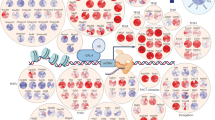
Abstract
Discovery of interaction sites between RNA-binding proteins (RBPs) and their RNA targets plays a critical role in enabling our understanding of how these RBPs control RNA processing and regulation. Cross-linking and immunoprecipitation (CLIP) provides a generalizable, transcriptome-wide method by which RBP/RNA complexes are purified and sequenced to identify sites of intermolecular contact. By simplifying technical challenges in prior CLIP methods and incorporating the generation of and quantitative comparison against size-matched input controls, the single-end enhanced CLIP (seCLIP) protocol allows for the profiling of these interactions with high resolution, efficiency and scalability. Here, we present a step-by-step guide to the seCLIP method, detailing critical steps and offering insights regarding troubleshooting and expected results while carrying out the ~4-d protocol. Furthermore, we describe a comprehensive bioinformatics pipeline that offers users the tools necessary to process two replicate datasets and identify reproducible and significant peaks for an RBP of interest in ~2 d.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Code availability
All code is made freely and publicly available under the BSD-3 license. Custom scripts and workflow definition files described in this paper may be found at https://doi.org/10.5281/zenodo.507659149. Up-to-date versions may be found on GitHub at https://github.com/yeolab/eclip.
References
Gerstberger, S., Hafner, M. & Tuschl, T. A census of human RNA-binding proteins. Nat. Rev. Genet. 15, 829–845 (2014).
Hentze, M. W., Castello, A., Schwarzl, T. & Preiss, T. A brave new world of RNA-binding proteins. Nat. Rev. Mol. Cell Biol. 19, 327–341 (2018).
Lukong, K. E., Chang, K., Khandjian, E. W. & Richard, S. RNA-binding proteins in human genetic disease. Trends Genet. 24, 416–425 (2008).
Nussbacher, J. K., Batra, R., Lagier-Tourenne, C. & Yeo, G. W. RNA-binding proteins in neurodegeneration: Seq and you shall receive. Trends Neurosci. 38, 226–236 (2015).
Brinegar, A. E. & Cooper, T. A. Roles for RNA-binding proteins in development and disease. Brain Res. 1647, 1–8 (2016).
Conlon, E. G. & Manley, J. L. RNA-binding proteins in neurodegeneration: mechanisms in aggregate. Genes Dev. 31, 1509–1528 (2017).
Lee, F. C. Y. & Ule, J. Advances in CLIP technologies for studies of protein–RNA interactions. Mol. Cell 69, 354–369 (2018).
Ule, J. et al. CLIP identifies Nova-regulated RNA networks in the brain. Science 302, 1212–1215 (2003).
Van Nostrand, E. L. et al. Robust transcriptome-wide discovery of RNA-binding protein binding sites with enhanced CLIP (eCLIP). Nat. Methods 13, 508–514 (2016).
Van Nostrand, E. L. et al. Robust, cost-effective profiling of RNA binding protein targets with single-end crosslinking and immunoprecipitation (seCLIP). Methods Mol. Biol. 1648, 177–200 (2017).
Van Nostrand, E. L. et al. Principles of RNA processing from analysis of enhanced CLIP maps for 150 RNA binding proteins. Genome Biol. 21, 90 (2020).
Shah, A., Qian, Y., Weyn-Vanhentenryck, S. M. & Zhang, C. CLIP Tool Kit (CTK): a flexible and robust pipeline to analyze CLIP sequencing data. Bioinformatics 33, 566–567 (2017).
Haberman, N. et al. Insights into the design and interpretation of iCLIP experiments. Genome Biol. 18, 7 (2017).
Wheeler, E. C., Van Nostrand, E. L. & Yeo, G. W. Advances and challenges in the detection of transcriptome-wide protein-RNA interactions. Wiley Interdiscip. Rev. RNA 9, 397–414 (2018).
Zarnegar, B. J. et al. irCLIP platform for efficient characterization of protein–RNA interactions. Nat. Methods 13, 489–492 (2016).
Huppertz, I. et al. iCLIP: protein-RNA interactions at nucleotide resolution. Methods 65, 274–287 (2014).
Buchbender, A. et al. Improved library preparation with the new iCLIP2 protocol. Methods 178, 33–48 (2020).
Kaczynski, T., Hussain, A. & Farkas, M. Quick-irCLIP: interrogating protein-RNA interactions using a rapid and simple cross-linking and immunoprecipitation technique. MethodsX 6, 1292–1304 (2019).
Martin, M. Cutadapt removes adapter sequences from high-throughput sequencing reads. EMBnet J. 17, e10 (2011).
Dobin, A. et al. STAR: ultrafast universal RNA-seq aligner. Bioinformatics 29, 15–21 (2013).
Smith, T., Heger, A. & Sudbery, I. UMI-tools: modeling sequencing errors in Unique Molecular Identifiers to improve quantification accuracy. Genome Res. 27, 491–499 (2017).
Merkel, D. Docker: lightweight Linux containers for consistent development and deployment. Linux J. 2014, 81–84 (2014).
Kurtzer, G. M., Sochat, V. & Bauer, M. W. Singularity: scientific containers for mobility of compute. PLoS One 12, e0177459 (2017).
Crusoe, M. R. Methods included: standardizing computational reuse and portability with the Common Workflow Language. Preprint at https://doi.org/10.48550/arXiv.2105.07028 (2021).
Köster, J. & Rahmann, S. Snakemake—a scalable bioinformatics workflow engine. Bioinformatics 28, 2520–2522 (2012).
Di Tommaso, P. et al. Nextflow enables reproducible computational workflows. Nat. Biotechnol. 35, 316–319 (2017).
Voss, K., Van der Auwera, G. & Gentry, J. Full-stack Genomics Pipelining with GATK4 + WDL + Cromwell [version 1; not peer reviewed]. F1000Res https://f1000research.com/slides/6-1381 (2017).
Deelman, E. et al. Pegasus, a workflow management system for science automation. Future Gener. Comput. Syst. 46, 17–35 (2015).
Van Nostrand, E. L. et al. A large-scale binding and functional map of human RNA-binding proteins. Nature 583, 711–719 (2020).
Conway, A. E. et al. Enhanced CLIP uncovers IMP protein-RNA targets in human pluripotent stem cells important for cell adhesion and survival. Cell Rep. 15, 666–679 (2016).
Van Nostrand, E. L. et al. CRISPR/Cas9-mediated integration enables TAG-eCLIP of endogenously tagged RNA binding proteins. Methods 118–119, 50–59 (2017).
Krach, F. et al. Transcriptome-pathology correlation identifies interplay between TDP-43 and the expression of its kinase CK1E in sporadic ALS. Acta Neuropathol. 136, 405–423 (2018).
Di Stefano, B. et al. The RNA helicase DDX6 controls cellular plasticity by modulating P-body homeostasis. Cell Stem Cell 25, 622–638.e13 (2019).
Xu, Q. et al. Enhanced crosslinking immunoprecipitation (eCLIP) method for efficient identification of protein-bound RNA in mouse testis. J. Vis. Exp. 2019, e59681 (2019).
Ke, S. et al. A majority of m6A residues are in the last exons, allowing the potential for 3′ UTR regulation. Genes Dev. 29, 2037–2053 (2015).
Linder, B. et al. Single-nucleotide-resolution mapping of m6A and m6Am throughout the transcriptome. Nat. Methods 12, 767–772 (2015).
Patil, D. P., Pickering, B. F. & Jaffrey, S. R. Reading m6A in the transcriptome: m6A-binding proteins. Trends Cell Biol. 28, 113–127 (2018).
Li, X. et al. Base-resolution mapping reveals distinct m1A methylome in nuclear- and mitochondrial-encoded transcripts. Mol. Cell 68, 993–1005.e9 (2017).
Roberts, J. T., Porman, A. M. & Johnson, A. M. Identification of m6A residues at single-nucleotide resolution using eCLIP and an accessible custom analysis pipeline. RNA 27, 527–541 (2021).
Kadumuri, R. V. & Janga, S. C. Epitranscriptomic code and its alterations in human disease. Trends Mol. Med. 24, 886–903 (2018).
Tran, S. S. et al. Widespread RNA editing dysregulation in brains from autistic individuals. Nat. Neurosci. 22, 25–36 (2019).
Landt, S. G. et al. ChIP-seq guidelines and practices of the ENCODE and modENCODE consortia. Genome Res. 22, 1813–1831 (2012).
Li, Q., Brown, J. B., Huang, H. & Bickel, P. J. Measuring reproducibility of high-throughput experiments. Ann. Appl. Stat. 5, 1752–1779 (2011).
Vivian, J. et al. Toil enables reproducible, open source, big biomedical data analyses. Nat. Biotechnol. 35, 314–316 (2017).
Sundararaman, B. et al. Resources for the comprehensive discovery of functional RNA elements. Mol. Cell 61, 903–913 (2016).
Bao, W., Kojima, K. K. & Kohany, O. Repbase Update, a database of repetitive elements in eukaryotic genomes. Mob. DNA 6, 11 (2015).
Smith, T. & Sudbery, I. FAQ. UMI-tools. https://umi-tools.readthedocs.io/en/latest/faq.html (2020).
Wang, Z. F., Whitfield, M. L., Ingledue, T. C., Dominski, Z. & Marzluff, W. F. The protein that binds the 3′ end of histone mRNA: a novel RNA-binding protein required for histone pre-mRNA processing. Genes Dev. 10, 3028–3040 (1996).
Yee, B., Domissy, A. & Crusoe, M. R. YeoLab/eclip. https://github.com/yeolab/eclip (2021).
Acknowledgements
The authors thank Yeo laboratory member K. Rothamel for critical reading of the manuscript. This work was supported by grants from the US National Institutes of Health (HG009889 and HG004659 to G.W.Y. and HG009530 to E.L.V.N.).
Author information
Authors and Affiliations
Contributions
S.M.B. wrote the sections of the manuscript pertaining to the experimental protocol and contributed to the development of the experimental methods. B.A.Y. wrote the sections of the manuscript pertaining to the bioinformatics methods. G.A.P. contributed to the development of bioinformatics methods. J.R.M. and S.S.P. contributed to developing the experimental methods and produced the data in Fig. 3. A.A.S. contributed to the development of experimental methods. A.C.S. produced the data presented in Supplementary Figure 2. E.L.V.N. contributed to the development of all experimental and bioinformatics methods described and directed writing of the manuscript. G.W.Y. directed the development of all experimental and bioinformatics methods described and the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
G.W.Y. is co-founder, member of the Board of Directors, on the Science Advisory Board, an equity holder and a paid consultant for Locanabio and Eclipse BioInnovations. G.W.Y. is a visiting professor at the National University of Singapore. G.W.Y.’s interest(s) have been reviewed and approved by the University of California, San Diego in accordance with its conflict-of-interest policies. A.A.S. is co-founder and Research & Development Director for Eclipse Bioinnovations. E.L.V.N. is co-founder, member of the Board of Directors, on the Science Advisory Board, an equity holder and a paid consultant for Eclipse BioInnovations. E.L.V.N.’s interest(s) have been reviewed and approved by Baylor College of Medicine in accordance with its conflict-of-interest policies. The authors declare no other competing interests.
Peer review
Peer review information
Nature Protocols thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Van Nostrand, E. et al. Nat. Methods 13, 508–514 (2016): https://doi.org/10.1038/nmeth.3810
Van Nostrand, E. et al. Nature 583, 711–719 (2020): https://doi.org/10.1038/s41586-020-2077-3
Van Nostrand, E. et al. Methods Mol. Biol. 1648, 177–200 (2017): https://doi.org/10.1007/978-1-4939-7204-3_14
Supplementary information
Supplementary Information
Supplementary Figs. 1 and 2.
Supplementary Data 1
Source data for Supplementary Fig. 2.
Rights and permissions
About this article
Cite this article
Blue, S.M., Yee, B.A., Pratt, G.A. et al. Transcriptome-wide identification of RNA-binding protein binding sites using seCLIP-seq. Nat Protoc 17, 1223–1265 (2022). https://doi.org/10.1038/s41596-022-00680-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-022-00680-z
This article is cited by
-
Mapping protein–RNA binding in plants with individual-nucleotide-resolution UV cross-linking and immunoprecipitation (plant iCLIP2)
Nature Protocols (2024)
-
hnRNP A1 dysfunction alters RNA splicing and drives neurodegeneration in multiple sclerosis (MS)
Nature Communications (2024)
-
Profiling of RNA-binding protein binding sites by in situ reverse transcription-based sequencing
Nature Methods (2024)
-
A super-enhancer-regulated RNA-binding protein cascade drives pancreatic cancer
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.