPORS-AS1 and PGK which drives a report gene, GFP, serving as a transfection control. The expression of TOPORS-AS1 from the plasmid was verified by direct sequencing and gel electrophoresis after restriction enzyme digestion. For plasmid amplification, please see details in Supplemental Materials and Methods S1.
Plasmid transfection and stable cell selection
Ovarian cancer cell lines, IGROV1 and SKOV3, were selected for transfection either with pCDH_TOPORS-AS1 or pCDH_vector (mock), using the Lipofectamine 3000 reagent (Thermo Fisher Scientific). Details of plasmid transfection and cell selection are provided in Supplemental Materials and Methods S1.
Cell proliferation assay
Ovarian cancer cells in 100 μl complete medium were added into the wells of 96-well plates at a density of 3 × 103 cells per well. After 0, 24, 48, and 72 h of incubation, 10 μl cell proliferation reagent WST-1 was added into each well following the manufacturer’s instructions (Roche). The cells were incubated with WST-1 for 2 more hours at 37 °C in a humidified atmosphere containing 5% CO2 before the plate was read with a microplate reader (BioTek Synergy 2, Winooski, VT) at 450 nm absorbance. Cell proliferation at each time point was normalized to the mean viable cells at 0 h. Each proliferation assay was performed in triplicate.
Cell migration and invasion assays
Cell migration and invasion were analyzed in 24-well plates using the Costar Transwell permeable membrane support with 8.0-μm pore size (Corning). In the invasion assay, ovarian cancer cells, 1 × 104 cells per well, in 200 μl serum-free medium were placed in the upper chambers coated with growth-factor-reduced Matrigel at 1 mg/ml (BD Pharmingen). The lower chambers were filled with 600 μl complete culture media containing 10% FBS. Cell migration assays were performed similarly without Matrigel coating. Invaded or migrated cells were stained with HEME 3 Solution (Fisher Diagnostics) after 36 h of culture, and the stained cells were photographed using the Olympus CKX41 microscopy with an Infinity 2 camera. Cell numbers were counted using the ImageJ software. All the experiments were repeated 3 times, and each experiment was done in triplicate.
Colony formation assay
The assay has been described elsewhere8. In brief, ovarian cancer cells (5 × 103) were seeded on 0.3% agarose overlaid onto solidified 0.6% agarose in PRMI 1640 with 10% FBS. Complete medium (200 µl) was added into the well every three days. Cell colonies were counted after three weeks. The experiments were repeated 3 times.
siRNA assay
Ovarian cancer cells with stable expression of TOPORS-AS1 were transfected with the Target Long Noncoding RNA siRNA-SMART pool (#R-189076-00-0005) to knockdown TOPORS-AS1 expression, and Lincode Non-targeting Pool (#D-001320-10-05) was used as a negative control, both of which were purchased from Dharmacon. The transfection was performed following the manufacturer’s protocol for Lipofectamine RNAiMAX (Themo Fisher Scientific). The transfected cells were prepared for analysis after 36 h of incubation.
Analysis of cell transcriptome
The transcriptomes of IGROV1 and SKOV3 with and without TOPORS-AS1 overexpression were analyzed with the Affymetrix Human Transcriptome Array 2.0 (Affymetrix). DNA labeling, probe hybridization, and signal scanning were completed by the Genomics Shared Resource at UHCC. The initial array data were normalized using the robust multi-array average RMA algorithm in the Affymetrix Expression Console (Affymetrix). Gene-based differential expression were analyzed using the Transcriptome Analysis Console (TAC) v3.0 (Affymetrix). Differentially expressed genes, defined as fold change ≥ 1.5 and p value < 0.05, were interrogated with the Ingenuity Pathway Analysis (IPA) software to predict the possible signaling pathways and regulatory mechanisms associated with TOPORS-AS1 overexpression. PROMO (http://alggen.lsi.upc.es/cgi-bin/promo_v3/promo/promoinit.cgi?dirDB=TF_8.3) was employed with 5% dissimilarity to predict the potential transcription factors and their possible binding sites in the TOPORS-AS1 promoter45,46. NPInter (https://www.bioinfo.org/NPInter/) was used to show possible interactions between TOPORS-AS1 and other molecules19.
RNA immunoprecipitation (RIP)
EZ-Magna RIP RNA-Binding Protein Immunoprecipitation kit (#17-701, Millipore) was used for RIP assay. Antibody against β-catenin (ab6302) was purchased from Abcam. Rabbit IgG antibody (#12-370 from EMD Millipore) was used as control. RIP assay was performed according to the manufacturer's instructions.
Chromatin immunoprecipitation (ChIP)
ChIP assay was carried out using a commercial kit from EMD Millipore. Plasmids (pCMV-vector or pCMV-VDR) were transfected to 293 T cells using the Lipofectamine 3000 reagent (Themo Fisher Scientific). Details of the ChIP assay are described in Supplemental Materials and Methods S1.
Dual luciferase reporter assay
A plasmid (pCMV-VDR) containing a full-length human VDR and a vector (pGL4.27[luc2P/minP/Hygro]) were purchased from Origene Technologies and Promega, respectively. GENEWIZ synthesized wild and mutant TOPORS-AS1 promoters and inserted the sequences into the pGL4.27 vector to make pGL4.27-TOPORS-AS1-wt and pGL4.27-TOPORS-AS1-mut plasmids. pCMV-VDR or pCMV-vector were transiently transfected to the 293 T cells using Lipofectamine 3000 reagent (Themo Fisher Scientific). After transfection, cells were incubated for 36 h and further transfected with plasmid pGL4.27-TOPORS-AS1-wt or pGL4.27-TOPORS-AS1-mut, together with the renilla reporter vector. After incubation, renilla and firefly luciferase activities were measured using the Dual-Luciferase kit (Promega). The results were normalized to the renilla reporter to adjust for transfection efficiency. Each assay was performed in triplicate, and the assays were repeated three times.
Immunoblotting
Cell proteins were extracted using the RIPA Lysis and Extraction Buffer (Themo Fisher Scientific). Concentrations of the extracted proteins were measured with the BCA assay, and 40–60 µg of the proteins were boiled in the Laemmli Sample Buffer (Bio-Rad) at 95℃ for 5 min, followed by 10% SDS–PAGE. The gel results were transferred onto the polyvinylidene difluoride (PVDF) membranes (Millipore) which were then blocked with 5% non-fat milk for 30 min and incubated with a primary antibody, followed by a secondary antibody to generate the signals detectable by an enhanced chemiluminescence system (Pierce). Primary antibodies used for blotting included anti-VDR (#12550 from Cell Signaling Technology), anti-hnRNPA2B1 (ab6102 from Abcam), anti-β-catenin (ab6302 from Abcam), anti-p-β-catenin (ser33/37/thr41) (#9561 from Cell Signaling Technology), anti-β-catenin (#8480 from Cell Signaling Technology), anti-LEF1 (#2230 from Cell Signaling Technology), anti-TCF1/TCF7(#2203 from Cell Signaling Technology), anti-cMYC (#18583 from Cell Signaling Technology), anti-p-GSK3β(Y216) (ab75745 from Abcam), anti-GSK3β (#12456 from Cell Signaling Technology, and anti-β-actin (A2228 from Sigma-Aldrich) antibodies.
Immunoprecipitation (IP)
Cells were cultured to 70% confluence and lysed in the Pierce IP Buffer (Themo Fisher Scientific). To purify proteins, cell lysates (600–800 μg) were incubated with 30 μl of the Pierce Protein A/G Magnetic Beads (Themo Fisher Scientific) for 30 min at 4 °C on rotation. After bead removal and overnight incubation at 4 °C with anti-β-catenin antibody (2–4 μg/mg protein), the protein solution was mixed with 30 μl of the Protein A/G Magnetic Beads, and the mixture was incubated for another 2 h. Then, the beads were washed three times with washing buffer and once with PBS, and were further mixed with the Laemmli Sample Buffer, followed by boiling at 95 °C for 5 min. After that, the solution was processed with 10% SDS–PAGE, and the resulting gels were transferred to the PVDF membranes, followed by incubation with another antibody (anti-hnRNPA2B1) and ECL measurement (Bio-Rad).
Meta-analysis
Using the keywords “ovarian cancer” and “ovarian carcinoma”, we searched the Gene Expression Omnibus (GEO) in NCBI (https://www.ncbi.nlm.nih.gov/gds). Datasets with more than 50 tumor samples as well as information on TOPORS-AS1 expression and survival outcomes were selected for analysis. Five datasets meeting the criteria were identified in the database, including GSE14764, GSE30161, GSE18520, GSE26193, and GSE26712. The Cancer Genome Atlas (TCGA) data on TOPORS-AS1 expression in ovarian cancer were also downloaded from TANRIC (https://www.tanric.org), and clinical information related to the dataset was retrieved from cBioPortal (http://www.cbioportal.org)47,48. Hazard ratios (HR) and 95% confidence intervals (95% CI) were calculated for each of the datasets, and these results were entered into the Review Manager 5.3 to obtain summary HR and 95% CI, using the random-effect model49.
Statistical analysis
Expression index (EI) was calculated as levels of TOPORS-AS1 expression after adjusting for the GAPDH expression, and the calculation was based on the formula 1000 × 2(−∆Ct), where ∆Ct is the difference in cycle thresholds (Ct) between CtTOPORS-AS1 and CtGAPDH. For data analysis, the EI values were classified into low, middle, and high groups, based on the tertile distribution of TOPORS-AS1 expression. The expression data were analyzed for associations with clinical and pathological variables, using the chi-square test. Log-rank test was used to compare the Kaplan–Meier survival curves between high and low expression of TOPORS-AS1, and the median was used as cutoff. Hazard ratios (HR) and 95% confidence interval (CI) were calculated using the Cox proportional hazards regression model, univariate and multivariate. In the multivariate model, age at surgery, disease stage, tumor grade and histology type were included as covariates to control for confounding factors. Two survival outcomes, progression-free and overall survivals, were used in survival analysis. Progression-free survival was the time interval from the date of surgery to the date of disease progression or last follow-up. Overall survival was the duration between surgery and last follow-up or death. SAS (version 9.4) and R (version 3.0.2) were used for statistical analyses. All p-values were two-sided.
Abbreviations
- lncRNAs:
-
Long non-coding RNAs
- TOPORS-AS1:
-
TOPORS Antisense RNA
- VDR:
-
Vitamin D receptor
- MEG3:
-
Maternally expressed gene 3
- HR:
-
Hazard ratio
- CI:
-
Confidence interval
- OS:
-
Overall survival
- PFS:
-
Progression free survival
- DEGs:
-
Differentially expressed genes
- LEF1:
-
Lymphoid enhancer-binding factor 1
- TCF1/TCF7:
-
Transcription Factor 7
- GSK3β:
-
Glycogen synthase kinase 3 beta
- hnRNPA2B1:
-
Heterogeneous nuclear ribonucleoproteins A2/B1
- IP:
-
Immunoprecipitation
- RIP:
-
RNA immunoprecipitation chip
- ChIP:
-
Chromatin immunoprecipitation
References
ACS. American Cancer Society. Cancer Facts & Figures 2018. American Cancer Society 2018 (2018).
Harrow, J. et al. GENCODE: The reference human genome annotation for The ENCODE Project. Genome Res. 22, 1760–1774. https://doi.org/10.1101/gr.135350.111 (2012).
Gao, Y. et al. LncRNA-HOST2 regulates cell biological behaviors in epithelial ovarian cancer through a mechanism involving microRNA let-7b. Hum. Mol. Genet. 24, 841–852. https://doi.org/10.1093/hmg/ddu502 (2015).
Ji, Q. et al. Long non-coding RNA MALAT1 promotes tumour growth and metastasis in colorectal cancer through binding to SFPQ and releasing oncogene PTBP2 from SFPQ/PTBP2 complex. Br. J. Cancer 111, 736–748. https://doi.org/10.1038/bjc.2014.383 (2014).
Kawasaki, Y. et al. MYU, a Target lncRNA for Wnt/c-Myc signaling, mediates induction of CDK6 to promote cell cycle progression. Cell. Rep. 16, 2554–2564. https://doi.org/10.1016/j.celrep.2016.08.015 (2016).
Kim, J. et al. Long noncoding RNA MALAT1 suppresses breast cancer metastasis. Nat. Genet. 50, 1705–1715. https://doi.org/10.1038/s41588-018-0252-3 (2018).
Luo, M. et al. Long non-coding RNA H19 increases bladder cancer metastasis by associating with EZH2 and inhibiting E-cadherin expression. Cancer Lett. 333, 213–221. https://doi.org/10.1016/j.canlet.2013.01.033 (2013).
Wang, Z. et al. ERalpha upregulates the expression of long non-coding RNA LINC00472 which suppresses the phosphorylation of NF-kappaB in breast cancer. Breast Cancer Res. Treat. https://doi.org/10.1007/s10549-018-05108-5 (2019).
Fu, Y. et al. Long non-coding RNAs, ASAP1-IT1, FAM215A, and LINC00472, in epithelial ovarian cancer. Gynecol. Oncol. 143, 642–649. https://doi.org/10.1016/j.ygyno.2016.09.021 (2016).
Carlberg, C. & Campbell, M. J. Vitamin D receptor signaling mechanisms: Integrated actions of a well-defined transcription factor. Steroids 78, 127–136. https://doi.org/10.1016/j.steroids.2012.10.019 (2013).
Feldman, D., Krishnan, A. V., Swami, S., Giovannucci, E. & Feldman, B. J. The role of vitamin D in reducing cancer risk and progression. Nat. Rev. Cancer 14, 342–357. https://doi.org/10.1038/nrc3691 (2014).
Villena-Heinsen, C. et al. Immunohistochemical analysis of 1,25-dihydroxyvitamin-D3-receptors, estrogen and progesterone receptors and Ki-67 in ovarian carcinoma. Anticancer Res. 22, 2261–2267 (2002).
Irani, M. & Merhi, Z. Role of vitamin D in ovarian physiology and its implication in reproduction: A systematic review. Fertil. Steril. 102, 460–468. https://doi.org/10.1016/j.fertnstert.2014.04.046 (2014).
Nandi, A., Sinha, N., Ong, E., Sonmez, H. & Poretsky, L. Is there a role for vitamin D in human reproduction?. Horm. Mol. Biol. Clin. Investig. 25, 15–28. https://doi.org/10.1515/hmbci-2015-0051 (2016).
Ahonen, M. H., Zhuang, Y. H., Aine, R., Ylikomi, T. & Tuohimaa, P. Androgen receptor and vitamin D receptor in human ovarian cancer: growth stimulation and inhibition by ligands. Int. J. Cancer 86, 40–46. https://doi.org/10.1002/(sici)1097-0215(20000401)86:1%3c40::aid-ijc6%3e3.0.co;2-e (2000).
Jiang, Y. J. & Bikle, D. D. LncRNA profiling reveals new mechanism for VDR protection against skin cancer formation. J. Steroid. Biochem. Mol. Biol. 144(Pt A), 87–90. https://doi.org/10.1016/j.jsbmb.2013.11.018 (2014).
Xiu, Y. L. et al. Upregulation of the lncRNA Meg3 induces autophagy to inhibit tumorigenesis and progression of epithelial ovarian carcinoma by regulating activity of ATG3. Oncotarget 8, 31714–31725. https://doi.org/10.18632/oncotarget.15955 (2017).
Zuo, S., Wu, L., Wang, Y. & Yuan, X. Long non-coding RNA MEG3 activated by vitamin D suppresses glycolysis in colorectal cancer via promoting c-Myc degradation. Front. Oncol. 10, 274. https://doi.org/10.3389/fonc.2020.00274 (2020).
Hao, Y. et al. NPInter v3.0: an upgraded database of noncoding RNA-associated interactions. Database (Oxford). https://doi.org/10.1093/database/baw057 (2016).
Su, X. et al. Comprehensive analysis of long non-coding RNAs in human breast cancer clinical subtypes. Oncotarget 5, 9864–9876. https://doi.org/10.18632/oncotarget.2454 (2014).
Sorensen, K. P. et al. Long non-coding RNA expression profiles predict metastasis in lymph node-negative breast cancer independently of traditional prognostic markers. Breast Cancer Res. 17, 55. https://doi.org/10.1186/s13058-015-0557-4 (2015).
Deuster, E., Jeschke, U., Ye, Y., Mahner, S. & Czogalla, B. Vitamin D and VDR in gynecological cancers-a systematic review. Int. J. Mol. Sci. 18. https://doi.org/10.3390/ijms18112328 (2017).
Whitfield, G. K., Jurutka, P. W., Haussler, C. A. & Haussler, M. R. Steroid hormone receptors: evolution, ligands, and molecular basis of biologic function. J. Cell. Biochem. Suppl. 32–33, 110–122 (1999).
Silvagno, F. et al. Analysis of vitamin D receptor expression and clinical correlations in patients with ovarian cancer. Gynecol. Oncol. 119, 121–124. https://doi.org/10.1016/j.ygyno.2010.06.008 (2010).
Hu, L., Bikle, D. D. & Oda, Y. Reciprocal role of vitamin D receptor on beta-catenin regulated keratinocyte proliferation and differentiation. J. Steroid. Biochem. Mol. Biol. 144(Pt A), 237–241. https://doi.org/10.1016/j.jsbmb.2013.11.002 (2014).
Larriba, M. J. et al. Vitamin D receptor deficiency enhances Wnt/beta-catenin signaling and tumor burden in colon cancer. PLoS ONE 6, e23524. https://doi.org/10.1371/journal.pone.0023524 (2011).
MacDonald, B. T., Tamai, K. & He, X. Wnt/beta-catenin signaling: Components, mechanisms, and diseases. Dev. Cell 17, 9–26. https://doi.org/10.1016/j.devcel.2009.06.016 (2009).
Boyer, A., Goff, A. K. & Boerboom, D. WNT signaling in ovarian follicle biology and tumorigenesis. Trends Endocrinol. Metab. 21, 25–32. https://doi.org/10.1016/j.tem.2009.08.005 (2010).
Arend, R. C., Londono-Joshi, A. I., Straughn, J. M. Jr. & Buchsbaum, D. J. The Wnt/beta-catenin pathway in ovarian cancer: A review. Gynecol. Oncol. 131, 772–779. https://doi.org/10.1016/j.ygyno.2013.09.034 (2013).
Anastas, J. N. & Moon, R. T. WNT signalling pathways as therapeutic targets in cancer. Nat. Rev. Cancer 13, 11–26. https://doi.org/10.1038/nrc3419 (2013).
Kinzler, K. W. & Vogelstein, B. Lessons from hereditary colorectal cancer. Cell 87, 159–170 (1996).
Polakis, P. Drugging Wnt signalling in cancer. EMBO J. 31, 2737–2746. https://doi.org/10.1038/emboj.2012.126 (2012).
Gatcliffe, T. A., Monk, B. J., Planutis, K. & Holcombe, R. F. Wnt signaling in ovarian tumorigenesis. Int. J. Gynecol. Cancer 18, 954–962. https://doi.org/10.1111/j.1525-1438.2007.01127.x (2008).
Rask, K. et al. Wnt-signalling pathway in ovarian epithelial tumours: Increased expression of beta-catenin and GSK3beta. Br. J. Cancer 89, 1298–1304. https://doi.org/10.1038/sj.bjc.6601265 (2003).
Wend, P., Holland, J. D., Ziebold, U. & Birchmeier, W. Wnt signaling in stem and cancer stem cells. Semin. Cell Dev. Biol. 21, 855–863. https://doi.org/10.1016/j.semcdb.2010.09.004 (2010).
Ong, M. S. et al. ’Lnc’-ing Wnt in female reproductive cancers: Therapeutic potential of long non-coding RNAs in Wnt signalling. Br. J. Pharmacol. 174, 4684–4700. https://doi.org/10.1111/bph.13958 (2017).
Shen, P., Pichler, M., Chen, M., Calin, G. A. & Ling, H. To Wnt or lose: the missing non-coding linc in colorectal cancer. Int. J. Mol. Sci. 18. https://doi.org/10.3390/ijms18092003 (2017).
Hu, P. et al. NBAT1 suppresses breast cancer metastasis by regulating DKK1 via PRC2. Oncotarget 6, 32410–32425. https://doi.org/10.18632/oncotarget.5609 (2015).
Ohtsuka, M. et al. H19 noncoding RNA, an independent prognostic factor, regulates essential Rb-E2F and CDK8-beta-catenin signaling in colorectal cancer. EBioMedicine 13, 113–124. https://doi.org/10.1016/j.ebiom.2016.10.026 (2016).
Di Cecilia, S. et al. RBM5-AS1 is critical for self-renewal of colon cancer stem-like cells. Cancer Res. 76, 5615–5627. https://doi.org/10.1158/0008-5472.CAN-15-1824 (2016).
Fang, C. et al. Long non-coding RNA HNF1A-AS1 mediated repression of miR-34a/SIRT1/p53 feedback loop promotes the metastatic progression of colon cancer by functioning as a competing endogenous RNA. Cancer Lett. 410, 50–62. https://doi.org/10.1016/j.canlet.2017.09.012 (2017).
Stockley, J. et al. The RNA-binding protein hnRNPA2 regulates beta-catenin protein expression and is overexpressed in prostate cancer. RNA Biol. 11, 755–765 (2014).
Scully, R. E. & Sobin, L. H. Histologic typing of ovarian tumors. Arch. Pathol. Lab. Med. 111, 794–795 (1987).
Shepherd, J. H. Revised FIGO staging for gynaecological cancer. Br. J. Obstet. Gynaecol. 96, 889–892 (1989).
Messeguer, X. et al. PROMO: Detection of known transcription regulatory elements using species-tailored searches. Bioinformatics 18, 333–334 (2002).
Farre, D. et al. Identification of patterns in biological sequences at the ALGGEN server: PROMO and MALGEN. Nucleic Acids Res. 31, 3651–3653 (2003).
Gao, J. et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal 6, pl1. https://doi.org/10.1126/scisignal.2004088 (2013).
Cerami, E. et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2, 401–404. https://doi.org/10.1158/2159-8290.CD-12-0095 (2012).
DerSimonian, R. & Laird, N. Meta-analysis in clinical trials. Control Clin. Trials 7, 177–188. https://doi.org/10.1016/0197-2456(86)90046-2 (1986).
Funding
The study was funded by University of Hawaii Cancer Center.
Author information
Authors and Affiliations
Contributions
Research design: Y.F., Z.W., H.Yu. Data acquisition: Y.F., D.K., N.B., Z.W., M.Tiirikainen., H.Yu. Data analysis: Y.F., Z.W., I.P., H.Yu. Data interpretation: Y.F., D.K., N.B., Z.W., I.P., M.Tius., M.Tiirikainen., CR, H.Yang., H.Yu. Manuscript preparation: Y.F., D.K., N.B., Z.W., I.P., M.Tius., M.Tiirikainen., C.R., H.Yang., H.Yu.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fu, Y., Katsaros, D., Biglia, N. et al. Vitamin D receptor upregulates lncRNA TOPORS-AS1 which inhibits the Wnt/β-catenin pathway and associates with favorable prognosis of ovarian cancer. Sci Rep 11, 7484 (2021). https://doi.org/10.1038/s41598-021-86923-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-021-86923-7
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.