Abstract
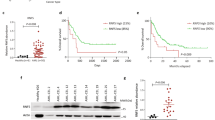
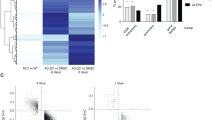
Genetic and epigenetic aberrations contribute to the initiation and progression of acute myeloid leukemia (AML). GFI1, a zinc-finger transcriptional repressor, exerts its function by recruiting histone deacetylases to target genes. We present data that low expression of GFI1 is associated with an inferior prognosis of AML patients. To elucidate the mechanism behind this, we generated a humanized mouse strain with reduced GFI1 expression (GFI1-KD). Here we show that AML development induced by onco-fusion proteins such as MLL-AF9 or NUP98-HOXD13 is accelerated in mice with low human GFI1 expression. Leukemic cells from animals that express low levels of GFI1 show increased H3K9 acetylation compared to leukemic cells from mice with normal human GFI1 expression, resulting in the upregulation of genes involved in leukemogenesis. We investigated a new epigenetic therapy approach for this subgroup of AML patients. We could show that AML blasts from GFI1-KD mice and from AML patients with low GFI1 levels were more sensitive to treatment with histone acetyltransferase inhibitors than cells with normal GFI1 expression levels. We suggest therefore that GFI1 has a dose-dependent role in AML progression and development. GFI1 levels are involved in epigenetic regulation, which could open new therapeutic approaches for AML patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Estey E, Dohner H . Acute myeloid leukaemia. Lancet 2006; 368: 1894–1907.
Dohner H, Estey EH, Amadori S, Appelbaum FR, Buchner T, Burnett AK et al. Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood 2010; 115: 453–474.
Tefferi A, Vardiman JW . Myelodysplastic syndromes. N Engl J Med 2009; 361: 1872–1885.
Malcovati L, Hellstrom-Lindberg E, Bowen D, Ades L, Cermak J, Del Canizo C et al. Diagnosis and treatment of primary myelodysplastic syndromes in adults: recommendations from the European LeukemiaNet. Blood 2013; 122: 2943–2964.
Sekeres MA, Swern AS, Fenaux P, Greenberg PL, Sanz GF, Bennett JM et al. Validation of the IPSS-R in lenalidomide-treated, lower-risk myelodysplastic syndrome patients with del(5q). Blood Cancer J 2014; 4: e242.
Steidl U, Steidl C, Ebralidze A, Chapuy B, Han HJ, Will B et al. A distal single nucleotide polymorphism alters long-range regulation of the PU.1 gene in acute myeloid leukemia. J Clin Invest 2007; 117: 2611–2620.
Bejar R, Levine R, Ebert BL . Unraveling the molecular pathophysiology of myelodysplastic syndromes. J Clin Oncol 2011; 29: 504–515.
Celton M, Forest A, Gosse G, Lemieux S, Hebert J, Sauvageau G et al. Epigenetic regulation of GATA2 and its impact on normal karyotype acute myeloid leukemia. Leukemia 2014; 28: 1617–1626.
van Riel B, Rosenbauer F . Epigenetic control of hematopoiesis: the PU.1 chromatin connection. Biol Chem 2014; 395: 1265–1274.
Rosenbauer F, Wagner K, Kutok JL, Iwasaki H, Le Beau MM, Okuno Y et al. Acute myeloid leukemia induced by graded reduction of a lineage-specific transcription factor, PU.1. Nat Genet 2004; 36: 624–630.
Kuo MC, Liang DC, Huang CF, Shih YS, Wu JH, Lin TL et al. RUNX1 mutations are frequent in chronic myelomonocytic leukemia and mutations at the C-terminal region might predict acute myeloid leukemia transformation. Leukemia 2009; 23: 1426–1431.
Wouters BJ, Lowenberg B, Erpelinck-Verschueren CA, van Putten WL, Valk PJ, Delwel R . Double CEBPA mutations, but not single CEBPA mutations, define a subgroup of acute myeloid leukemia with a distinctive gene expression profile that is uniquely associated with a favorable outcome. Blood 2009; 113: 3088–3091.
Lessard J, Sauvageau G . Bmi-1 determines the proliferative capacity of normal and leukaemic stem cells. Nature 2003; 423: 255–260.
Bullinger L, Dohner K, Bair E, Frohling S, Schlenk RF, Tibshirani R et al. Use of gene-expression profiling to identify prognostic subclasses in adult acute myeloid leukemia. N Engl J Med 2004; 350: 1605–1616.
Heyd F, Chen R, Afshar K, Saba I, Lazure C, Fiolka K et al. The p150 subunit of the histone chaperone Caf-1 interacts with the transcriptional repressor Gfi1. Biochim Biophys Acta 2011; 1809: 255–261.
Montoya-Durango DE, Velu CS, Kazanjian A, Rojas ME, Jay CM, Longmore GD et al. Ajuba functions as a histone deacetylase-dependent co-repressor for autoregulation of the growth factor-independent-1 transcription factor. J Biol Chem 2008; 283: 32056–32065.
McGhee L, Bryan J, Elliott L, Grimes HL, Kazanjian A, Davis JN et al. Gfi-1 attaches to the nuclear matrix, associates with ETO (MTG8) and histone deacetylase proteins, and represses transcription using a TSA-sensitive mechanism. J Cell Biochem 2003; 89: 1005–1018.
Bell DW, Taguchi T, Jenkins NA, Gilbert DJ, Copeland NG, Gilks CB et al. Chromosomal localization of a gene, GF1, encoding a novel zinc finger protein reveals a new syntenic region between man and rodents. Cytogenet Cell Genet 1995; 70: 263–267.
Phelan JD, Saba I, Zeng H, Kosan C, Messer MS, Olsson HA et al. Growth factor independent-1 maintains Notch1-dependent transcriptional programming of lymphoid precursors. PLoS Genet 2013; 9: e1003713.
Karsunky H, Zeng H, Schmidt T, Zevnik B, Kluge R, Schmid KW et al. Inflammatory reactions and severe neutropenia in mice lacking the transcriptional repressor Gfi1. Nat Genet 2002; 30: 295–300.
Person RE, Li FQ, Duan Z, Benson KF, Wechsler J, Papadaki HA et al. Mutations in proto-oncogene GFI1 cause human neutropenia and target ELA2. Nat Genet 2003; 34: 308–312.
Zeng H, Yucel R, Kosan C, Klein-Hitpass L, Moroy T . Transcription factor Gfi1 regulates self-renewal and engraftment of hematopoietic stem cells. EMBO J 2004; 23: 4116–4125.
Hock H, Hamblen MJ, Rooke HM, Schindler JW, Saleque S, Fujiwara Y et al. Gfi-1 restricts proliferation and preserves functional integrity of haematopoietic stem cells. Nature 2004; 431: 1002–1007.
Marteijn JA, van der Meer LT, van Emst L, van Reijmersdal S, Wissink W, de Witte T et al. Gfi1 ubiquitination and proteasomal degradation is inhibited by the ubiquitin ligase Triad1. Blood 2007; 110: 3128–3135.
Zhu J, Jankovic D, Grinberg A, Guo L, Paul WE . Gfi-1 plays an important role in IL-2-mediated Th2 cell expansion. Proc Natl Acad Sci USA 2006; 103: 18214–18219.
Rathinam C, Klein C . Transcriptional repressor Gfi1 integrates cytokine-receptor signals controlling B-cell differentiation. PLoS One 2007; 2: e306.
Pargmann D, Yucel R, Kosan C, Saba I, Klein-Hitpass L, Schimmer S et al. Differential impact of the transcriptional repressor Gfi1 on mature CD4+ and CD8+ T lymphocyte function. Eur J Immunol 2007; 37: 3551–3563.
Zarebski A, Velu CS, Baktula AM, Bourdeau T, Horman SR, Basu S et al. Mutations in growth factor independent-1 associated with human neutropenia block murine granulopoiesis through colony stimulating factor-1. Immunity 2008; 28: 370–380.
Horman SR, Velu CS, Chaubey A, Bourdeau T, Zhu J, Paul WE et al. Gfi1 integrates progenitor versus granulocytic transcriptional programming. Blood 2009; 113: 5466–5475.
Spooner CJ, Cheng JX, Pujadas E, Laslo P, Singh H . A recurrent network involving the transcription factors PU.1 and Gfi1 orchestrates innate and adaptive immune cell fates. Immunity 2009; 31: 576–586.
Hock H, Orkin SH . Zinc-finger transcription factor Gfi-1: versatile regulator of lymphocytes, neutrophils and hematopoietic stem cells. Curr Opin Hematol 2006; 13: 1–6.
Phelan JD, Shroyer NF, Cook T, Gebelein B, Grimes HL . Gfi1-cells and circuits: unraveling transcriptional networks of development and disease. Curr Opin Hematol 2010; 17: 300–307.
van der Meer LT, Jansen JH, van der Reijden BA . Gfi1 and Gfi1b: key regulators of hematopoiesis. Leukemia 2010; 24: 1843–1834.
Karsunky H, Mende I, Schmidt T, Moroy T . High levels of the onco-protein Gfi-1 accelerate T-cell proliferation and inhibit activation induced T-cell death in Jurkat T-cells. Oncogene 2002; 21: 1571–1579.
Hock H, Hamblen MJ, Rooke HM, Traver D, Bronson RT, Cameron S et al. Intrinsic requirement for zinc finger transcription factor Gfi-1 in neutrophil differentiation. Immunity 2003; 18: 109–120.
Khandanpour C, Krongold J, Schutte J, Bouwman F, Vassen L, Gaudreau MC et al. The human GFI136N variant induces epigenetic changes at the Hoxa9 locus and accelerates K-RAS driven myeloproliferative disorder in mice. Blood 2012; 120: 4006–4017.
Mori N, Morosetti R, Mizoguchi H, Koeffler HP . Progression of myelodysplastic syndrome: allelic loss on chromosomal arm 1p. Br J Haematol 2003; 122: 226–230.
Huh HJ, Chae SL, Lee M, Hong KS, Mun YC, Seong CM et al. CD34, RAB20, PU.1 and GFI1 mRNA expression in myelodysplastic syndrome. Int J Lab Hematol 2009; 31: 344–351.
Valk PJ, Verhaak RG, Beijen MA, Erpelinck CA, Barjesteh van Waalwijk van Doorn-Khosrovani S, Boer JM et al. Prognostically useful gene-expression profiles in acute myeloid leukemia. N Engl J Med 2004; 350: 1617–1628.
Verhaak RG, Wouters BJ, Erpelinck CA, Abbas S, Beverloo HB, Lugthart S et al. Prediction of molecular subtypes in acute myeloid leukemia based on gene expression profiling. Haematologica 2009; 94: 131–134.
Yucel R, Kosan C, Heyd F, Moroy T . Gfi1:green fluorescent protein knock-in mutant reveals differential expression and autoregulation of the growth factor independence 1 (Gfi1) gene during lymphocyte development. J Biol Chem 2004; 279: 40906–40917.
Khandanpour C, Phelan JD, Vassen L, Schutte J, Chen R, Horman SR et al. Growth factor independence 1 antagonizes a p53-induced DNA damage response pathway in lymphoblastic leukemia. Cancer Cell 2013; 23: 200–214.
Wilson NK, Foster SD, Wang X, Knezevic K, Schutte J, Kaimakis P et al. Combinatorial transcriptional control in blood stem/progenitor cells: genome-wide analysis of ten major transcriptional regulators. Cell Stem Cell 2010; 7: 532–544.
Eppert K, Takenaka K, Lechman ER, Waldron L, Nilsson B, van Galen P et al. Stem cell gene expression programs influence clinical outcome in human leukemia. Nat Med 2011; 17: 1086–1093.
Holzenberger M, Lenzner C, Leneuve P, Zaoui R, Hamard G, Vaulont S et al. Cre-mediated germline mosaicism: a method allowing rapid generation of several alleles of a target gene. Nucleic Acids Res 2000; 28: E92.
Goardon N, Marchi E, Atzberger A, Quek L, Schuh A, Soneji S et al. Coexistence of LMPP-like and GMP-like leukemia stem cells in acute myeloid leukemia. Cancer Cell 2011; 19: 138–152.
Ye M, Zhang H, Yang H, Koche R, Staber PB, Cusan M et al. Hematopoietic differentiation is required for initiation of acute myeloid leukemia. Cell Stem Cell 2015; 17: 611–623.
Krivtsov AV, Twomey D, Feng Z, Stubbs MC, Wang Y, Faber J et al. Transformation from committed progenitor to leukaemia stem cell initiated by MLL-AF9. Nature 2006; 442: 818–822.
Chen W, Kumar AR, Hudson WA, Li Q, Wu B, Staggs RA et al. Malignant transformation initiated by Mll-AF9: gene dosage and critical target cells. Cancer Cell 2008; 13: 432–440.
Lin YW, Slape C, Zhang Z, Aplan PD . NUP98-HOXD13 transgenic mice develop a highly penetrant, severe myelodysplastic syndrome that progresses to acute leukemia. Blood 2005; 106: 287–295.
Saleque S, Kim J, Rooke HM, Orkin SH . Epigenetic regulation of hematopoietic differentiation by Gfi-1 and Gfi-1b is mediated by the cofactors CoREST and LSD1. Mol Cell 2007; 27: 562–572.
Lane AA, Chabner BA . Histone deacetylase inhibitors in cancer therapy. J Clin Oncol 2009; 27: 5459–5468.
Silva G, Cardoso BA, Belo H, Almeida AM . Vorinostat induces apoptosis and differentiation in myeloid malignancies: genetic and molecular mechanisms. PLoS One 2013; 8: e53766.
Arif M, Vedamurthy BM, Choudhari R, Ostwal YB, Mantelingu K, Kodaganur GS et al. Nitric oxide-mediated histone hyperacetylation in oral cancer: target for a water-soluble HAT inhibitor, CTK7A. Chem Biol 2010; 17: 903–913.
Garnett MJ, Edelman EJ, Heidorn SJ, Greenman CD, Dastur A, Lau KW et al. Systematic identification of genomic markers of drug sensitivity in cancer cells. Nature 2012; 483: 570–575.
Kok CH, Watkins DB, Leclercq T, D’Andrea RJ, Hughes TP, White DL . Low GFI1 expression in white blood cells of CP-CML patients at diagnosis is strongly associated with subsequent blastic transformation. Leukemia 2013; 27: 1427–1430.
Bachas C, Schuurhuis GJ, Zwaan CM, van den Heuvel-Eibrink MM, den Boer ML, de Bont ES et al. Gene expression profiles associated with pediatric relapsed AML. PLoS One 2015; 10: e0121730.
Sanda T, Li X, Gutierrez A, Ahn Y, Neuberg DS, O’Neil J et al. Interconnecting molecular pathways in the pathogenesis and drug sensitivity of T-cell acute lymphoblastic leukemia. Blood 2010; 115: 1735–1745.
Fiolka K, Hertzano R, Vassen L, Zeng H, Hermesh O, Avraham KB et al. Gfi1 and Gfi1b act equivalently in haematopoiesis, but have distinct, non-overlapping functions in inner ear development. EMBO Rep 2006; 7: 326–333.
Kirschbaum M, Gojo I, Goldberg SL, Bredeson C, Kujawski LA, Yang A et al. A phase 1 clinical trial of vorinostat in combination with decitabine in patients with acute myeloid leukaemia or myelodysplastic syndrome. Br J Haematol 2014; 167: 185–193.
Acknowledgements
We thank all patients for agreeing to participate in the different studies. We thank Saskia Grunwald and Renata Köster for excellent technical assistance and the team of the animal facility of University Hospital Essen for genotyping, technical and administrative assistance during the whole project. We thank Joachim Göthert, Bob Löwenberg, Peter Valk and Lucio Castilla for sharing resources, mice, technical information and patient data. The MCSV-MLL-AF9-IRES-GFP construct was kindly provided by Jay Hess and Lee Grimes. Cyrus Khandanpour was supported by a Max Eder Grant from the Deutsche Krebshilfe, Germany. Tarik Möröy’s lab was supported by a grant from the Canadian Institute for Health Research (MOP – 111011) and by the Leukemia & Lymphoma Society (LLS) and holds a Tier1 Canada Research Chair.
Author contributions
CK, TM. Methodology: CK, TM. Investigation: CK, TM, JMH, LB, HM, AH, CV, LV, FR,TR, BG, AG, SvdCC, YSA-M, AT, SMH, JS, RFL, JF, KL, BP, LKH, MH, GE, CT, UD. Writing Original Draft: CK, JMH, TM, LB, CV, AH. Writing & Editing: CK, JMH, TR, TM. Funding Acquisition: CK, JM, TM, UD, JR. Resources: CK, JM, TM, UD, CT, GE, MH, CT, GE, UD, BG. Supervision: CK, JM, UD, TM.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Cyrus Khandanpour received travel reimbursement for attending scientific conferences from Amgen and Chugai. Jaroslaw Maciejewski received speaker honoraria from Celgene and Alexion. The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Hönes, J., Botezatu, L., Helness, A. et al. GFI1 as a novel prognostic and therapeutic factor for AML/MDS. Leukemia 30, 1237–1245 (2016). https://doi.org/10.1038/leu.2016.11
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.11
This article is cited by
-
The prognostic and therapeutic potential of HO-1 in leukemia and MDS
Cell Communication and Signaling (2023)
-
GFI1B acts as a metabolic regulator in hematopoiesis and acute myeloid leukemia
Leukemia (2022)
-
Superior efficacy of co-targeting GFI1/KDM1A and BRD4 against AML and post-MPN secondary AML cells
Blood Cancer Journal (2021)
-
Reduced expression but not deficiency of GFI1 causes a fatal myeloproliferative disease in mice
Leukemia (2019)
-
LSD1 inhibition by tranylcypromine derivatives interferes with GFI1-mediated repression of PU.1 target genes and induces differentiation in AML
Leukemia (2019)