Abstract
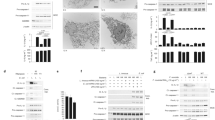
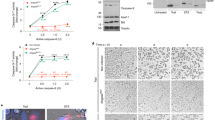
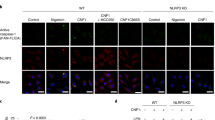
Macrophages mediate crucial innate immune responses via caspase-1-dependent processing and secretion of interleukin 1β (IL-1β) and IL-18. Although infection with wild-type Salmonella typhimurium is lethal to mice, we show here that a strain that persistently expresses flagellin was cleared by the cytosolic flagellin-detection pathway through the activation of caspase-1 by the NLRC4 inflammasome; however, this clearance was independent of IL-1β and IL-18. Instead, caspase-1-induced pyroptotic cell death released bacteria from macrophages and exposed the bacteria to uptake and killing by reactive oxygen species in neutrophils. Similarly, activation of caspase-1 cleared unmanipulated Legionella pneumophila and Burkholderia thailandensis by cytokine-independent mechanisms. This demonstrates that activation of caspase-1 clears intracellular bacteria in vivo independently of IL-1β and IL-18 and establishes pyroptosis as an efficient mechanism of bacterial clearance by the innate immune system.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Miao, E.A., Andersen-Nissen, E., Warren, S.E. & Aderem, A. TLR5 and Ipaf: dual sensors of bacterial flagellin in the innate immune system. Semin Immunopathol 29, 275–288 (2007).
Dinarello, C.A. Immunological and inflammatory functions of the interleukin-1 family. Annu. Rev. Immunol. 27, 519–550 (2009).
Szretter, K.J. et al. Role of host cytokine responses in the pathogenesis of avian H5N1 influenza viruses in mice. J. Virol. 81, 2736–2744 (2007).
Schmitz, N., Kurrer, M., Bachmann, M.F. & Kopf, M. Interleukin-1 is responsible for acute lung immunopathology but increases survival of respiratory influenza virus infection. J. Virol. 79, 6441–6448 (2005).
Kozak, W. et al. Thermal and behavioral effects of lipopolysaccharide and influenza in interleukin-1β-deficient mice. Am. J. Physiol. 269, R969–R977 (1995).
Van Der Sluijs, K.F. et al. Enhanced viral clearance in interleukin-18 gene-deficient mice after pulmonary infection with influenza A virus. Immunology 114, 112–120 (2005).
Liu, B. et al. Interleukin-18 improves the early defence system against influenza virus infection by augmenting natural killer cell-mediated cytotoxicity. J. Gen. Virol. 85, 423–428 (2004).
Sansonetti, P.J. et al. Caspase-1 activation of IL-1β and IL-18 are essential for Shigella flexneri-induced inflammation. Immunity 12, 581–590 (2000).
Bergsbaken, T., Fink, S.L. & Cookson, B.T. Pyroptosis: host cell death and inflammation. Nat. Rev. Microbiol. 7, 99–109 (2009).
Hueck, C.J. Type III protein secretion systems in bacterial pathogens of animals and plants. Microbiol. Mol. Biol. Rev. 62, 379–433 (1998).
Miao, E.A. & Warren, S.E. Innate immune detection of bacterial virulence factors via the NLRC4 inflammasome. J. Clin. Immunol. 30, 502–506 (2010).
Miao, E.A. et al. Innate immune detection of the type III secretion apparatus through the NLRC4 inflammasome. Proc. Natl. Acad. Sci. USA 107, 3076–3080 (2010).
Lara-Tejero, M. et al. Role of the caspase-1 inflammasome in Salmonella typhimurium pathogenesis. J. Exp. Med. 203, 1407–1412 (2006).
Raupach, B., Peuschel, S.K., Monack, D.M. & Zychlinsky, A. Caspase-1-mediated activation of interleukin-1β (IL-1β) and IL-18 contributes to innate immune defenses against Salmonella enterica serovar Typhimurium infection. Infect. Immun. 74, 4922–4926 (2006).
Ibarra, J.A. & Steele-Mortimer, O. Salmonella–the ultimate insider. Salmonella virulence factors that modulate intracellular survival. Cell. Microbiol. 11, 1579–1586 (2009).
Cummings, L.A., Wilkerson, W.D., Bergsbaken, T. & Cookson, B.T. In vivo, fliC expression by Salmonella enterica serovar Typhimurium is heterogeneous, regulated by ClpX, and anatomically restricted. Mol. Microbiol. 61, 795–809 (2006).
Miao, E.A. et al. Cytoplasmic flagellin activates caspase-1 and secretion of interleukin 1β via Ipaf. Nat. Immunol. 7, 569–575 (2006).
Miggin, S.M. et al. NF-κB activation by the Toll-IL-1 receptor domain protein MyD88 adapter-like is regulated by caspase-1. Proc. Natl. Acad. Sci. USA 104, 3372–3377 (2007).
Mariathasan, S. et al. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430, 213–218 (2004).
Suzuki, T. et al. Differential regulation of caspase-1 activation, pyroptosis, and autophagy via Ipaf and ASC in Shigella-infected macrophages. PLoS Pathog. 3, e111 (2007).
Franchi, L. et al. Critical role for Ipaf in Pseudomonas aeruginosa-induced caspase-1 activation. Eur. J. Immunol. 37, 3030–3039 (2007).
Case, C.L., Shin, S. & Roy, C.R. Asc and Ipaf Inflammasomes direct distinct pathways for caspase-1 activation in response to Legionella pneumophila. Infect. Immun. 77, 1981–1991 (2009).
Sumimoto, H., Miyano, K. & Takeya, R. Molecular composition and regulation of the Nox family NAD(P)H oxidases. Biochem. Biophys. Res. Commun. 338, 677–686 (2005).
Fischbach, M.A. et al. The pathogen-associated iroA gene cluster mediates bacterial evasion of lipocalin 2. Proc. Natl. Acad. Sci. USA 103, 16502–16507 (2006).
Gunn, J.S. The Salmonella PmrAB regulon: lipopolysaccharide modifications, antimicrobial peptide resistance and more. Trends Microbiol. 16, 284–290 (2008).
Bueno, S.M., Gonzalez, P.A., Schwebach, J.R. & Kalergis, A.M. T cell immunity evasion by virulent Salmonella enterica. Immunol. Lett. 111, 14–20 (2007).
Mariathasan, S., Weiss, D.S., Dixit, V.M. & Monack, D.M. Innate immunity against Francisella tularensis is dependent on the ASC/caspase-1 axis. J. Exp. Med. 202, 1043–1049 (2005).
Duprez, L., Wirawan, E., Vanden Berghe, T. & Vandenabeele, P. Major cell death pathways at a glance. Microbes Infect. 11, 1050–1062 (2009).
Zychlinsky, A., Prevost, M.C. & Sansonetti, P.J. Shigella flexneri induces apoptosis in infected macrophages. Nature 358, 167–169 (1992).
Schroder, K., Muruve, D.A. & Tschopp, J. Innate immunity: cytoplasmic DNA sensing by the AIM2 inflammasome. Curr. Biol. 19, R262–R265 (2009).
Wu, J., Fernandes-Alnemri, T. & Alnemri, E.S. Involvement of the AIM2, NLRC4, and NLRP3 inflammasomes in caspase-1 activation by Listeria monocytogenes. J. Clin. Immunol. 30, 693–702 (2010).
Warren, S.E. et al. Cutting edge: Cytosolic bacterial DNA activates the inflammasome via Aim2. J. Immunol. 185, 818–821 (2010).
Tsuchiya, K. et al. Involvement of absent in melanoma 2 in inflammasome activation in macrophages infected with Listeria monocytogenes. J. Immunol. 185, 1186–1195 (2010).
Sauer, J.D. et al. Listeria monocytogenes triggers AIM2-mediated pyroptosis upon infrequent bacteriolysis in the macrophage cytosol. Cell Host Microbe 7, 412–419 (2010).
Rathinam, V.A. et al. The AIM2 inflammasome is essential for host defense against cytosolic bacteria and DNA viruses. Nat. Immunol. 11, 395–402 (2010).
Kim, S. et al. Listeria monocytogenes is sensed by the NLRP3 and AIM2 inflammasome. Eur. J. Immunol. 40, 1545–1551 (2010).
Jones, J.W. et al. Absent in melanoma 2 is required for innate immune recognition of Francisella tularensis. Proc. Natl. Acad. Sci. USA 107, 9771–9776 (2010).
Fernandes-Alnemri, T. et al. The AIM2 inflammasome is critical for innate immunity to Francisella tularensis. Nat. Immunol. 11, 385–393 (2010).
Warren, S.E., Mao, D.P., Rodriguez, A.E., Miao, E.A. & Aderem, A. Multiple Nod-like receptors activate caspase 1 during Listeria monocytogenes infection. J. Immunol. 180, 7558–7564 (2008).
Craven, R.R. et al. Staphylococcus aureus α-hemolysin activates the NLRP3-inflammasome in human and mouse monocytic cells. PLoS One 4, e7446 (2009).
Munoz-Planillo, R., Franchi, L., Miller, L.S. & Nunez, G. A critical role for hemolysins and bacterial lipoproteins in Staphylococcus aureus-induced activation of the Nlrp3 inflammasome. J. Immunol. 183, 3942–3948 (2009).
Broz, P. et al. Redundant roles for inflammasome receptors NLRP3 and NLRC4 in host defense against Salmonella. J. Exp. Med. 207, 1745–1755 (2010).
Beuzon, C.R., Unsworth, K.E. & Holden, D.W. In vivo genetic analysis indicates that PhoP-PhoQ and the Salmonella pathogenicity island 2 type III secretion system contribute independently to Salmonella enterica serovar Typhimurium virulence. Infect. Immun. 69, 7254–7261 (2001).
Miao, E.A. et al. Salmonella typhimurium leucine-rich repeat proteins are targeted to the SPI1 and SPI2 type III secretion systems. Mol. Microbiol. 34, 850–864 (1999).
Shornick, L.P. et al. Mice deficient in IL-1β manifest impaired contact hypersensitivity to trinitrochlorobenzone. J. Exp. Med. 183, 1427–1436 (1996).
Yamamoto, M. et al. Essential role for TIRAP in activation of the signalling cascade shared by TLR2 and TLR4. Nature 420, 324–329 (2002).
Uematsu, S. et al. Detection of pathogenic intestinal bacteria by Toll-like receptor 5 on intestinal CD11c+ lamina propria cells. Nat. Immunol. 7, 868–874 (2006).
Kuida, K. et al. Altered cytokine export and apoptosis in mice deficient in interleukin-1 beta converting enzyme. Science 267, 2000–2003 (1995).
Beuzon, C.R. & Holden, D.W. Use of mixed infections with Salmonella strains to study virulence genes and their interactions in vivo. Microbes Infect. 3, 1345–1352 (2001).
Acknowledgements
We thank D. Rodriguez for managing the mouse colony and members of the Aderem laboratory for critical review of the manuscript. Supported by the US National Institutes of Health (U54 AI057141 and AI052286).
Author information
Authors and Affiliations
Contributions
E.A.M., I.A.L. and A.A. conceived of the research plan and wrote the manuscript; E.A.M., I.A.L., D.P.M., M.D., A.S. and M.D.W. planned and did experiments; and P.M.T. planned and did histological analyses.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–2 and Tables 1–3 (PDF 491 kb)
Rights and permissions
About this article
Cite this article
Miao, E., Leaf, I., Treuting, P. et al. Caspase-1-induced pyroptosis is an innate immune effector mechanism against intracellular bacteria. Nat Immunol 11, 1136–1142 (2010). https://doi.org/10.1038/ni.1960
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1960
This article is cited by
-
cGAS-STING, inflammasomes and pyroptosis: an overview of crosstalk mechanism of activation and regulation
Cell Communication and Signaling (2024)
-
Salidroside protects RGC from pyroptosis in diabetes-induced retinopathy associated with NLRP3, NFEZL2 and NGKB1, revealed by network pharmacology analysis and experimental validation
European Journal of Medical Research (2024)
-
Vitamin A deficiency impairs neutrophil-mediated control of Salmonella via SLC11A1 in mice
Nature Microbiology (2024)
-
Gasdermin E dictates inflammatory responses by controlling the mode of neutrophil death
Nature Communications (2024)
-
Preparation and characterization of monoclonal antibodies against porcine gasdermin D protein
Applied Microbiology and Biotechnology (2024)