Abstract
Thiols are important precursors for the synthesis of a variety of pharmaceutically important sulfur-containing compounds. In view of the versatile reactivity of free thiols, here we report the development of a visible light-mediated direct decarboxylative thiolation reaction of alkyl redox-active esters to free thiols based on the abundant carboxylic acid feedstock. This transformation is applicable to various carboxylic acids, including primary, secondary, and tertiary acids as well as natural products and drugs, forging a general and facile access to free thiols with diverse structures. Moreover, the direct access to free thiols affords an advantage of rapid in situ diversification with high efficiency to other important thiol derivatives such as sulfide, disulfide, thiocyanide, thioselenide, etc.
Similar content being viewed by others
Introduction
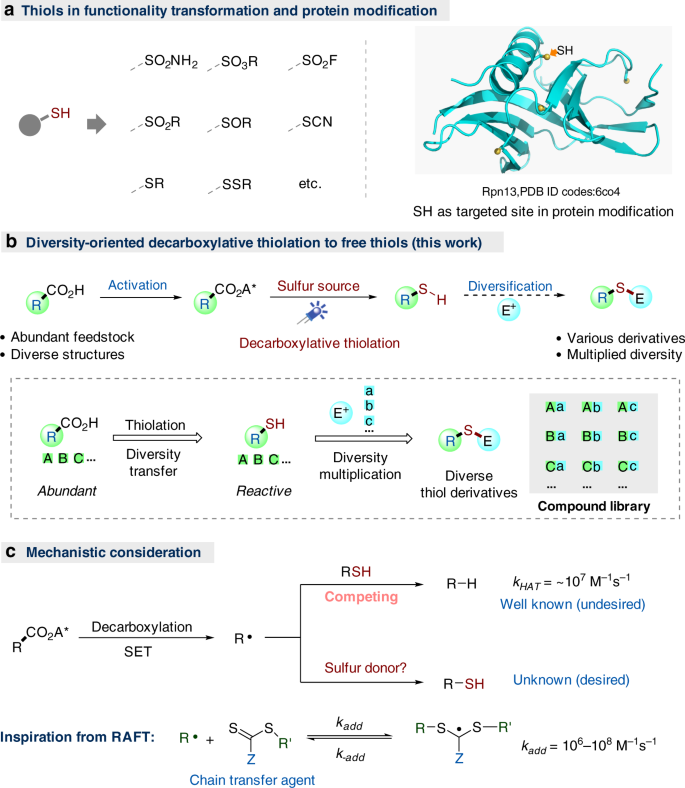
The construction of molecule libraries with structural and functional diversity is crucial for the study in the context of chemical biology and drug discovery1,2,3. Thiols are important precursors for the synthesis of a variety of pharmaceutically important sulfur-containing compounds, including sulfonamides, sulfonyl fluorides, sulfoxides, sulfides, disulfides, and so on, by virtue of their high reactivity and valence labile nature, and widely employed in organic synthesis, polymer preparation, materials science, and biomedicine4,5,6,7,8,9,10,11,12,13,14,15,16,17. In fact, besides their well-known roles in protein structure stabilizations18,19 and many enzymatic processes20, thiol is also one of the most targeted sites in post-translational protein modification (Fig. 1a)21,22,23. Inspired by the versatile reactivity of thiols, we conceived that, based on the feedstock of abundant carboxylic acid, a decarboxylative thiolation of acid-derived redox-active esters (RAEs)24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39 (RCO2A*) to free thiols could forge a novel access to various thiols and related derivatives with considerable structural diversity. In particular, the decarboxylative access to free thiols could allow a further diversification to other sulfur-containing compounds40,41,42 with a multiplied diversity by varying the coupling agents (e.g., with various electrophiles E+, Fig. 1b).
A number of radical C–S bond formation reactions43,44,45,46,47,48,49 have been reported, including the related decarboxylative transformations pioneered by Barton in 1980s46,47,48,49, but a direct radical thiolation to free thiols remains elusive so far. The challenges for the proposed radical decarboxylative thiolation to free thiols probably lie in the labile nature of free thiols, which can lead to dimerization, undesired hydrogen transfer, and other side reactions43. In fact, free thiols are commonly used as hydrogen atom transfer (HAT) catalysts or reagents in radical chemistry50,51,52,53,54, and the HAT from a primary alkyl thiol to alkyl radicals is a fast process (ca. 107 M−1s−1)52,53,54. Therefore, in the decarboxylative thiolation process, the desired thiol product (RSH) formed earlier may intercept the newly generated alkyl radicals (R∙), thus leading to the undesired alkane (R-H) formation (Fig. 1c). Nevertheless, in radical polymerization, the chain-transfer agents (CTA) employed in reversible addition-fragmentation chain-transfer polymerization can readily alter the radical addition rate by adjusting the Z group and increase kadd to above 108 M−1 s−1 (Fig. 1, C, below)55,56, which inspired us to focus on the sulfur donor search in the beginning. Herein, we report our efforts in the successful identification of aryl thioamides as an effective sulfur donor, and the invention of visible light-mediated direct decarboxylative thiolation of alkyl RAEs to free thiols. Moreover, further diversification to other thiol derivatives, such as sulfide, disulfide, thiocyanide, and thioselenide via in situ trapping, is also demonstrated.
Results
Reaction optimization
We commenced our study with the search for sulfur donors suitable for the radical thiolation reaction, by employing dihydrocinnamic acid-derived RAE (1) as the model substrate and Eosin Y-Na2/diisopropylethylamine (DIPEA) as the photoredox catalytic system (Table 1). Initially, thiourea 2a, which is frequently used as a sulfur donor in the nucleophilic substitution reactions of alkyl halides57,58, was examined first in the reaction, but only the alkane product 3′ was observed (entry 1), indicating the radical reactivity is substantially different from the polar substitution reactions. Other thioureas like 2b and 2c were also examined, but neither of them afforded the desired thiol product (entries 2 and 3). We then turned our attention to other types of sulfur donor (for more details about the reaction development, please see the Supplementary Figs. 1–9 and Table 1). To our delight, benzothioamide was found being a promising sulfur donor for this decarboxylative thiolation reaction, and the desired thiol 3 could be obtained as the predominant product in 77% yield (entry 4). We then carried out several modifications on the phenyl group of benzothioamides (entries 4–6). Electron-withdrawing group (-CF3, 2e) led to a decreased yield of 35%, while the introduction of an electron-donating group (2f) was found beneficial and further increased the yield to 81% (entry 5 vs. entry 6). The N-H group proved to be crucial for this transformation. Replacement with either one or two methyl groups (2g and 2h), both resulted in a sharp drop in yield (entries 7 and 8). Moreover, sulfur powder was also tested, but no desired thiol product was observed (entry 9). With 2f as the sulfur donor, we conducted a further reaction optimization, including
photocatalyst, solvent, light source, and so on (for details, please see the Supplementary Tables 2 and 3). Other photocatalysts, such as Ru(bpy)3Cl2·6H2O and Ir(ppy)3, gave lower yields (entries 10–13), while Eosin Y was found equally efficient (entry 14). Running the reaction in CH3CN could slightly enhanced the selectivity (entry 15). Without light or photocatalyst, no reaction or a low yield was observed (entries 16 and 17). To our delight, the employment of two equivalents of sulfur donor 2f could further suppress the undesired alkane formation and increase the yield of the desired thiol product to a decent level of 88% in the end (entry 18).
Substrate scope
With the optimized reaction conditions in hand, we next examined the reaction scope with a variety of primary, secondary, and tertiary acid-derived RAEs (Fig. 2). Some free thiols are volatile and thus isolated in their disulfide form by in situ trapping with diphenyl disulfide. These results are also included in Fig. 2. In cases of primary acids (3–18), we could see a good functional group tolerance. Br, Cl, ether, ester, and also a triple C–C bond are all compatible in the reaction, and the desired free thiols (4–10) or disulfides (11–18) could be isolated in moderate to good yields. Through this decarboxylative thiolation, simple propionic acid can also be converted to the corresponding disulfide in 40% yield (14). The reaction can be well applied to cyclic carboxylic acids (19–30), and different ring sizes (22–26), including cyclopropane (22), cyclobutane (23), benzocyclopentane (21), and cycloheptane (26), all can be converted to the desired product in reasonable yields. Heterocyclic free thiols or disulfides, such as 19, 20, and 30, are also accessible from the corresponding carboxylic acids. Pleasingly, tertiary thiols or disulfide (31–36) can be prepared as well with this method. In particular, this visible light-mediated radical decarboxylative thiolation reaction could afford a facile access to tertiary bridgehead thiols (such as bicyclic thiol 31, 1-adamantanethiol 33 and 34) under mild reaction conditions, which are often difficult to prepare via traditional nucleophilic substitution reactions due to the steric shielding of the bridgehead position that prevents the backside attack of the nucleophiles59. The reported synthesis of 33 from 1-adamantyl bromide or alcohol was performed under very harsh conditions (reflux in AcOH/conc. aq. HBr), and under the same conditions, only a trace amount of product was obtained in the synthesis of bicyclo[2.2.2]octane-1-thiol59,60. Moreover, the decarboxylative thiolation reaction can be well extended to natural occurring acids, such as oleic acid (39), aspartic acid (40), glutamic acid (41), dipeptide Glu-Pro (42), and nutriacholic acid (46). It is worth mentioning that the conversion of aspartic acid to the thiol product 40 is resembling a transformation of aspartic acid to cysteine via a residue manipulation. To our delight, this decarboxylative thiolation can be adopted for the late-stage modification of drugs, such as gemfibrozil (43), oxaprozin (44), and indometacin (45). In cases of 35, 36, and 43, we could observe less alkane formation when the reactions were performed in CF3CH2OH instead of CH3CN, and thus led to a better yield. As outlined in Fig. 2, under this reaction condition, primary, secondary, and tertiary carbon radicals can all be readily generated from the corresponding alkyl NHPI esters and subsequently trapped by the sulfur donor and converted to the desired thiol or disulfide products.
Product diversification
Disulfides are important motifs in life and biological active molecules, due to their unique pharmacological and physiochemical properties61,62,63,64,65,66. To further explore the scope of this transformation, more diaryl disulfide-trapping agents were examined with dihydrocinnamic acid-derived ester 1 as a model substrate. As shown in Fig. 3a, the reactions worked well with various diaryl disulfides processing different electronic nature, affording the desired unsymmetric alkyl aryl disulfides 47–51 readily in good yields. Importantly, the conversion of carboxylic acids to disulfides via this dicarboxylic thiolation/in situ trapping protocol provides a facile approach to synthesize this type of molecules with high structural diversity. Thiols are key precursors to many pharmaceutically important compounds4,5,6,7,8,9,10,11,12,13,14,15,16,17,40,41,42,43,44,45. The direct decarboxylative thiolation to free thiols allows for the establishment of a rapid, in situ diversification to various thiol derivatives without isolating the free thiols, which are often smelly and unstable. As shown in Fig. 3b, the 2-phenylethane-1-thiol can readily undergo alkylation in situ with various electrophiles or Michael acceptors, to provide the corresponding sulfide products (52–57). Moreover, trapping with 4-methylbenzenesulfonyl cyanide enable the conversion of carboxylic acid to thiocyanide 58. An access to thioselenide from the corresponding carboxylic acid via an in situ reaction with diphenyl diselenide was also demonstrated with the synthesis of thioselenide 59.
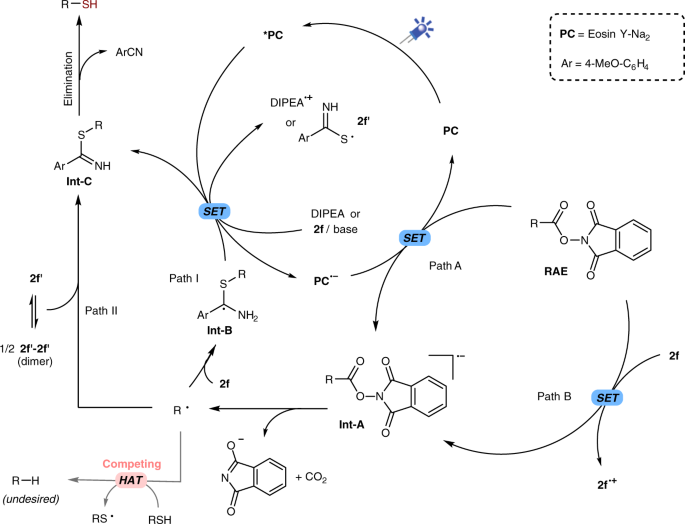
A possible reaction mechanism is proposed as outlined in Fig. 4. Under the irradiation of light, the photocatalyst Eosin Y (PC) is excited and subsequently reductively quenched by DIPEA or 2f, affording PC•− and 2f′ in the presence of base. The fluorescence quenching experiments also clearly showed that both DIPEA and sulfur donor 2f can quench the fluorescence of the photocatalyst (Supplementary Figs. 7–9). A single electron transfer (SET) from PC•− to the carboxylic acid-derived RAE afford the corresponding radical anion Int-A and concurrently regenerate the photocatalyst (Path A). As product also observed in the absence of PC and 2f showed substantial absorption in the blue light region (Supplementary Fig. 5), Path B might also involve to some extent. Int-A then undergoes fragmentation to give the alkyl radical R∙ via the N–O bond cleavage followed by the extrusion of CO2. The alkyl radical can be trapped with TEMPO (Supplementary Fig. 3). The addition of R∙ to the sulfur donor 2f generate the radical intermediate Int-B67, which can be oxidized to imine Int-C by a SET to excited photocatalyst as shown as Path I. The higher yields with aryl thioamides than alkyl ones may be ascribed to the stabilization of the aryl group to radical Int-B. The beneficial effect of the electron-donating group (2d–f, entries 4–5, Table 1) might result from a favored one electron oxidation of Int-B to Int-C. Alternatively, a radical coupling of R∙ and 2 f′ could also afford the intermediate Int-C (Path II). As a competing process, the HAT from thiol (R-SH) to R∙ will lead to the formation of the alkane side product (R-H)51,52, which can be suppressed by a rapid trapping of R∙ with active thiolating agents via Path I or II. In the end, the desired thiol is produced after the elimination of one molecule of ArCN68,69, which could be a relatively slow step, and a slow release of free thiols can decrease the formation of the undesired alkane product. The nitrile byproduct formation was confirmed by gas chromatography-mass spectrometry (GC-MS) analysis, and it can also be isolated by column chromatography. Therefore, the use of N-unsubstituted thioamides is crucial for this transformation. In contrast, N-substituted thioamide 2g and 2h are unable to form the corresponding N-H imine intermediate Int-C.
Discussion
In conclusion, a visible light-mediated direct decarboxylative thiolation of carboxylic acid-derived RAEs to free thiols has been developed. Aryl thioamides have been identified as an effective sulfur donor and crucial to this thiolation reaction. The transformation of abundant carboxylic acid feedstock to the corresponding free thiols and their further in situ diversification allows for a rapid and general access to various pharmaceutically important compounds, such as sulfide, disulfide, thiocyanide, and thioselenide, with diverse structures, which may be utilized for the molecule library construction and benefit the related study in chemical biology and discovery of novel, biologically interesting small molecules.
Methods
General procedure for decarboxylative thiolation
To an oven-dried 10-ml Schlenk tube equipped with a magnetic stir bar and a Teflon-coated septum screwcap was added the NHPI redox-active ester (0.2 mmol, 1.0 equiv.), 4-methoxythiobenzamide (2f, 0.4 mmol, 2.0 equiv.), Eosin Y-Na2 (2.5 mol%). The tube was evacuated and backfilled with argon for three cycles. The DIPEA (0.22 mmol, 1.1 equiv.) and dry CH3CN (2.0 ml) was added via a gastight syringe under argon atmosphere. Make sure the screwcap was closed, and the solvent was frozen by liquid nitrogen. Then, the screwcap was opened and the tube was evacuated for about 3 min. The screwcap was closed and let the solvent melts in a tepid water bath. Repeat above freeze-pump-thaw procedures for 3–5 times until you no longer see the evolution of gas as the solution thaws. The tube was filled with argon and sealed, irradiated with 6 W blue light-emitting diode (LED) reactor and stirred at ambient temperature for 24 h. Full experimental details (Supplementary Figs. 1–9 and Supplementary Tables 1–3) and characterization of new compounds (Supplementary Figs. 10–68) can be found in the Supplementary Methods section.
General procedure for the synthesis of disulfides
To an oven-dried 10-ml Schlenk tube equipped with a magnetic stir bar and a Teflon-coated septum screwcap was added the NHPI redox-active ester (0.2 mmol, 1.0 equiv.), 4-methoxythiobenzamide (2f, 0.4 mmol, 2.0 equiv.), and Eosin Y-Na2 (2.5 mol%). The tube was evacuated and backfilled with argon for three cycles. The DIPEA (0.22 mmol, 1.1 equiv.) and dry CH3CN (2.0 ml) was added via a gastight syringe under argon atmosphere. Make sure the screwcap was closed, and the solvent was frozen by liquid nitrogen. Then, the screwcap was opened and the tube was evacuated for ~3 min. The screwcap was then closed and the solvents were let to melt in a tepid water bath. Repeat above freeze-pump-thaw procedures for 3–5 times until you no longer see the evolution of gas as the solution thaws. The tube was filled with argon and sealed, irradiated with 6 W blue LED reactor and stirred at ambient temperature for 24 h. Then, the K2CO3 (0.4 mmol, 2.0 equiv.) and diaryl disulfide (0.4 mmol, 2.0 equiv.) was added under argon atmosphere. The tube stirred at ambient temperature for 6 h in the dark. Upon completion, the reaction mixture was carefully concentrated and the residue was further purified by flash chromatography to give the desired disulfide products. Full experimental details and characterization of new compounds can be found in the Supplementary Methods section.
Data availability
The authors declare that all data supporting the findings of this study are available within the article and Supplementary information files, and are also available from the corresponding author upon reasonable request.
References
Kennedy, J. P. et al. Application of combinatorial chemistry science on modern drug discovery. J. Comb. Chem. 10, 345–354 (2008).
Boström, J., Brown, D., Young, R. & Keserü, G. M. Expanding the medicinal chemistry synthetic toolbox. Nat. Rev. Drug Discov. 17, 709–727 (2018).
Li, G. & Lou, H.-X. Strategies to diversify natural products for drug discovery. Med. Res. Rev. 38, 1255–1294 (2018).
Dénès, F., Pichowicz, M., Povie, G. & Renaud, P. Thiyl radicals in organic synthesis. Chem. Rev. 114, 2587–2693 (2014).
Dunbar, K. L., Scharf, D. H., Litomska, A. & Hertweck, C. Enzymatic carbon–sulfur bond formation in natural product biosynthesis. Chem. Rev. 117, 5521–5577 (2017).
Franc, G. & Kakkar, A. K. “Click” methodologies: efficient, simple and greener routes to design dendrimers. Chem. Soc. Rev. 39, 1536–1544 (2010).
Hoyle, C. E. & Bowman, C. N. Thiol–ene click chemistry. Angew. Chem. Int. Ed. 49, 1540–1573 (2010).
Dreaden, E. C., Alkilany, A. M., Huang, X., Murphy, C. J. & EI-Sayed, M. A. The golden age: gold nanoparticles for biomedicine. Chem. Soc. Rev. 41, 2740–2779 (2012).
Wang, X. et al. Palladium-catalyzed enantioselective thiocarbonylation of styrenes. Angew. Chem. Int. Ed. 58, 12264–12270 (2019).
Ouhib, F. et al. A switchable domino process for the construction of novel CO2‐sourced sulfur‐containing building blocks and polymers. Angew. Chem. Int. Ed. 131, 11894–11899 (2019).
Laudadio, G. et al. Sulfonyl fluoride synthesis through electrochemical oxidative coupling of thiols and potassium fluoride. J. Am. Chem. Soc. 141, 11832–11836 (2019).
Kang, Y. S. et al. Ligand-promoted RhIII-catalyzed thiolation of benzamides with a broad disulfide scope. Angew. Chem. Int. Ed. 58, 9099–9103 (2019).
Liu, D., Ma, H.-X., Fang, P. & Mei, T.-S. Nickel-catalyzed thiolation of aryl halides and heteroaryl halides through electrochemistry. Angew. Chem. Int. Ed. 58, 5033–5037 (2019).
Yang, X.-H. et al. Catalytic hydrothiolation: counterion-controlled regioselectivity. J. Am. Chem. Soc. 141, 3006–3013 (2019).
Zhang, Y., Xu, X. & Zhu, S. Nickel-catalysed selective migratory hydrothiolation of alkenes and alkynes with thiols. Nat. Commun. 10, 1752 (2019).
McKinnell, R. M. et al. Discovery of TD-0212, an orally active dual pharmacology AT1 antagonist and neprilysin inhibitor (ARNI). ACS Med. Chem. Lett. 10, 86–91 (2019).
He, J. et al. Catalytic decarboxylative radical sulfonylation. Chem 6, 1149–1159 (2020).
Alegre-Cebollada, J., Kosuri, P., Rivas-Pardo, J. A. & Fernández, J. M. Direct observation of disulfide isomerization in a single protein. Nat. Chem. 3, 882–887 (2011).
Góngora-Benítez, M., Tulla-Puche, J. & Albericio, F. Multifaceted roles of disulfide bonds. Peptides as therapeutics. Chem. Rev. 114, 901–926 (2014).
Poole, L. B. The basics of thiols and cysteines in redox biology and chemistry. Free Radic. Biol. Med. 80, 148–157 (2015).
Spicer, C. D. & Davis, B. G. Selective chemical protein modification. Nat. Commun. 5, 4740 (2014).
Krall, N., da Cruz, F., Boutureira, O. & Bernardes, G. Site-selective protein-modification chemistry for basic biology and drug development. Nat. Chem. 8, 103–113 (2015).
deGruyter, J. N., Malins, L. R. & Baran, P. S. Residue-specific peptide modification: a chemist’s guide. Biochemistry 56, 3863–3873 (2017).
Xuan, J., Zhang, Z.-G. & Xiao, W.-J. Visible‐light‐induced decarboxylative functionalization of carboxylic acids and their derivatives. Angew. Chem. Int. Ed. 54, 15632–15641 (2015).
Huang, H., Jia, K. & Chen, Y. Radical decarboxylative functionalizations enabled by dual photoredox catalysis. ACS Catal. 6, 4983–4988 (2016).
Schwarz, J. & König, B. Decarboxylative reactions with and without light—a comparison. Green. Chem. 20, 323–361 (2018).
Murarka, S. N‐(acyloxy) phthalimides as redox‐active esters in cross‐coupling reactions. Adv. Synth. Catal. 360, 1735–1753 (2018).
Fawcett, A. et al. Photoinduced decarboxylative borylation of carboxylic acids. Science 357, 283–286 (2017).
Candish, L., Teders, M. & Glorius, F. Transition-metal-free, visible-light-enabled decarboxylative borylation of aryl N-hydroxyphthalimide esters. J. Am. Chem. Soc. 139, 7440–7443 (2017).
Wang, D., Zhu, N., Chen, P., Lin, Z. & Liu, G. Enantioselective decarboxylative cyanation employing cooperative photoredox catalysis and copper catalysis. J. Am. Chem. Soc. 139, 15632–15635 (2017).
Mao, R., Frey, A., Balon, J. & Hu, X. Decarboxylative C (sp3)–N cross-coupling via synergetic photoredox and copper catalysis. Nat. Catal. 1, 120–126 (2018).
Ma, J. et al. Synthesis of β-substituted γ-aminobutyric acid derivatives through enantioselective photoredox catalysis. Angew. Chem. Int. Ed. 57, 11193–11197 (2018).
Wang, C. et al. Visible‐light‐driven, copper‐catalyzed decarboxylative C (sp3)−H alkylation of glycine and peptides. Angew. Chem. Int. Ed. 57, 15841–15846 (2018).
Proctor, R. S. J., Davis, H. J. & Phipps, R. J. Catalytic enantioselective minisci-type addition to heteroarenes. Science 360, 419–422 (2018).
Fu, M.-C., Shang, R., Zhao, B., Wang, B. & Fu, Y. Photocatalytic decarboxylative alkylations mediated by triphenylphosphine and sodium iodide. Science 363, 1429–1434 (2019).
Qin, T. et al. A general alkyl-alkyl cross-coupling enabled by redox-active esters and alkylzinc reagents. Science 352, 801–805 (2016).
Huihui, K. M. M. et al. Decarboxylative cross-electrophile coupling of N‑hydroxyphthalimide esters with aryl iodides. J. Am. Chem. Soc. 138, 5016–5019 (2016).
Xue, W. & Oestreich, M. Copper-catalyzed decarboxylative radical silylation of redox-active aliphatic carboxylic acid derivatives. Angew. Chem. Int. Ed. 56, 11649–11652 (2017).
Liu, X. G. et al. Decarboxylative Negishi coupling of redox-active aliphatic esters by cobalt catalysis. Angew. Chem. Int. Ed. 57, 13096–13100 (2018).
Scott, K. A. & Njardarson, J. T. Analysis of US FDA-approved drugs containing sulfur atoms. Top. Curr. Chem. 376, 1–34 (2018).
Feng, M., Tang, B., Liang, S. & Jiang, X. Sulfur containing scaffolds in drugs: synthesis and application in medicinal chemistry. Curr. Top. Med. Chem. 16, 1200–1216 (2016).
Ilardi, E. A., Vitaku, E. & Njardarson, J. T. Data-mining for sulfur and fluorine: an evaluation of pharmaceuticals to reveal opportunities for drug design and discovery: miniperspective. J. Med. Chem. 57, 2832–2842 (2014).
Wimmer, A. & König, B. Photocatalytic formation of carbon–sulfur bonds. Beilstein J. Org. Chem. 14, 54–83 (2018).
Jin, Y., Yang, H. & Fu, H. An N-(acetoxy) phthalimide motif as a visible-light pro-photosensitizer in photoredox decarboxylative arylthiation. Chem. Commun. 52, 12909–12912 (2016).
Wei, L., Wu, C., Tung, C.-H., Wang, W. & Xu, Z. Decarboxylative sulfenylation of amino acids via metallaphotoredox catalysis. Org. Chem. Front. 6, 3224–3227 (2019).
Na, C. G., Ravelli, D. & Alexanian, E. J. Direct decarboxylative functionalization of carboxylic acids via O–H hydrogen atom transfer. J. Am. Chem. Soc. 142, 44–49 (2020).
Gianatassio, R. et al. Simple sulfinate synthesis enables C-H trifluoromethylcyclopropanation. Angew. Chem. Int. Ed. 53, 9851–9855 (2014).
Barton, D. H. R., Crich, D. & Motherwell, W. B. New and improved methods for the radical decarboxylation of acids. J. Chem. Soc. Chem. Commun. 939–941 https://pubs.rsc.org/en/content/articlelanding/1983/c3/c39830000939#!divAbstract (1983).
Barton, D. H. R., Lacher, B., Misterkiewicz, B. & Zard, S. Z. The invention of radical reactions: Part XVII. A decarboxylative sulphonylation of carboxylic acids. Tetrahedron 44, 1153–1158 (1988).
Guin, J., Fröhlich, R. & Studer, A. Thiol-catalyzed stereoselective transfer hydroamination of olefins with N-aminated dihydropyridines. Angew. Chem. Int. Ed. 47, 779–782 (2008).
Okada, K., Okamoto, K. & Oda, M. A new and practical method of decarboxylation: photosensitized decarboxylation of N-acyloxyphthalimides via electron-transfer mechanism. J. Am. Chem. Soc. 110, 8736–8738 (1988).
Newcomb, M., Glenn, A. G. & Manek, M. B. Rate constants and arrhenius functions for hydrogen atom transfer from tert-butyl thiol to primary alkyl radicals. J. Org. Chem. 54, 4603–4606 (1989).
Chauvin, J.-P. R., Griesser, M. & Pratt, D. A. Hydropersulfides: H-atom transfer agents par excellence. J. Am. Chem. Soc. 139, 6484–6493 (2017).
Poon, J.-F. & Pratt, D. A. Recent insights on hydrogen atom transfer in the inhibition of hydrocarbon autoxidation. Acc. Chem. Res. 51, 1996–2005 (2018).
Roth, P. J., Boyer, C., Lowe, A. B. & Davis, T. P. RAFT polymerization and thiol chemistry: a complementary pairing for implementing modern macromolecular design. Macromol. Rapid Commun. 32, 1123–1143 (2011).
Moad, G., Rizzardo, E. & Thang, S. H. Radical addition–fragmentation chemistry in. Polym. Synth. Polym. 49, 1079–1131 (2008).
Cossar, B. C., Fournier, J. O., Fields, D. L. & Reynolds, D. D. Preparation of thiols. J. Org. Chem. 27, 93–95 (1962).
Wardell, J. L. In The Chemistry of the Thiol Group (ed. Patai, S.), Part 1, 179–211 (Wiley, New York, 1974).
Tkachenko, B. A. et al. Functionalized nanodiamonds part 3: thiolation of tertiary/bridgehead alcohols. Org. Lett. 8, 1767–1770 (2006).
Moya-López, J. F. et al. Studies on the diastereoselective oxidation of 1-thio-β-d-glucopyranosides: synthesis of the usually less favoured RS sulfoxide as a single diastereoisomer. Org. Biomol. Chem. 13, 1904–1914 (2015).
Musiejuka, M. & Witt, D. Recent developments in the synthesis of unsymmetrical disulfanes (disulfides). A review. Org. Prep. Proc. Int. 47, 95–131 (2015).
Xiao, X., Feng, M. & Jiang, X. New design of a disulfurating reagent: facile and straightforward pathway to unsymmetrical disulfanes by copper‐catalyzed oxidative cross‐coupling. Angew. Chem. Int. Ed. 55, 14121–14125 (2016).
Xiao, X., Xue, J. & Jiang, X. Polysulfurating reagent design for unsymmetrical polysulfide construction. Nat. Commun. 9, 2191 (2018).
Wang, W., Lin, Y., Ma, Y., Tung, C.-H. & Xu, Z. Cu-catalyzed electrophilic disulfur transfer: synthesis of unsymmetrical disulfides. Org. Lett. 20, 3829–3832 (2018).
Huang, P., Wang, P., Tang, S., Fu, Z. & Lei, A. Electro‐oxidative S−H/S−H cross‐coupling with hydrogen evolution: facile access to unsymmetrical disulfides. Angew. Chem. Int. Ed. 57, 8115–8119 (2018).
Chauvin, J.-P. R., Griesser, M. & Pratt, D. A. The antioxidant activity of polysulfides: it’s radical! Chem. Sci. 10, 4999–5010 (2019).
Smith, R. A., Fu, G., McAteer, O., Xu, M. & Gutekunst, W. R. Radical approach to thioester containing polymers. J. Am. Chem. Soc. 141, 1446–1451 (2019).
Khodade, V. S. & Toscano, J. P. Development of S-substituted thioisothioureas as efficient hydropersulfide precursors. J. Am. Chem. Soc. 140, 17333–17337 (2018).
Rad, M. N. S. & Maghsoudi, S. Two-step three-component process for one-pot synthesis of 8-alkylmercaptocaffeine derivatives. RSC Adv. 6, 70335–70342 (2016).
Acknowledgements
We gratefully acknowledge National Natural Science Foundation of China (No. 21602028), the Recruitment Program of Global Experts, and Fuzhou University for the financial support.
Author information
Authors and Affiliations
Contributions
T.C. and T.X. contributed equally to this work. T.C. developed the reactions, and contributed to the reaction scope investigation, mechanistic study, and product derivatization. T.X. carried out most substrate synthesis, and contributed to the study of reaction scope, product diversification, and reaction mechanism. R.X. and X.S. participated in the synthesis of substrates. S.L. conceived this concept and prepared this manuscript with feedback from T.C. and T.X.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Communications thanks Hua Wang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Cao, T., Xu, T., Xu, R. et al. Decarboxylative thiolation of redox-active esters to free thiols and further diversification. Nat Commun 11, 5340 (2020). https://doi.org/10.1038/s41467-020-19195-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-020-19195-w
This article is cited by
-
Photocatalytic divergent decarboxylative amination: a metal-free access to aliphatic amines and hydrazines
Science China Chemistry (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.