Featured
-
-
Article
| Open Access Direct catalytic asymmetric synthesis of α-chiral bicyclo[1.1.1]pentanes
Direct catalytic asymmetric synthesis of α-chiral bicyclo[1.1.1]pentanes
Bicyclo[1.1.1]pentanes (BCPs) are important motifs in contemporary drug design, however, approaches to BCPs featuring adjacent stereocenters are rather limited. Here, the authors report a photo- and organocatalyzed asymmetric addition of simple aldehydes to [1.1.1]propellane to generate enantioenriched α-chiral BCPs.
- Marie L. J. Wong
- , Alistair J. Sterling
- & Edward A. Anderson
-
Article
| Open Access Stereoselectivity in spontaneous assembly of rolled incommensurate carbon bilayers
Stereoselectivity in spontaneous assembly of rolled incommensurate carbon bilayers
Incommensurate pairing is a type of stereoisomerism, observed in carbon bilayers, that arises from the twisted orientations of the graphitic layers. Here, the authors create a finite molecular version of an incommensurate carbon bilayer in the form of two concentrically assembled cylindrical molecules.
- Taisuke Matsuno
- , Yutaro Ohtomo
- & Hiroyuki Isobe
-
Article
| Open Access Development of a mugineic acid family phytosiderophore analog as an iron fertilizer
Development of a mugineic acid family phytosiderophore analog as an iron fertilizer
Iron is an essential plant nutrient that is poorly bioavailable in alkaline soils, resulting in reduced agricultural productivity. Here, the authors report the synthesis of stable and cheap iron-chelator, proline-2’-deoxymugineic acid (PDMA), and demonstrate its utility as potential fertilizer.
- Motofumi Suzuki
- , Atsumi Urabe
- & Kosuke Namba
-
Article
| Open Access Selective E to Z isomerization of 1,3-Dienes Enabled by A Dinuclear Mechanism
Selective E to Z isomerization of 1,3-Dienes Enabled by A Dinuclear Mechanism
Geometric E to Z double C=C bond isomerization is challenging as it requires kinetic trapping of the Z-isomer with injection of chemical energy. Here, the authors report a dinuclear Pd(I)−Pd(I) complex that mediates selective isomerization of E-1,3-dienes to the Z-isomers without photoirradiation.
- Eiji Kudo
- , Kota Sasaki
- & Tetsuro Murahashi
-
Article
| Open Access Imine as a linchpin approach for meta-C–H functionalization
Imine as a linchpin approach for meta-C–H functionalization
Site-selective C–H functionalization still faces some challenges, such as the introduction and removal of an appropriate directing group. Here, the authors introduce a temporary directing group for selective meta-C–H functionalization of 2-arylbenzaldehydes via reversible imine formation.
- Sukdev Bag
- , Sadhan Jana
- & Debabrata Maiti
-
Article
| Open Access Catalytic asymmetric reductive hydroalkylation of enamides and enecarbamates to chiral aliphatic amines
Catalytic asymmetric reductive hydroalkylation of enamides and enecarbamates to chiral aliphatic amines
Enantiopure aliphatic amines are frequently encountered as chiral auxiliaries and synthetic intermediates for bioactive compounds. Here, the authors report a mild nickel-catalysed asymmetric reductive hydroalkylation to convert enamides and enecarbamates into α-branched chiral amines and derivatives.
- Jia-Wang Wang
- , Yan Li
- & Yao Fu
-
Article
| Open Access Marine furanocembranoids-inspired macrocycles enabled by Pd-catalyzed unactivated C(sp3)-H olefination mediated by donor/donor carbenes
Marine furanocembranoids-inspired macrocycles enabled by Pd-catalyzed unactivated C(sp3)-H olefination mediated by donor/donor carbenes
Furanocembranoid-like natural products with the alkene-substituted furan scaffold display a range of biological activities, but are difficult to access. Here, the authors report a modular biomimetic strategy to synthesise diverse alkene-substituted furan-containing macrolactams via palladium-catalysed unactivated Csp3-H olefination.
- Jiping Hao
- , Xueying Guo
- & Weibo Yang
-
Article
| Open Access Enantioselective construction of six- and seven-membered triorgano-substituted silicon-stereogenic heterocycles
Enantioselective construction of six- and seven-membered triorgano-substituted silicon-stereogenic heterocycles
Enantioenriched medium-sized silicon-stereogenic heterocycles are in high demand due to the potential use in functional materials. Here, the authors show a rhodium-catalyzed enantioselective synthesis of six- and seven-membered tri-organosubstituted silicon-stereogenic heterocycles.
- Shuyou Chen
- , Delong Mu
- & Chuan He
-
Matters Arising
| Open Access Reply to “A Thermodynamic assessment of the reported room-temperature chemical synthesis of C2”
Reply to “A Thermodynamic assessment of the reported room-temperature chemical synthesis of C2”
- Kazunori Miyamoto
- , Shodai Narita
- & Masanobu Uchiyama
-
Article
| Open Access Gold(I)-catalyzed intramolecular cyclization/intermolecular cycloaddition cascade as a fast track to polycarbocycles and mechanistic insights
Gold(I)-catalyzed intramolecular cyclization/intermolecular cycloaddition cascade as a fast track to polycarbocycles and mechanistic insights
Metal carbene is usually employed as a 1-carbon synthon or 3-carbon synthon in a variety of cycloaddition reactions. Here, the authors report a gold-catalyzed cascade protocol for the assembly of polycarbocyclic frameworks via a β-aryl gold-carbene intermediate which reacts as a 4-carbon synthon with alkenes in [4 + 2]-cycloadditions.
- Cheng Zhang
- , Kemiao Hong
- & Xinfang Xu
-
Article
| Open Access Three-component radical homo Mannich reaction
Three-component radical homo Mannich reaction
Due to the ionic nature of its mechanism, the Mannich reaction can only use non-enolizable aldehydes as substrates. Here, the authors expand the scope of the classical Mannich reaction to enolizable aldehydes by employing a radical process resulting in a streamlined synthesis of γ-amino-carbonyl compounds.
- Shuai Shi
- , Wenting Qiu
- & Zhankui Sun
-
Article
| Open Access Pd-catalyzed formal Mizoroki–Heck coupling of unactivated alkyl chlorides
Pd-catalyzed formal Mizoroki–Heck coupling of unactivated alkyl chlorides
The use of alkyl chlorides in Pd-catalyzed Mizoroki–Heck coupling reactions remains an unsolved problem despite their significant synthetic potential. Here, the authors show that alkyl chlorides can efficiently participate in Mizoroki–Heck cross-coupling reactions with excellent functional group compatibility via photoinduced Pd catalysis.
- Geun Seok Lee
- , Daeun Kim
- & Soon Hyeok Hong
-
Article
| Open Access Silane- and peroxide-free hydrogen atom transfer hydrogenation using ascorbic acid and cobalt-photoredox dual catalysis
Silane- and peroxide-free hydrogen atom transfer hydrogenation using ascorbic acid and cobalt-photoredox dual catalysis
Hydrogen atom transfer (HAT) hydrogenation of alkenes usually requires stoichiometric silanes and peroxides. Here, the authors report a silane- and peroxide-free HAT hydrogenation method using a combined cobalt/photoredox catalysis and vitamin C as a sole stoichiometric reactant.
- Yuji Kamei
- , Yusuke Seino
- & Shigeki Matsunaga
-
Article
| Open Access Ni-catalyzed regio- and stereo-defined intermolecular cross-electrophile dialkylation of alkynes without directing group
Ni-catalyzed regio- and stereo-defined intermolecular cross-electrophile dialkylation of alkynes without directing group
The synthesis of regio- and stereo-defined alkenes with multiple alkyl substituents is an unmet challenge. Here, the authors report a nickel-catalyzed intermolecular cross-dialkylation of alkynes devoid of directing or activating groups to afford multiple aliphatic substituted alkenes in a syn-selective fashion.
- Yi-Zhou Zhan
- , Nan Xiao
- & Wei Shu
-
Article
| Open Access General synthetic strategy for regioselective ultrafast formation of disulfide bonds in peptides and proteins
General synthetic strategy for regioselective ultrafast formation of disulfide bonds in peptides and proteins
Synthesis of peptides and proteins containing multiple disulfide bonds is challenging, limiting the elucidation of their biological functions. Here, the authors report a general synthetic strategy for fast formation of two and three disulfide bonds in peptides and proteins, and apply it to prepare several therapeutically important peptides.
- Shay Laps
- , Fatima Atamleh
- & Ashraf Brik
-
Article
| Open Access Palladium-catalyzed allene synthesis enabled by β-hydrogen elimination from sp2-carbon
Palladium-catalyzed allene synthesis enabled by β-hydrogen elimination from sp2-carbon
β-Hydrogen elimination is a fundamental reaction in palladium catalysis, however, the eliminated β-hydrogen is usually attached to a sp3-carbon. Here, the authors report a palladium-catalyzed cross-coupling reaction involving a β-vinylic hydrogen elimination from an allylic palladium intermediate.
- Ge Zhang
- , Yi-Kang Song
- & Guo-Qiang Lin
-
Article
| Open Access NiH-catalyzed asymmetric hydroarylation of N-acyl enamines to chiral benzylamines
NiH-catalyzed asymmetric hydroarylation of N-acyl enamines to chiral benzylamines
Chiral benzylamines are found in many pharmacologically active molecules. Here, the authors report an enantio- and regio-selective reductive hydroarylation of N-acyl enamines with a NiH/chiral bis-imidazoline catalytic system affording a range of enantioenriched benzylamines.
- Yuli He
- , Huayue Song
- & Shaolin Zhu
-
Article
| Open Access Molecular embroidering of graphene
Molecular embroidering of graphene
Covalently 2D-patterning graphene with different chemical functionalities is an attractive way to tailor its physical and chemical properties. Here, the authors realize spatially defined 2D-hetereoarchitectures of graphene via a strategy of molecular embroidering.
- Tao Wei
- , Malte Kohring
- & Andreas Hirsch
-
Perspective
| Open Access Catalytic enantioselective C(sp3)–H functionalization involving radical intermediates
Catalytic enantioselective C(sp3)–H functionalization involving radical intermediates
Enantioselective functionalization of ubiquitous C(sp3)–H bonds via radical species has witnessed a renaissance in the past years. Here, the authors summarize the main achievements in the field by identifying two main reaction pathways determining the stereochemistry and give an outlook on future challenges and opportunities.
- Chi Zhang
- , Zhong-Liang Li
- & Xin-Yuan Liu
-
Article
| Open Access Passerini-type reaction of boronic acids enables α-hydroxyketones synthesis
Passerini-type reaction of boronic acids enables α-hydroxyketones synthesis
Multicomponent reactions enable the rapid construction of diverse molecular scaffolds with modularity and step economy. In this work, the authors report the use of boronic acids as carbon nucleophiles in a Passerini-type three-component coupling reaction towards an expanded inventory of α-hydroxyketones.
- Kai Yang
- , Feng Zhang
- & Qiuling Song
-
Article
| Open Access Metal-free atom transfer radical polymerization with ppm catalyst loading under sunlight
Metal-free atom transfer radical polymerization with ppm catalyst loading under sunlight
Organocatalytic atom transfer radical polymerization (O-ATRP) is attractive due to its metal-free nature but catalysts are rarely applied at a low loading. Here the authors introduce a catalyst design logic based on heteroatom-doping of polycyclic arenes, which led to the discovery of oxygen-doped anthanthrene as an organic photoredox catalysts for O-ATRP.
- Qiang Ma
- , Jinshuai Song
- & Saihu Liao
-
Article
| Open Access Enantioselective preparation of mechanically planar chiral rotaxanes by kinetic resolution strategy
Enantioselective preparation of mechanically planar chiral rotaxanes by kinetic resolution strategy
Since the discovery of mechanically planar chiral rotaxanes and topologically chiral catenanes, their asymmetric synthesis has been a long-standing challenge. Here, the authors report enantioselective preparation of mechanically planar chiral rotaxanes with up to 99.9% ee in 29% yield.
- Ayumi Imayoshi
- , Bhatraju Vasantha Lakshmi
- & Takeo Kawabata
-
Article
| Open Access Collective synthesis of acetylenic pharmaceuticals via enantioselective Nickel/Lewis acid-catalyzed propargylic alkylation
Collective synthesis of acetylenic pharmaceuticals via enantioselective Nickel/Lewis acid-catalyzed propargylic alkylation
Chiral acetylenic derivatives are found in many bioactive compounds and are versatile building blocks in organic synthesis. Here, the authors report a mild enantioselective nickel/Lewis acid-catalyzed asymmetric propargylic substitution reaction which is applied to the collective synthesis of seven biologically active compounds.
- Xihao Chang
- , Jiayin Zhang
- & Chang Guo
-
Article
| Open Access Biomimetic selenocystine based dynamic combinatorial chemistry for thiol-disulfide exchange
Biomimetic selenocystine based dynamic combinatorial chemistry for thiol-disulfide exchange
Thiol-disulfide exchange is an extensively used reversible reaction in dynamic combinatorial chemistry, but usually requires long time to reach equilibrium. Here, the authors employ selenocystine as a catalyst of thiol-disulfide exchange at low temperatures and basic pH, and show that it can promote disulfide bond formation during folding of a scrambled RNase A.
- Andrea Canal-Martín
- & Ruth Pérez-Fernández
-
Article
| Open Access Enantioselective extraction of unprotected amino acids coupled with racemization
Enantioselective extraction of unprotected amino acids coupled with racemization
Dynamic kinetic resolution is a common approach for the preparation of optically pure amino acids using enzymes. Here the authors report an alternative method based on enantioselective extraction coupled with racemization, in which a bulky extractant affords hydrophobic extractable imines with amino acids rapidly, reversibly and enantioselectively.
- Haofei Huang
- , Yingji Jin
- & Kwan Mook Kim
-
Article
| Open Access Unified divergent strategy towards the total synthesis of the three sub-classes of hasubanan alkaloids
Unified divergent strategy towards the total synthesis of the three sub-classes of hasubanan alkaloids
Several Hasubanan alkaloids have been synthesized in the past decades, however a divergent approach to access the 3 subclasses of such natural products has not been reported yet. Here, the authors show the enantioselective total syntheses of four representative members via a unified strategy leading to the three topologically different classes of alkaloids.
- Guang Li
- , Qian Wang
- & Jieping Zhu
-
Article
| Open Access Bis-silylation of internal alkynes enabled by Ni(0) catalysis
Bis-silylation of internal alkynes enabled by Ni(0) catalysis
Bis-silylated alkenes offer the advantage of two functional handles with distinguished reactivity for downstream functionalization. Here, the authors report a nickel-catalyzed bis-silylation of internal alkynes to versatile silylated alkene intermediates which can be chemoselectively manipulated.
- Yun Zhang
- , Xi-Chao Wang
- & Dongbing Zhao
-
Article
| Open Access Anti-selective [3+2] (Hetero)annulation of non-conjugated alkenes via directed nucleopalladation
Anti-selective [3+2] (Hetero)annulation of non-conjugated alkenes via directed nucleopalladation
Dihydrobenzofurans and indolines are common substructures in medicines and natural products. Herein, the authors report a palladium-catalyzed [3 + 2] (hetero)annulation proceeding in an anti-selective fashion and enabling direct access to these valuable heterocycles with broad reaction scope.
- Hui-Qi Ni
- , Ilia Kevlishvili
- & Keary M. Engle
-
Article
| Open Access Nature-inspired remodeling of (aza)indoles to meta-aminoaryl nicotinates for late-stage conjugation of vitamin B3 to (hetero)arylamines
Nature-inspired remodeling of (aza)indoles to meta-aminoaryl nicotinates for late-stage conjugation of vitamin B3 to (hetero)arylamines
Vitamin B3 derivatives display a range of biological activities. Here, the authors report the synthesis of meta-aminoaryl nicotinates, derivatives of vitamin B3, and their late-stage conjugation with (hetero)arylamines, ultimately expanding the chemical space for biomedical research.
- Begur Vasanthkumar Varun
- , Kannan Vaithegi
- & Seung Bum Park
-
Article
| Open Access Reverse C-glycosidase reaction provides C-nucleotide building blocks of xenobiotic nucleic acids
Reverse C-glycosidase reaction provides C-nucleotide building blocks of xenobiotic nucleic acids
C-nucleosides are analogues of the canonical N-nucleosides and, despite their synthetic value, biocatalysis has not targeted them yet. Here, the authors report a pseudouridine monophosphate C-glycosidase enzyme for selective 5-β-C-glycosylation of uracil and its derivatives from pentose 5- phosphate substrates.
- Martin Pfeiffer
- & Bernd Nidetzky
-
Article
| Open Access Xenon binding by a tight yet adaptive chiral soft capsule
Xenon binding by a tight yet adaptive chiral soft capsule
Xenon binding carries potential for xenon separation and emerging applications in magnetic resonance imaging. Here, the authors report a rare example of a tight yet soft capsule, assembled from two chiral bisurea-bisthiourea macrocycle components, that can efficiently and adaptively bind xenon in both the solid state and solution.
- Shi-Xin Nie
- , Hao Guo
- & Qi-Qiang Wang
-
Article
| Open Access Benzene- and pyridine-incorporated octaphyrins with different coordination modes toward two PdII centers
Benzene- and pyridine-incorporated octaphyrins with different coordination modes toward two PdII centers
The reactivity of expanded porphyrins is highly influenced by their scaffold architecture. Here, the authors construct two nonaromatic dumbbell-shaped benzene- and pyridine-incorporating octaphyrins via Suzuki-Miyaura cross-coupling; depending on the framework, different coordination modes are obtained upon metallation with Pd(II).
- Le Liu
- , Zhiwen Hu
- & Jianxin Song
-
Article
| Open Access Visible-light photoredox-catalyzed C–O bond cleavage of diaryl ethers by acridinium photocatalysts at room temperature
Visible-light photoredox-catalyzed C–O bond cleavage of diaryl ethers by acridinium photocatalysts at room temperature
The high bond energies of C–O bonds in biomass and its derivatives make their cleavage very challenging. Here, the authors report a photoredox-catalyzed C–O bond cleavage in diaryl ethers by acidolysis with an aryl carboxylic acid followed by hydrolysis in a one-pot manner.
- Fang-Fang Tan
- , Xiao-Ya He
- & Yang Li
-
Article
| Open Access Empowering alcohols as carbonyl surrogates for Grignard-type reactions
Empowering alcohols as carbonyl surrogates for Grignard-type reactions
Alcohols are more naturally abundant than carbonyl compounds, which in turn are well known for their reactivity in Grignard reactions. Here, the authors showcase a distinct Grignard-like reactivity by using alcohols as coupling partners with hydrazones and synthesize more complex alcohols under ruthenium catalysis.
- Chen-Chen Li
- , Haining Wang
- & Chao-Jun Li
-
Article
| Open Access Vicinal difunctionalization of carbon–carbon double bond for the platform synthesis of trifluoroalkyl amines
Vicinal difunctionalization of carbon–carbon double bond for the platform synthesis of trifluoroalkyl amines
Metal-free olefin diamination may display challenges, especially when targeting fluorinated amines. Here, the authors report a double nucleophilic functionalization of an activated alkene originated from a trifluoropropenyliodonium salt with two nucleophiles for the selective synthesis of trifluoromethylated ethylene amines and diamines.
- Ferenc Béke
- , Ádám Mészáros
- & Zoltán Novák
-
Article
| Open Access Ni-catalyzed asymmetric hydrogenation of N-aryl imino esters for the efficient synthesis of chiral α-aryl glycines
Ni-catalyzed asymmetric hydrogenation of N-aryl imino esters for the efficient synthesis of chiral α-aryl glycines
Chiral α-amino acids find application in the fields of pharmaceutical, biological and synthetic chemistry. Here, the authors report a nickel-catalyzed asymmetric hydrogenation of N-aryl imino esters affording chiral α-aryl glycines in high yields and enantioselectivities.
- Dan Liu
- , Bowen Li
- & Wanbin Zhang
-
Article
| Open Access Alkyl halides as both hydride and alkyl sources in catalytic regioselective reductive olefin hydroalkylation
Alkyl halides as both hydride and alkyl sources in catalytic regioselective reductive olefin hydroalkylation
Methods that regioselectively install a functionalized alkyl unit at the 2-position of a terminal unactivated C=C bond are scarce. Here, the authors report a Markovnikov-selective hydroalkylation of unactivated amide-tethered alkenes catalyzed by nickel in conjunction with a stoichiometric reductant.
- Xianxiao Chen
- , Weidong Rao
- & Ming Joo Koh
-
Article
| Open Access Site-switchable mono-O-allylation of polyols
Site-switchable mono-O-allylation of polyols
Selective manipulation of one functional group, out of many repeatedly occurring in a substrate, represents a grand challenge in chemistry. Here, the authors report a Pd/Lewis acid cocatalytic system that achieves not only site-selective, but also site-switchable mono-O-allylation of polyols.
- Hua Tang
- , Yu-Biao Tian
- & Dawen Niu
-
Article
| Open Access Sequentially amplified circularly polarized ultraviolet luminescence for enantioselective photopolymerization
Sequentially amplified circularly polarized ultraviolet luminescence for enantioselective photopolymerization
Chiral functional materials with circularly polarized luminescence can be used in various applications but rarely reported. Here the authors show, a complex system, which show intense circularly polarized ultraviolet luminescence with large glum value, enabling a chiral UV light triggered enantioselective polymerization.
- Dongxue Han
- , Xuefeng Yang
- & Pengfei Duan
-
Article
| Open Access Quadruple C-H activation coupled to hydrofunctionalization and C-H silylation/borylation enabled by weakly coordinated palladium catalyst
Quadruple C-H activation coupled to hydrofunctionalization and C-H silylation/borylation enabled by weakly coordinated palladium catalyst
Combining the Heck reaction with other transformations provides a powerful strategy to access diverse, complex compounds. Here, the authors report a weak coordination dominated Pd(0)-catalyzed quadruple C-H activation followed by hydro-functionalization, C-H silylation, and C-H borylation.
- Bo-Cheng Tang
- , Wen-Xuan Lin
- & An-Xin Wu
-
Article
| Open Access Skeletal reorganization divergence of N-sulfonyl ynamides
Skeletal reorganization divergence of N-sulfonyl ynamides
Skeletal reorganizations are intriguing processes in chemical synthesis due to their mechanism, atom-economy and synthetic versatility. Herein, the authors describe a divergent skeletal reorganization of N-sulfonyl ynamides to thiete sulfones and propargyl sulfonamides.
- Linwei Zeng
- , Yuxin Lin
- & Sunliang Cui
-
Article
| Open Access State-of-the-art augmented NLP transformer models for direct and single-step retrosynthesis
State-of-the-art augmented NLP transformer models for direct and single-step retrosynthesis
Development of algorithms to predict reactant and reagents given a target molecule is key to accelerate retrosynthesis approaches. Here the authors demonstrate that applying augmentation techniques to the SMILE representation of target data significantly improves the quality of the reaction predictions.
- Igor V. Tetko
- , Pavel Karpov
- & Guillaume Godin
-
Article
| Open Access Chiral phosphoric acid-catalyzed stereodivergent synthesis of trisubstituted allenes and computational mechanistic studies
Chiral phosphoric acid-catalyzed stereodivergent synthesis of trisubstituted allenes and computational mechanistic studies
Despite of the high demand of chiral allenes, their asymmetric synthesis remains a challenge for organic chemists. Here, the authors report a stereodivergent synthesis of trisubstituted allenes via asymmetric additions of oxazolones to activated 1,3-enynes enabled by modification of chiral phosphoric acid catalysts.
- Jiawen Wang
- , Sujuan Zheng
- & Xiaoyu Yang
-
Article
| Open Access Copper(I)-catalyzed asymmetric 1,6-conjugate allylation
Copper(I)-catalyzed asymmetric 1,6-conjugate allylation
Catalytic asymmetric conjugate allylation of unsaturated carbonyl compounds is usually difficult to achieve due to competing 1,2-addition. Here, the authors report a copper(I)-catalyzed asymmetric 1,6-conjugate allylation of 2,2-dimethyl-6-alkenyl-4H-1,3-dioxin-4-ones with good scope and high enantioselectivity.
- Chang-Yun Shi
- , Zhi-Zhou Pan
- & Liang Yin
-
Article
| Open Access The role of allyl ammonium salts in palladium-catalyzed cascade reactions towards the synthesis of spiro-fused heterocycles
The role of allyl ammonium salts in palladium-catalyzed cascade reactions towards the synthesis of spiro-fused heterocycles
Synthetic methods aiming at minimizing reaction steps while increasing molecular complexity are highly sought after by organic chemists. Here, the authors report two cascade procedures combining nucleophilic substitution, palladium-catalyzed Heck and C–H activation reactions for the synthesis of spiro-fused heterocycles.
- Fei Ye
- , Yao Ge
- & Matthias Beller
-
Article
| Open Access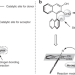 Diastereodivergent chiral aldehyde catalysis for asymmetric 1,6-conjugated addition and Mannich reactions
Diastereodivergent chiral aldehyde catalysis for asymmetric 1,6-conjugated addition and Mannich reactions
Chiral aldehyde catalysis is a useful strategy in the catalytic asymmetric α-functionalization of amino methyl compounds but these reaction types are limited. Here, the authors report two chiral aldehyde-catalysed diastereodivergent reactions, namely, a 1,6-conjugate addition and a bio-inspired Mannich reaction.
- Wei Wen
- , Ming-Jing Luo
- & Qi-Xiang Guo
-
Article
| Open Access Decarboxylative thiolation of redox-active esters to free thiols and further diversification
Decarboxylative thiolation of redox-active esters to free thiols and further diversification
Thiols are important precursors for the synthesis of a variety of pharmaceutically important sulfur-containing compounds. Here, the authors report a visible light-mediated decarboxylative thiolation of alkyl redox-active esters to free thiols and the in situ product diversification of a number of thiol derivatives.
- Tianpeng Cao
- , Tianxiao Xu
- & Saihu Liao
-
Article
| Open Access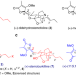 Enantioselective total syntheses of (+)-stemofoline and three congeners based on a biogenetic hypothesis
Enantioselective total syntheses of (+)-stemofoline and three congeners based on a biogenetic hypothesis
Stemofoline alkaloids are promising lead structures for further development in the fields of agriculture and medicine. Here, the authors report the enantioselective total syntheses of four stemofoline alkaloids in 19 steps based on a biogenetic hypothesis.
- Xiong-Zhi Huang
- , Long-Hui Gao
- & Pei-Qiang Huang
-
Article
| Open Access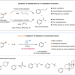 Direct C(sp2)–H alkylation of unactivated arenes enabled by photoinduced Pd catalysis
Direct C(sp2)–H alkylation of unactivated arenes enabled by photoinduced Pd catalysis
Direct catalytic C(sp2)–H alkylation of unactivated arenes with alkyl halides remains elusive despite the progress in C-H functionalization. Here, the authors report the catalytic C(sp2)–H alkylation of unactivated arenes with alkyl bromides via visible-light induced Pd catalysis.
- Daeun Kim
- , Geun Seok Lee
- & Soon Hyeok Hong

 Dual Ni/photoredox-catalyzed asymmetric cross-coupling to access chiral benzylic boronic esters
Dual Ni/photoredox-catalyzed asymmetric cross-coupling to access chiral benzylic boronic esters