Featured
-
-
Article
| Open Access Rehybridization dynamics into the pericyclic minimum of an electrocyclic reaction imaged in real-time
Rehybridization dynamics into the pericyclic minimum of an electrocyclic reaction imaged in real-time
Electrocyclic reactions proceed through critical geometries, which are known as pericyclic transition states in thermal reactions and pericyclic minima in photochemical reactions. Here, the authors image the structure of a pericyclic minimum in real time using a combination of ultrafast electron diffraction and ab initio dynamics simulations.
- Y. Liu
- , D. M. Sanchez
- & T. J. A. Wolf
-
Article
| Open Access Flattened 1D fragments of fullerene C60 that exhibit robustness toward multi-electron reduction
Flattened 1D fragments of fullerene C60 that exhibit robustness toward multi-electron reduction
Fullerenes are compelling molecular materials, but the reason for their high robustness to multi-electron reduction is still debated. Here, the authors synthesize flattened 1D fragments of fullerene C60 with high robustness against multi-electron reduction.
- Masahiro Hayakawa
- , Naoyuki Sunayama
- & Aiko Fukazawa
-
Article
| Open Access Computational study on the catalytic control of endo/exo Diels-Alder reactions by cavity quantum vacuum fluctuations
Computational study on the catalytic control of endo/exo Diels-Alder reactions by cavity quantum vacuum fluctuations
Strong light-matter interactions are a pathway to chemical control at the molecular level. Here, authors theoretically show that an optical cavity allows to control catalysis, inhibition, and endo/exo stereoselectivity in Diels-Alder reactions.
- Fabijan Pavošević
- , Robert L. Smith
- & Angel Rubio
-
Article
| Open Access Switching imidazole reactivity by dynamic control of tautomer state in an allosteric foldamer
Switching imidazole reactivity by dynamic control of tautomer state in an allosteric foldamer
Molecular biology achieves control over complex reaction networks by means of molecular systems that translate a chemical input into an orthogonal chemical output but the application of dynamic foldamers has been limited by the restriction to stereochemical changes that produce spectroscopic signals. Here, the authors present an artificial molecular translation device that converts a chemical input into an unrelated chemical output.
- David P. Tilly
- , Jean-Paul Heeb
- & Jonathan Clayden
-
Article
| Open Access Ni-catalyzed mild hydrogenolysis and oxidations of C–O bonds via carbonate redox tags
Ni-catalyzed mild hydrogenolysis and oxidations of C–O bonds via carbonate redox tags
Catalytic redox transformations of C–O bonds are pertinent to the valorization of oxygenated hydrocarbons, but typically rely on hazardous redox agents which bring about numerous drawbacks ranging from safety hazards to waste management. Here, the authors demonstrate that such reagents can be rendered redundant by catalytically harnessing internal C–H bonds as redox equivalents.
- Georgios Toupalas
- , Loélie Ribadeau-Dumas
- & Bill Morandi
-
Article
| Open Access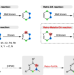 C(alkyl)–C(vinyl) bond cleavage enabled by Retro-Pallada-Diels-Alder reaction
C(alkyl)–C(vinyl) bond cleavage enabled by Retro-Pallada-Diels-Alder reaction
Inert C–C bond cleavage remains a challenge in organic chemistry. Herein, the authors report a retro-Diels-Alder process via palladium catalysis to achieve selective C(alkyl)–C(vinyl) bond cleavage.
- Qingyang Zhao
- , Le Yu
- & Zheng-Hui Guan
-
Article
| Open Access Direct nucleophilic and electrophilic activation of alcohols using a unified boron-based organocatalyst scaffold
Direct nucleophilic and electrophilic activation of alcohols using a unified boron-based organocatalyst scaffold
Boronic acids are promising catalysts for the direct functionalization of alcohols without requiring precious metals. Here, the authors report an easily synthesized class of cyclic hemiboronic acid catalysts which are applicable in both nucleophilic and electrophilic modes of alcohol activation.
- Jason P. G. Rygus
- & Dennis G. Hall
-
Article
| Open Access Photocatalyzed regioselective hydrosilylation for the divergent synthesis of geminal and vicinal borosilanes
Photocatalyzed regioselective hydrosilylation for the divergent synthesis of geminal and vicinal borosilanes
Geminal and vicinal borosilanes are useful building blocks in synthetic chemistry. But in contrast to the well-established transition-metal catalyzed methods, radical approaches are rarely explored. Here, the authors report the synthesis of geminal borosilanes from α-selective hydrosilylation of alkenyl boronates via photoinduced hydrogen atom transfer (HAT) catalysis
- Degong Kong
- , Muliang Zhang
- & Jie Wu
-
Article
| Open Access Allylic hydroxylation of enones useful for the functionalization of relevant drugs and natural products
Allylic hydroxylation of enones useful for the functionalization of relevant drugs and natural products
Enones are privileged structural motifs in bioactive natural products and pharmaceuticals, but the γ-hydroxylation of enones is challenging. Here, the authors report on a mild and efficient method for the direct C(sp3)–H hydroxylation of enones via visible-light-induced hydrogen-atom transfer, without the need for a metal and peroxide.
- Cheng-Yu Zheng
- & Jian-Min Yue
-
Article
| Open Access Nickel/biimidazole-catalyzed electrochemical enantioselective reductive cross-coupling of aryl aziridines with aryl iodides
Nickel/biimidazole-catalyzed electrochemical enantioselective reductive cross-coupling of aryl aziridines with aryl iodides
Metal reductants show low efficiency in enantioselective nickel-catalyzed reductive cross-coupling of aryl aziridines with aryl iodides. Here, the authors improve the reaction efficiency through an electrochemical method.
- Yun-Zhao Wang
- , Zhen-Hua Wang
- & Tian-Sheng Mei
-
Article
| Open Access Design of C1-symmetric tridentate ligands for enantioselective dearomative [3 + 2] annulation of indoles with aminocyclopropanes
Design of C1-symmetric tridentate ligands for enantioselective dearomative [3 + 2] annulation of indoles with aminocyclopropanes
Polycyclic indolines are valuable skeletons in drug discovery. Here, the authors report an asymmetric dearomative [3 + 2] annulation of indoles with aminocyclopropanes to construct tricyclic indolines.
- Hai-Xia Wang
- , Chun Yang
- & Hai-Ming Guo
-
Article
| Open Access Copper(I)-catalyzed asymmetric alkylation of α-imino-esters
Copper(I)-catalyzed asymmetric alkylation of α-imino-esters
Asymmetric alkylation of enolates is an important method for the formation of α-stereogenic carbonyls but generally requires the introduction and then removal of chiral auxiliaries. Here, the authors report a copper-catalyzed asymmetric alkylation of α-amino esters.
- Zong-Ci Liu
- , Zi-Qing Wang
- & Liang Yin
-
Article
| Open Access Asymmetric hydrogenation of 1,1-diarylethylenes and benzophenones through a relay strategy
Asymmetric hydrogenation of 1,1-diarylethylenes and benzophenones through a relay strategy
Distinguishing the prochiral faces of a double bond can be challenging for hydrogenation catalysts. Here, authors show a relay strategy for constructing compounds with a chiral gem-diaryl carbon center via selective arene exchange between 1,1-diarylethylenes or benzophenones with (naphthalene)Cr(CO)3 and subsequent asymmetric hydrogenation.
- Ke Li
- , Wen-Qiang Wu
- & Hang Shi
-
Article
| Open Access Stereodefined polymetalloid alkenes synthesis via stereoselective boron-masking of polyborylated alkenes
Stereodefined polymetalloid alkenes synthesis via stereoselective boron-masking of polyborylated alkenes
Polyborylated-containing alkenes, hold promise as polymetalated building blocks for a wide variety of organic motifs, biologically-active compounds, and natural product synthesis, though strategies for their stereoselective synthesis are lacking. Here, the authors report concise, highly site-selective, and stereoselective boron-masking strategies for polyborylated alkenes.
- Nadim Eghbarieh
- , Nicole Hanania
- & Ahmad Masarwa
-
Article
| Open Access An α-chloroaldehyde-based formal synthesis of eribulin
An α-chloroaldehyde-based formal synthesis of eribulin
Eribulin is an anticancer drug and the most structurally complex non-peptidic drug made by chemical synthesis. Here, the authors report a streamlined synthesis of eribulin that creates new opportunities for drug discovery and development.
- Anissa Kaghad
- , Dimitrios Panagopoulos
- & Robert Britton
-
Article
| Open Access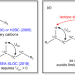 Unequivocal identification of two-bond heteronuclear correlations in natural products at nanomole scale by i-HMBC
Unequivocal identification of two-bond heteronuclear correlations in natural products at nanomole scale by i-HMBC
The inability to distinguish two-bond from long-range HMBC correlation has historically impeded NMR structure elucidation. Here the authors developed a practical iHMBC methodology utilizing accurate isotope shift measurement to overcome this limitation.
- Yunyi Wang
- , Aili Fan
- & Xiao Wang
-
Article
| Open Access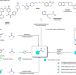 Transition-metal-free silylboronate-mediated cross-couplings of organic fluorides with amines
Transition-metal-free silylboronate-mediated cross-couplings of organic fluorides with amines
C–N bond cross-coupling is fundamental in organic chemistry, but the readily available organic fluorides are unsuitable as coupling substrates. Here, we realize defluorinative cross-coupling with amines via a silylboronate-mediated strategy.
- Jun Zhou
- , Zhengyu Zhao
- & Norio Shibata
-
Article
| Open Access Mn(I)-catalyzed sigmatropic rearrangement of β, γ-unsaturated alcohols
Mn(I)-catalyzed sigmatropic rearrangement of β, γ-unsaturated alcohols
Molecular rearrangements are ubiquitous in modern synthetic chemistry, providing a powerful strategy to arrive at complex structures in an atom- and step-economic process. Here, the authors disclose a Mn(I)- catalyzed sigmatropic rearrangement of β, γ-unsaturated alcohols via C-C σ-bond activation to site-selectively reorganize carbon skeletons.
- Can Yang
- , Xiaoyu Zhou
- & Wei Zeng
-
Article
| Open Access 3-sulfonyloxyaryl(mesityl)iodonium triflates as 1,2-benzdiyne precursors with activation via ortho-deprotonative elimination strategy
3-sulfonyloxyaryl(mesityl)iodonium triflates as 1,2-benzdiyne precursors with activation via ortho-deprotonative elimination strategy
Preparation of functionalized analogues of 1,2-benzdiyne, from heavily substituted as well as benzofused scaffolds, is challenging. Here, the authors develop an array of 3-sulfonyloxyaryl(mesityl)iodonium triflates as 1,2-benzdiyne precursors, triggered from ortho-deprotonative elimination.
- Haoyin Yuan
- , Wenhao Yin
- & Yang Li
-
Article
| Open Access Modular and practical diamination of allenes
Modular and practical diamination of allenes
Diamination of allenes with two different acyclic and cyclic alkyl amines remains a daunting challenge. Here, the authors report a modular and practical diamination of allenes, which offers efficient syntheses of β,γ-diamino carboxylates and sulfones.
- Jian-Jun Dai
- , Xianglin Yin
- & Mingji Dai
-
Article
| Open Access Screw sense excess and reversals of helical polymers in solution
Screw sense excess and reversals of helical polymers in solution
Reversals are structural motifs found in helical polymers that are difficult to determine experimentally. Here, the authors turn to photochemical electrocyclization as a tool to establish their presence and estimate excess screw sense in solution.
- Francisco Rey-Tarrío
- , Rafael Rodríguez
- & Félix Freire
-
Article
| Open Access Modulation of metal species as control point for Ni-catalyzed stereodivergent semihydrogenation of alkynes with water
Modulation of metal species as control point for Ni-catalyzed stereodivergent semihydrogenation of alkynes with water
Generally, transition metal-catalyzed stereodivergent transfer semihydrogenation of alkynes realized regulation of stereoselectivity by a Z to E isomerization process. Here, authors report a Ni-catalyzed stereodivergent transfer semihydrogenation of alkynes with a base-assisted metal species modulation mechanism, delivering both olefinic isomers smoothly in orthogonal catalytic pathways using cheap and nontoxic catalysts and additives
- Yuanqi Wu
- , Yuhui Ao
- & Yu Liu
-
Article
| Open Access Electrochemical oxidative difunctionalization of diazo compounds with two different nucleophiles
Electrochemical oxidative difunctionalization of diazo compounds with two different nucleophiles
The difunctionalization of diazo compounds, traditionally with one nucleophile and one electrophile, is a powerful strategy. Here, the authors develop a path of electrochemical oxidative difunctionalization of diazo compounds with two different nucleophiles.
- Dongfeng Yang
- , Zhipeng Guan
- & Aiwen Lei
-
Article
| Open Access Poly(heptazine imide) ligand exchange enables remarkable low catalyst loadings in heterogeneous metallaphotocatalysis
Poly(heptazine imide) ligand exchange enables remarkable low catalyst loadings in heterogeneous metallaphotocatalysis
The development of heterogeneous metallaphotocatalysis is of great interest for sustainable organic synthesis. Herein, the authors demonstrate the incorporation of privileged homogeneous bipyridyl-based Ni-catalysts into highly ordered and crystalline potassium poly(heptazine imide) (K-PHI).
- Liuzhuang Xing
- , Qian Yang
- & Yunfei Cai
-
Article
| Open Access Stereoselective C–O silylation and stannylation of alkenyl acetates
Stereoselective C–O silylation and stannylation of alkenyl acetates
Carbon-heteroatom bond formation reactions are of high interest in synthetic organic chemistry. Here, the authors report the cobalt-catalyzed synthesis of tri- and tetrasubstituted alkenyl silanes and stannanes via C–O functionalization of vinyl acetates using silylzinc pivalate and stannylzinc chloride as the nucleophiles.
- Ying Hu
- , Jiali Peng
- & Jie Li
-
Article
| Open Access Ex-situ generation and synthetic utilization of bare trifluoromethyl anion in flow via rapid biphasic mixing
Ex-situ generation and synthetic utilization of bare trifluoromethyl anion in flow via rapid biphasic mixing
The trifluoromethyl anion rapidly decomposes into difluorocarbene (:CF2) and fluorine ion, limiting its applicability in synthesis. Here, the authors report a strategy to generate and use the short-lived CF3- intermediate from stable CF3H gas via fast biphasic mixing in precisely customized flow dissolvers.
- Hyune-Jea Lee
- , Jeong-Un Joo
- & Heejin Kim
-
Article
| Open Access Polyfunctionalization of vicinal carbon centers and synthesis of unsymmetric 1,2,3,4-tetracarbonyl compounds
Polyfunctionalization of vicinal carbon centers and synthesis of unsymmetric 1,2,3,4-tetracarbonyl compounds
The synthesis of polycarbonyl compounds featuring four or more carbonyl groups is a synthetic challenge for organic chemists. Here, the authors describe how unsymmetric polycarbonyl compounds bearing up to 4 adjacent carbonyl groups can be accessed within a two-step synthesis from readily available starting materials and discuss their reactivity.
- Luca De Angelis
- , Chao Pei
- & Michael P. Doyle
-
Article
| Open Access Red light-driven electron sacrificial agents-free photoreduction of inert aryl halides via triplet-triplet annihilation
Red light-driven electron sacrificial agents-free photoreduction of inert aryl halides via triplet-triplet annihilation
The functionalization of aryl halides via photocatalysis typically involves the use of blue light. Here, the authors report the photoreduction of aryl halides enabled by red light; the method does not require a sacrificial electron donor.
- Le Zeng
- , Ling Huang
- & Gang Han
-
Article
| Open Access Cobalt(III)-catalyzed asymmetric ring-opening of 7-oxabenzonorbornadienes via indole C–H functionalization
Cobalt(III)-catalyzed asymmetric ring-opening of 7-oxabenzonorbornadienes via indole C–H functionalization
Asymmetric ring-opening (ARO) reactions, which can help quickly build molecular complexity, have not been extensively shown via β-oxygen elimination. Here, the authors report a cobalt(III)-catalyzed ARO reaction, providing mechanistic insights and the origins of the diastereo- and enantioselectivity.
- Yang Zheng
- , Wen-Yun Zhang
- & Shu-Li You
-
Article
| Open Access Interplay of diruthenium catalyst in controlling enantioselective propargylic substitution reactions with visible light-generated alkyl radicals
Interplay of diruthenium catalyst in controlling enantioselective propargylic substitution reactions with visible light-generated alkyl radicals
Construction of stereogenic carbon centers of propargylic alcohols with free radicals could mark a simple route to molecular complexity from easily obtained starting materials. Here the authors present a strategy to control enantioselective propargylic substitution reactions with alkyl radicals under photoredox conditions.
- Yulin Zhang
- , Yoshiaki Tanabe
- & Yoshiaki Nishibayashi
-
Article
| Open Access Chromium-catalyzed stereodivergent E- and Z-selective alkyne hydrogenation controlled by cyclic (alkyl)(amino)carbene ligands
Chromium-catalyzed stereodivergent E- and Z-selective alkyne hydrogenation controlled by cyclic (alkyl)(amino)carbene ligands
The stereoselective synthesis of olefins from alkynes is a long-standing challenge in organic synthesis. Here, the authors report the chromium-catalyzed alkyne hydrogenation; E- or Z-olefins are obtained depending on the carbene ligand used.
- Liang Ling
- , Chenyang Hu
- & Xiaoming Zeng
-
Article
| Open Access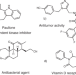 An unexpected synthesis of azepinone derivatives through a metal-free photochemical cascade reaction
An unexpected synthesis of azepinone derivatives through a metal-free photochemical cascade reaction
Photochemical nitrene transfer offers a green avenue for heterocyclic syntheses. Here, the authors developed a metal-free, visible light-mediated cascade reaction for the preparation of azepinone derivatives.
- Lina Song
- , Xianhai Tian
- & A. Stephen K. Hashmi
-
Article
| Open Access Enzyme-like polyene cyclizations catalyzed by dynamic, self-assembled, supramolecular fluoro alcohol-amine clusters
Enzyme-like polyene cyclizations catalyzed by dynamic, self-assembled, supramolecular fluoro alcohol-amine clusters
Terpene cyclases enable the synthesis of (poly)cyclic carbon frameworks via ring closure of linear polyenes. Here, the authors report in-situ formed fluorinated-alcohol-amine supramolecular clusters that mimic terpene cyclases for shape-controlled polyene cyclizations.
- Andreas M. Arnold
- , Philipp Dullinger
- & Tanja Gulder
-
Article
| Open Access Direct synthesis of sila-benzoazoles through hydrosilylation and rearrangement cascade reaction of benzoazoles and silanes
Direct synthesis of sila-benzoazoles through hydrosilylation and rearrangement cascade reaction of benzoazoles and silanes
Sila-substituted compounds have potential application in medicinal chemistry, but their preparation from the corresponding carbon analogues remains challenging. Here, the authors report the synthesis of sila-benzoazoles via hydrosilylation and rearrangement of benzoazoles; the reaction is enabled by a Lewis acid.
- Tianwei Liu
- , Mo Yang
- & Yuetao Zhang
-
Article
| Open Access Dearomative triple elementalization of quinolines driven by visible light
Dearomative triple elementalization of quinolines driven by visible light
Organoboron and organosilicon compounds have potential applications in various fields including materials science and medicinal chemistry. Here the authors report a protocol that enables the dearomative carbo-sila-boration of quinolines.
- Shiho Ishigaki
- , Yuki Nagashima
- & Ken Tanaka
-
Article
| Open Access Nickel-catalyzed divergent Mizoroki–Heck reaction of 1,3-dienes
Nickel-catalyzed divergent Mizoroki–Heck reaction of 1,3-dienes
Developing efficient strategies to realize divergent arylation of dienes has been a longstanding synthetic challenge. Herein, a nickel-catalyzed divergent Mizoroki–Heck reaction of 1,3-dienes has been demonstrated through the modification of ligands and additives.
- Wei-Song Zhang
- , Ding-Wei Ji
- & Qing-An Chen
-
Article
| Open Access Modulating stereoselectivity in allylic C(sp3)-H bond arylations via nickel and photoredox catalysis
Modulating stereoselectivity in allylic C(sp3)-H bond arylations via nickel and photoredox catalysis
The selective construction of C-C double bonds in alkenes, which can be one of two isomers, is a fundamental focus of organic chemistry. Here, the authors systematically investigated the stereoselectivity in allylic C(sp3)-H bond arylations and demonstrated a ligand switch could allow a stereodivergent synthesis of silyl enol ethers.
- Long Huang
- , Marcin Szewczyk
- & Magnus Rueping
-
Article
| Open Access The rocky road to organics needs drying
The rocky road to organics needs drying
How complex organics form in a prebiotic world remains a missing key to establish where life emerged. The authors present a road to abiotic organic synthesis and diversification in hydrothermal contexts involving magmatism and rock hydration.
- Muriel Andreani
- , Gilles Montagnac
- & Bénédicte Ménez
-
Article
| Open Access Preferential molecular recognition of heterochiral guests within a cyclophane receptor
Preferential molecular recognition of heterochiral guests within a cyclophane receptor
The design of synthetic supramolecular receptors capable of enantioselective molecular recognition is challenging. Here the authors report a perylene bisimide-based receptor that forms heterochiral host-guest complexes upon aromatic guests encapsulation; guest recognition can be modulated by peripheral substitution of the binding site.
- Manuel Weh
- , Kazutaka Shoyama
- & Frank Würthner
-
Article
| Open Access Additive-controlled asymmetric iodocyclization enables enantioselective access to both α- and β-nucleosides
Additive-controlled asymmetric iodocyclization enables enantioselective access to both α- and β-nucleosides
Nucleosides and their analogs are pharmacologically important molecules. Here, the authors report an additive-controlled stereodivergent iodocyclization method for the selective synthesis of α- or β-nucleosides.
- Qi Wang
- , Jiayi Mu
- & Fener Chen
-
Article
| Open Access Site- and enantioselective cross-coupling of saturated N-heterocycles with carboxylic acids by cooperative Ni/photoredox catalysis
Site- and enantioselective cross-coupling of saturated N-heterocycles with carboxylic acids by cooperative Ni/photoredox catalysis
Chiral α-acylated N-heterocycles are commonly found in pharmaceutical drugs, natural products, and catalysts for asymmetric synthesis. Here, the authors describe a cooperative nickel/photoredox catalysis strategy for the site- and enantioselective cross-coupling of saturated azacycles and carboxylic acids to access such valuable heterocyclic molecules.
- Xiaomin Shu
- , De Zhong
- & Haohua Huo
-
Article
| Open Access Formation and degradation of strongly reducing cyanoarene-based radical anions towards efficient radical anion-mediated photoredox catalysis
Formation and degradation of strongly reducing cyanoarene-based radical anions towards efficient radical anion-mediated photoredox catalysis
Cyanoarene-based photocatalysts are widely used due to their catalytic performance, but the formation of the active species and its potential degradation pathways are poorly understood. Here, the authors investigate these pathways under commonly-used photoredox-mediated reaction conditions.
- Yonghwan Kwon
- , Jungwook Lee
- & Min Sang Kwon
-
Article
| Open Access Ligand-controlled stereodivergent alkenylation of alkynes to access functionalized trans- and cis-1,3-dienes
Ligand-controlled stereodivergent alkenylation of alkynes to access functionalized trans- and cis-1,3-dienes
The stereoselective synthesis of 1,3-dienes represents a challenge in organic synthesis. Here, the authors report a ligand-controlled stereodivergent access to both Z- and E-sulfonyl-1,3-dienes via photoredox and nickel catalysis.
- Tianyu Long
- , Chen Zhu
- & Lingling Chu
-
Article
| Open Access Transition-metal free C–N bond formation from alkyl iodides and diazonium salts via halogen-atom transfer
Transition-metal free C–N bond formation from alkyl iodides and diazonium salts via halogen-atom transfer
The pursuit of efficient C–N bond formation is a prime focus of synthetic organic chemistry. Here, the authors documented a base promoted amination between alkyl iodides and diazonium salts via halogen-atom transfer (XAT) process.
- Jing Zhang
- , Min Jiang
- & Xinxin Shao
-
Article
| Open Access Iron-mediated ligand-to-metal charge transfer enables 1,2-diazidation of alkenes
Iron-mediated ligand-to-metal charge transfer enables 1,2-diazidation of alkenes
Alkene diazidations represent a promising strategy for the synthesis of 1,2-diamines. Here, the authors report a protocol that enables alkene diazidation via iron-mediated ligand-to-metal charge transfer, providing a versatile platform to access structurally diverse diazides without using external oxidants.
- Muliang Zhang
- , Jinghui Zhang
- & Yumeng Shi
-
Article
| Open Access Photochemical diazidation of alkenes enabled by ligand-to-metal charge transfer and radical ligand transfer
Photochemical diazidation of alkenes enabled by ligand-to-metal charge transfer and radical ligand transfer
The difunctionalization of alkenes allows for efficient construction of molecular complexity. Combining Ligand-to-Metal Charge transfer (LMCT) and Radical Ligand Transfer (RLT), the authors show a photochemical diazidation using iron salts, allowing rapid preparation of vicinal diamines and other privileged synthetic motifs.
- Kang-Jie Bian
- , Shih-Chieh Kao
- & Julian G. West
-
Article
| Open Access Ni-catalyzed benzylic β-C(sp3)–H bond activation of formamides
Ni-catalyzed benzylic β-C(sp3)–H bond activation of formamides
The development of 3d-metal-catalyzed β-C–H bond activation via 4-membered metallacycles remains an elusive challenge. Here, the authors report a Ni-Al bimetal-catalyzed β-C(sp3)–H bond activation of formamides via 4-membered nickelacycles.
- Rong-Hua Wang
- , Wei-Wei Xu
- & Mengchun Ye
-
Article
| Open Access Synthesis of macrocyclic nucleoside antibacterials and their interactions with MraY
Synthesis of macrocyclic nucleoside antibacterials and their interactions with MraY
MraY is a membrane enzyme required for bacterial cell wall synthesis. Here, the authors modify sphaerimicins as antibacterials targeting it via structure-based design and synthesis through two key reactions, showing a platform for further development of MraY inhibitors as antibacterials.
- Takeshi Nakaya
- , Miyuki Yabe
- & Satoshi Ichikawa
-
Article
| Open Access Rational enzyme design for enabling biocatalytic Baldwin cyclization and asymmetric synthesis of chiral heterocycles
Rational enzyme design for enabling biocatalytic Baldwin cyclization and asymmetric synthesis of chiral heterocycles
Chiral heterocyclic compounds are privileged structures in medicinal chemistry. Here, the authors report an in silico strategy for the enzymatic synthesis of pharmaceutically significant chiral N- and O-heterocycles via Baldwin cyclization of hydroxy- and amino-substituted epoxides and oxetanes using epoxide hydrolase mutants.
- Jun-Kuan Li
- , Ge Qu
- & Zhoutong Sun

 Asymmetric α-allylic allenylation of β-ketocarbonyls and aldehydes by synergistic Pd/chiral primary amine catalysis
Asymmetric α-allylic allenylation of β-ketocarbonyls and aldehydes by synergistic Pd/chiral primary amine catalysis