Article
|
Open Access
Featured
-
-
News & Views |
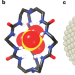 Clever cryptand cage coordinates contaminants
Clever cryptand cage coordinates contaminants
Replicating the ability of enzymes and transport proteins to effectively bind anions is a considerable challenge for supramolecular chemists. A neutral organic cage has now been developed that selectively binds sulfate anions in water.
- Rosemary J. Goodwin
- & Nicholas G. White
-
Article |
 A charge-neutral organic cage selectively binds strongly hydrated sulfate anions in water
A charge-neutral organic cage selectively binds strongly hydrated sulfate anions in water
Very few charge-neutral synthetic anion receptors can function in water, and those known typically select weakly hydrated anions such as iodide. Now a neutral molecular cage capable of donating 12 hydrogen bonds has been synthesized and found to bind highly hydrated sulfate in water with a strong selectivity over weakly hydrated anions.
- Liuyang Jing
- , Evelyne Deplazes
- & Xin Wu
-
Article |
 Non-statistical assembly of multicomponent [Pd2ABCD] cages
Non-statistical assembly of multicomponent [Pd2ABCD] cages
Metal-mediated self-assembly of organic building blocks is a powerful strategy to generate complex supramolecular objects. The non-statistical combination of multiple different components, however, has been a major challenge. Now integrative self-sorting of low-symmetry multicomponent cages has been achieved by combining shape complementarity and selective backbone interactions under thermodynamic control.
- Kai Wu
- , Elie Benchimol
- & Guido H. Clever
-
Perspective |
 Photoswitchable coordination cages
Photoswitchable coordination cages
The self-assembly of stimuli-responsive building blocks yields functional nano-systems and smart materials. This Perspective discusses how the integration of photoswitches into discrete coordination cages enables control over their assembly, guest binding and systems behaviour. Four scenarios are drawn to highlight the relationship between the photoswitching and dynamic assembly equilibria.
- Elie Benchimol
- , Jacopo Tessarolo
- & Guido H. Clever
-
News & Views |
 Stable organolithium gels
Stable organolithium gels
Organolithium reagents are characterized by their high reactivity towards air and moisture, traditionally requiring strict inert conditions for their handling and utilization. Now, these reagents can be encapsulated within an organogel, enhancing their stability and allowing their use and storage under ambient conditions.
- Andreu Tortajada
- & Eva Hevia
-
Article
| Open Access Organogel delivery vehicles for the stabilization of organolithium reagents
Organogel delivery vehicles for the stabilization of organolithium reagents
Organolithium compounds are important reagents but are often hazardous due to their high reactivity. Now, encapsulating organolithium reagents within a supramolecular organogel has been found to enhance their stability, facilitating their storage and handling under ambient conditions. The homogeneous gels can be easily subdivided and dosed into a wide range of synthetic reactions.
- Petr Slavík
- , Benjamin R. Trowse
- & David K. Smith
-
Article |
 Enantioselective fullerene functionalization through stereochemical information transfer from a self-assembled cage
Enantioselective fullerene functionalization through stereochemical information transfer from a self-assembled cage
The enantioselective functionalization of C60 is highly challenging, typically requiring complex chiral tethers or demanding chromatography. Fullerenes have now been shown to undergo Diels–Alder reactions in a chemo-, regio- and enantio-selective fashion through confinement within an enantiopure metal–organic cage functionalized with a chiral formylpyridine group.
- Zifei Lu
- , Tanya K. Ronson
- & Jonathan R. Nitschke
-
Article
| Open Access Dimeric and trimeric catenation of giant chiral [8 + 12] imine cubes driven by weak supramolecular interactions
Dimeric and trimeric catenation of giant chiral [8 + 12] imine cubes driven by weak supramolecular interactions
Interlocked shape-persistent organic cages are rare structures and the majority are formed using π-stacking as the driving force. Now it is shown that weak dispersion interactions—which are modulated by changing the 1,4-substituents of the constituent dialdehyde linkers—can be used to form interlocked dimeric and trimeric catenated cages.
- Bahiru Punja Benke
- , Tobias Kirschbaum
- & Michael Mastalerz
-
News & Views |
 Pop-up rings to rule reactivity
Pop-up rings to rule reactivity
An alcohol-functionalized cyclophane has been shown to form a bowl-shaped cavity large enough to host tris(bipyridine)ruthenium(ii). The macrocycle captures, and suppresses the activity of, the ruthenium complex in water but, when oxidized to its ketone-bearing counterpart, releases the ruthenium guest.
- David Van Craen
-
News & Views |
 Hopping protons in supramolecular catalysis
Hopping protons in supramolecular catalysis
Supramolecular catalysis can emulate many features of enzymatic transformations. Now, a complex proton wire mechanism — enabling the dual activation of a nucleophile and an electrophile through reciprocal proton transfer — has been shown to operate during the β-glycosylation of sugars within a self-assembled capsule.
- Cally J. E. Haynes
- & Larissa K. S. von Krbek
-
Article |
 Mimicry of the proton wire mechanism of enzymes inside a supramolecular capsule enables β-selective O-glycosylations
Mimicry of the proton wire mechanism of enzymes inside a supramolecular capsule enables β-selective O-glycosylations
Proton wires in enzyme active sites enable stereoselective reactions under mild conditions by facilitating dual activation of a nucleophile and an electrophile through reciprocal proton transfer. Now it has been shown that such an activation mode can be mimicked inside a supramolecular capsule, enabling a general approach to β-glycosides.
- Tian-Ren Li
- , Fabian Huck
- & Konrad Tiefenbacher
-
News & Views |
 A ravel alliance
A ravel alliance
A new class of interwoven metal–organic containers, including one with a cubic architecture, twelve crossing points and a large internal volume, has now been reported. Interconversion between different self-assembled structures can be triggered by simply exchanging the associated anions.
- Andrew W. Heard
- , Natasha M. A. Speakman
- & Jonathan R. Nitschke
-
-
-
Article |
 A biomimetic receptor for glucose
A biomimetic receptor for glucose
Synthetic receptors can be used to help understand biological systems, but rarely compete in terms of affinity or selectivity. Now, a glucose-binding compound has been prepared that, despite its symmetry and simplicity, can match all but the strongest glucose-binding proteins. The high binding affinity and outstanding selectivity of this receptor may translate into biomedical applications.
- Robert A. Tromans
- , Tom S. Carter
- & Anthony P. Davis
-
Article |
 Signal transduction in a covalent post-assembly modification cascade
Signal transduction in a covalent post-assembly modification cascade
Two coordination cages have been devised that undergo covalent modification during a cascade of two orthogonal Diels–Alder reactions. This results in increased lipophilicity for the second cage, enabling its phase transfer and separation from the first. The trigger, relay and inhibition features of this cascade system mimic key aspects of natural post-translational modification cascades.
- Ben S. Pilgrim
- , Derrick A. Roberts
- & Jonathan R. Nitschke
-
Article |
 Stereochemical plasticity modulates cooperative binding in a CoII12L6 cuboctahedron
Stereochemical plasticity modulates cooperative binding in a CoII12L6 cuboctahedron
An interconverting system of three distinct stereoisomers of a cuboctahedral CoII-based cage is able to regulate the binding affinities of large anionic guests. Through cooperative templation with fullerene guests, the cage converts into a desymmetrized cage that in turn exhibits positive cooperativity in binding of an icosahedral anion; this interaction is anti-cooperative in the fullerene-free parent.
- Felix J. Rizzuto
- & Jonathan R. Nitschke
-
Article |
 Reticular synthesis of porous molecular 1D nanotubes and 3D networks
Reticular synthesis of porous molecular 1D nanotubes and 3D networks
Porous molecular crystals have desirable properties, but are hard to form with the level of structural control seen for extended framework materials. Now, a ‘mix-and-match’ chiral recognition strategy has been used to form reticular porous supramolecular nanotubes and 3D networks, providing a blueprint for pore design in molecular crystals.
- A. G. Slater
- , M. A. Little
- & A. I. Cooper
-
Article |
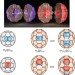 Synthesis of a distinct water dimer inside fullerene C70
Synthesis of a distinct water dimer inside fullerene C70
Endohedral C70 fullerenes containing either one or two water molecules have now been prepared using a molecular-surgery approach. The structure of H2O@C70 was determined by single-crystal X-ray analysis, revealing the encapsulated water molecule to be in an off-centre position. In (H2O)2@C70, the two water molecules form a discrete dimer held together with a single hydrogen bond.
- Rui Zhang
- , Michihisa Murata
- & Yasujiro Murata
-
Article |
 Highly efficient catalysis of the Kemp elimination in the cavity of a cubic coordination cage
Highly efficient catalysis of the Kemp elimination in the cavity of a cubic coordination cage
The Kemp elimination has been catalysed in the cavity of a coordination cage with a rate enhancement (kcat/kuncat) of 200,000 at pD 8.5. The catalysis requires two orthogonal interactions to bring together the components: hydrophobic binding of benzisoxazole, and accumulation of hydroxide ions at the cationic cage surface by ion-pairing.
- William Cullen
- , M. Cristina Misuraca
- & Michael D. Ward
-
Article |
 Self-assembled nanospheres with multiple endohedral binding sites pre-organize catalysts and substrates for highly efficient reactions
Self-assembled nanospheres with multiple endohedral binding sites pre-organize catalysts and substrates for highly efficient reactions
Preorganization of catalysts and substrates can lead to significant rate enhancement—an effect often observed in enzyme catalysis. Now, a self-assembled nanosphere equipped with 24 guanidinium binding sites is demonstrated to strongly bind sulfonate-containing gold catalysts. Base-triggered co-encapsulation of carboxylate containing substrates leads to pronounced gating effects and dramatically enhanced reaction rates.
- Qi-Qiang Wang
- , Sergio Gonell
- & Joost N. H. Reek
-
-
Article |
 Iterative design of a helically folded aromatic oligoamide sequence for the selective encapsulation of fructose
Iterative design of a helically folded aromatic oligoamide sequence for the selective encapsulation of fructose
Designing synthetic molecular receptors that can differentiate between specific monosaccharide guests is very challenging. Now, a helically folded oligoamide that selectively encapsulates fructose has been designed using an iterative approach that exploits the modular structure of folded synthetic oligomer sequences, in conjunction with molecular modelling and structural characterization.
- Nagula Chandramouli
- , Yann Ferrand
- & Ivan Huc
-
News & Views |
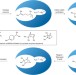 Terpenes in tight spaces
Terpenes in tight spaces
The ability of enzymes to direct the synthesis of complex natural products from simple starting materials is epitomized by terpene biosynthesis. Now, a supramolecular catalyst has been shown to mimic some of the reactivity of this process.
- Jeremy J. Roach
- & Ryan A. Shenvi
-
Article |
 Terpene cyclization catalysed inside a self-assembled cavity
Terpene cyclization catalysed inside a self-assembled cavity
A tail-to-head terpene cyclization, which is hard to control in solution, has now been catalysed inside a supramolecular structure. Evidence indicates that a direct isomerization of a geranyl cation to the cisoid-isomer, which so far was considered unlikely in the biosynthesis, is feasible in this system.
- Q. Zhang
- & K. Tiefenbacher
-
News & Views |
 Supramolecular joinery
Supramolecular joinery
Attempts to create a porous molecular crystal by removing solvent molecules from a solvate usually lead the host to reorganize into a non-porous close-packed structure. The 'virtual porosity' of such an organic cage crystal has now been trapped by introducing a judiciously chosen co-crystal former that prevents rearrangement of the host lattice.
- Leonard J. Barbour
-
Article |
 Trapping virtual pores by crystal retro-engineering
Trapping virtual pores by crystal retro-engineering
Organic molecular crystals with guest-occupied cavities are often observed, but the cavities tend to collapse when the guests are removed. Now, the porous domain of a crystalline solvate has been stabilized by formation of a cocrystal with a second molecule whose size and shape matches those of the unstable voids.
- Marc A. Little
- , Michael E. Briggs
- & Andrew I. Cooper
-
News & Views |
 Tipping a cell's ionic balance
Tipping a cell's ionic balance
A synthetic compound that transports chloride across membranes can kill both normal cells and cancer cells in vitro. The transporter works together with sodium channels to move NaCl into the cells, which triggers cell death.
- Jeffery T. Davis
-
Article |
 Acyclic cucurbit[n]uril molecular containers enhance the solubility and bioactivity of poorly soluble pharmaceuticals
Acyclic cucurbit[n]uril molecular containers enhance the solubility and bioactivity of poorly soluble pharmaceuticals
Acyclic cucurbituril-type molecular containers have been found to increase the solubility of insoluble pharmaceutical agents in water by up to 2,750-fold. In vitro and in vivo toxicology studies suggest that the containers are well tolerated, and paclitaxel solubilized in this manner efficiently kills HeLa and SK-OV-3 cancer cells.
- Da Ma
- , Gaya Hettiarachchi
- & Lyle Isaacs
-
News & Views |
 Under control
Under control
Macrocyclic compounds can serve as hosts for smaller organic molecules, but precise control over the uptake and release of the guests remains challenging. Now, a host–guest system has been built that responds to the addition of metal ions, showing promise for drug-delivery applications.
- Werner M. Nau

 Supramolecular trapping of a cationic all-metal σ-aromatic {Bi4} ring
Supramolecular trapping of a cationic all-metal σ-aromatic {Bi4} ring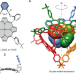 Double-walled design for tetrahedral cages
Double-walled design for tetrahedral cages Analysing allostery
Analysing allostery Coordination driven gelation
Coordination driven gelation