News & Views |
Featured
-
-
News & Views |
 ER remodelling by ER-phagy in neurogenesis
ER remodelling by ER-phagy in neurogenesis
The endoplasmic reticulum (ER) controls the synthesis of lipids and proteins and Ca2+ homeostasis, as well as contacting other organelles and the plasma membrane. A study now looks at a process by which this compartment is remodelled in axons during neurogenesis: the lysosomal clearance of ER subdomains, driven by FAM134 and CCPG1 proteins.
- Maurizio Molinari
-
Article
| Open Access Combinatorial selective ER-phagy remodels the ER during neurogenesis
Combinatorial selective ER-phagy remodels the ER during neurogenesis
Hoyer et al. establish that selective autophagy mechanisms are needed to remodel the ER and its proteome during in vitro neurogenesis across neuronal subcompartments and decode the substrate selectivity of ER-phagy receptors.
- Melissa J. Hoyer
- , Cristina Capitanio
- & J. Wade Harper
-
Article
| Open Access Arf1 coordinates fatty acid metabolism and mitochondrial homeostasis
Arf1 coordinates fatty acid metabolism and mitochondrial homeostasis
Enkler et al. show that a pool of Arf1 at lipid droplets is implicated in mitochondrial ATP production control through regulation of fatty acid metabolism and acetyl-CoA transfer to mitochondria.
- Ludovic Enkler
- , Viktoria Szentgyörgyi
- & Anne Spang
-
News & Views |
 Endoplasmic reticulum tethering by desmosomes
Endoplasmic reticulum tethering by desmosomes
Desmosomes and keratin are now found to regulate the distribution and dynamics of the endoplasmic reticulum (ER). This suggests that a range of ER functions may be coordinated by this intercellular adhesive and cytoskeletal network.
- Robert M. Harmon
- & Cara J. Gottardi
-
Article |
 Architecture and dynamics of a desmosome–endoplasmic reticulum complex
Architecture and dynamics of a desmosome–endoplasmic reticulum complex
Bharathan et al. discover that the endoplasmic reticulum associates with keratin intermediate filaments and desmosomal cell–cell junctions, and that desmosomes and the keratin cytoskeleton regulate the distribution, dynamics and function of the endoplasmic reticulum network.
- Navaneetha Krishnan Bharathan
- , William Giang
- & Andrew P. Kowalczyk
-
Research Briefing |
 Endoplasmic reticulum-associated degradation shown to regulate innate immunity
Endoplasmic reticulum-associated degradation shown to regulate innate immunity
The SEL1L–HRD1 endoplasmic reticulum-associated degradation pathway negatively regulates STING-mediated innate immunity by ubiquitinating and targeting STING for proteasomal degradation, thereby limiting its activation.
-
Article
| Open Access SEL1L–HRD1 endoplasmic reticulum-associated degradation controls STING-mediated innate immunity by limiting the size of the activable STING pool
SEL1L–HRD1 endoplasmic reticulum-associated degradation controls STING-mediated innate immunity by limiting the size of the activable STING pool
Ji et al. report that under basal conditions the SEL1L–HRD1 complex for endoplasmic reticulum-associated degradation ubiquitinates and negatively regulates STING protein levels in the endoplasmic reticulum, hence its activation.
- Yewei Ji
- , Yuan Luo
- & Ling Qi
-
Article
| Open Access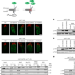 ER-associated RNA silencing promotes ER quality control
ER-associated RNA silencing promotes ER quality control
Efstathiou et al. describe an Argonaute-dependent endoplasmic reticulum (ER)-associated RNA silencing pathway that acts together with ER-associated protein degradation to preserve ER homeostasis and function.
- Sotirios Efstathiou
- , Franziska Ottens
- & Thorsten Hoppe
-
Article
| Open Access Identification of two pathways mediating protein targeting from ER to lipid droplets
Identification of two pathways mediating protein targeting from ER to lipid droplets
Song et al. identify two protein-targeting pathways from the endoplasmic reticulum to (1) early lipid droplets (LDs) and (2) mature lipid droplets. They define key factors mediating the second, late pathway and its many cargoes.
- Jiunn Song
- , Arda Mizrak
- & Tobias C. Walther
-
News & Views |
 Membranes regulate biomolecular condensates
Membranes regulate biomolecular condensates
Biomolecular condensation has emerged as a fundamental mechanism for cellular organization, but less is known about the regulation of condensate subcellular location and size. A new study reports that membrane tethering of protein and RNA directly influences the assembly, size and material properties of ribonucleic condensates.
- Lindsay B. Case
-
Article |
 Membrane surfaces regulate assembly of ribonucleoprotein condensates
Membrane surfaces regulate assembly of ribonucleoprotein condensates
Snead et al. report that membrane tethering facilitates assembly of ribonucleoprotein condensates while also restricting condensate size by reducing the diffusion of protein and RNA.
- Wilton T. Snead
- , Ameya P. Jalihal
- & Amy S. Gladfelter
-
Article |
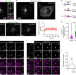 A Golgi-derived vesicle potentiates PtdIns4P to PtdIns3P conversion for endosome fission
A Golgi-derived vesicle potentiates PtdIns4P to PtdIns3P conversion for endosome fission
Gong et al. uncover a role for the Golgi network in ER-associated endosome fission, showing that a Golgi-derived SEC14L2 compartment mediates ER-associated endosome fission by promoting PtdIns4P to PtdIns3P conversion.
- Bo Gong
- , Yuting Guo
- & Shunji Jia
-
Letter |
 Protein quality control through endoplasmic reticulum-associated degradation maintains haematopoietic stem cell identity and niche interactions
Protein quality control through endoplasmic reticulum-associated degradation maintains haematopoietic stem cell identity and niche interactions
Xu, Liu, Pen, Zhang et al. demonstrate that the SEL1L–HRD1 complex, which is part of the ERAD protein quality control machinery, safeguards haematopoietic stem cell identity and niche location by ensuring the haematopoietic stem cell surface expression of MPL.
- Longyong Xu
- , Xia Liu
- & Xi Chen
-
Letter |
 Hrd1 forms the retrotranslocation pore regulated by auto-ubiquitination and binding of misfolded proteins
Hrd1 forms the retrotranslocation pore regulated by auto-ubiquitination and binding of misfolded proteins
Ubiquitin ligase Hrd1 is essential for endoplasmic-reticulum-associated protein degradation. Vasic et al. demonstrate that Hrd1 forms a retrotranslocation channel controlled by auto-ubiquitination and substrate binding.
- Vedran Vasic
- , Niels Denkert
- & Michael Meinecke
-
Article |
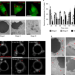 Phagolysosome resolution requires contacts with the endoplasmic reticulum and phosphatidylinositol-4-phosphate signalling
Phagolysosome resolution requires contacts with the endoplasmic reticulum and phosphatidylinositol-4-phosphate signalling
Levin-Konigsberg et al. show that resorption of the phagolysosome after degradation of its contents requires transfer of PI4P and tethering to the ER, both mediated by oxysterol-binding protein-related protein 1L (ORP1L).
- Roni Levin-Konigsberg
- , Fernando Montaño-Rendón
- & Sergio Grinstein
-
Article |
 ER–lysosome contacts enable cholesterol sensing by mTORC1 and drive aberrant growth signalling in Niemann–Pick type C
ER–lysosome contacts enable cholesterol sensing by mTORC1 and drive aberrant growth signalling in Niemann–Pick type C
Lim et al. show that OSBP and VAPA and VAPB deliver cholesterol across ER–lysosome contacts to activate mTORC1. OSBP-mediated cholesterol trafficking activates mTORC1 in a disease model caused by loss of Niemann–Pick C1.
- Chun-Yan Lim
- , Oliver B. Davis
- & Roberto Zoncu
-
News & Views |
 IRE1α modulates ER and mitochondria crosstalk
IRE1α modulates ER and mitochondria crosstalk
IRE1α is an endoplasmic reticulum (ER) transmembrane protein known for a crucial role in regulating the unfolding protein response. A study now shows that IRE1α interacts with the main ER Ca2+ channel InsP3Rs and facilitates the transfer of Ca2+ from the ER into mitochondria, thus driving cellular metabolism.
- Roland Malli
- & Wolfgang F. Graier
-
Article |
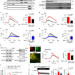 Non-canonical function of IRE1α determines mitochondria-associated endoplasmic reticulum composition to control calcium transfer and bioenergetics
Non-canonical function of IRE1α determines mitochondria-associated endoplasmic reticulum composition to control calcium transfer and bioenergetics
Carreras-Sureda et al. uncover a non-canonical role for IRE1α as a scaffold that stabilizes InsP3Rs at MAMs to control calcium uptake, fine-tunes ER–mitochondrial communication and regulates energy metabolism via AMPK.
- Amado Carreras-Sureda
- , Fabián Jaña
- & Claudio Hetz
-
Article |
 Ubiquilins regulate autophagic flux through mTOR signalling and lysosomal acidification
Ubiquilins regulate autophagic flux through mTOR signalling and lysosomal acidification
Şentürk et al. show that ubiquilins bind v-ATPase to control lysosome acidity, mTOR signalling and autophagic flux in neurons, and that feeding flies with acidic nanoparticles ameliorates defective autophagy in ubiquilin mutants.
- Mümine Şentürk
- , Guang Lin
- & Hugo J. Bellen
-
Letter |
 Single particle trajectories reveal active endoplasmic reticulum luminal flow
Single particle trajectories reveal active endoplasmic reticulum luminal flow
Using super-resolution microscopy, tracking of single particle trajectories of endoplasmic reticulum (ER) luminal proteins traversing tubular ER, and measuring ER dynamics, Holcman et al. show that ER content is propelled by active luminal flow.
- David Holcman
- , Pierre Parutto
- & Edward Avezov
-
Article |
 IRE1α governs cytoskeleton remodelling and cell migration
through a direct interaction with filamin A
IRE1α governs cytoskeleton remodelling and cell migration
through a direct interaction with filamin A
Urra et al. discover that IRE1α, an ER stress mediator, interacts with filamin A and controls actin dynamics and cell migration in mouse, Drosophila and zebrafish models in a manner independent of its canonical function.
- Hery Urra
- , Daniel R. Henriquez
- & Claudio Hetz
-
-
Review Article |
 Organelle biogenesis in the endoplasmic reticulum
Organelle biogenesis in the endoplasmic reticulum
In this Review, Prinz and co-authors discuss the role of the endoplasmic reticulum (ER) in the de novo generation of peroxisomes, lipid droplets and omegasomes, and how this requires subdomains with specific protein and lipid compositions.
- Amit S. Joshi
- , Hong Zhang
- & William A. Prinz
-
Letter |
 Endoplasmic-reticulum-mediated microtubule alignment governs cytoplasmic streaming
Endoplasmic-reticulum-mediated microtubule alignment governs cytoplasmic streaming
Through imaging and theoretical modelling, Kimura et al. discover that endoplasmic reticulum flow determines microtubule alignment to promote cytoplasmic streaming of yolk granules in Caenorhabditis elegans zygotes.
- Kenji Kimura
- , Alexandre Mamane
- & Akatsuki Kimura
-
News & Views |
 CPEB4 links the clock and the UPR to protect the liver
CPEB4 links the clock and the UPR to protect the liver
Under misfolded protein stress, the endoplasmic reticulum (ER) activates the unfolded protein response (UPR) to restore homeostasis, or commits the cell to apoptosis. A study now uncovers how the UPR is governed by the circadian clock to adjust ER protein-folding capacity to metabolic demand and protect against liver damage.
- Paul C. Moore
- & Scott A. Oakes
-
Article |
 Circadian- and UPR-dependent control of CPEB4 mediates a translational response to counteract hepatic steatosis under ER stress
Circadian- and UPR-dependent control of CPEB4 mediates a translational response to counteract hepatic steatosis under ER stress
Maillo et al. show that in hepatocytes ER stress upregulates CPEB4 through the UPR and circadian clock, leading to CPEB4-mediated translation for mitochondrial and ER homeostasis. CPEB4 loss leads to ageing- and high fat diet-induced liver steatosis.
- Carlos Maillo
- , Judit Martín
- & Raúl Méndez
-
News & Views |
 On keeping the right ER size
On keeping the right ER size
The endoplasmic reticulum (ER) is the largest membrane-bound organelle in cells, and its size needs to be carefully controlled. Downsizing the ER by autophagy is now shown to involve Sec62, a protein that also helps to build up the organelle. This link suggests a molecular switch for ER size control.
- Sebastian Schuck
-
Article |
 Translocon component Sec62 acts in endoplasmic reticulum turnover during stress recovery
Translocon component Sec62 acts in endoplasmic reticulum turnover during stress recovery
Fumagalli et al. show that Sec62 delivers ER components to the autolysosome for clearance by acting as a receptor for autophagy protein LC3-II. This identifies Sec62 as a critical factor for selective ER turnover.
- Fiorenza Fumagalli
- , Julia Noack
- & Maurizio Molinari
-
Article |
 Peroxin-dependent targeting of a lipid-droplet-destined membrane protein to ER subdomains
Peroxin-dependent targeting of a lipid-droplet-destined membrane protein to ER subdomains
Kopito and Schrul show that the peroxisome proteins PEX19 and PEX3 mediate the correct insertion of the lipid droplet protein UBXD8 into ER subdomains.
- Bianca Schrul
- & Ron R. Kopito
-
News & Views |
 Directing lipid transport at membrane contact sites
Directing lipid transport at membrane contact sites
Contact sites between the endoplasmic reticulum and the plasma membrane mediate receptor signalling. How this function is controlled physically and functionally is poorly understood. Extended synaptotagmins are now shown to shuttle the lipid metabolite diacylglycerol from the plasma membrane to the endoplasmic reticulum in receptor-stimulated cells.
- Michael Krauβ
- & Volker Haucke
-
Article |
 Control of plasma membrane lipid homeostasis by the extended synaptotagmins
Control of plasma membrane lipid homeostasis by the extended synaptotagmins
Saheki and colleagues show that extended synaptotagmins (E-Syts), ER proteins that function as tethers to the plasma membrane, can transfer lipids between bilayers in a Ca2+- and SMP-domain-dependent manner, thus regulating plasma membrane lipid homeostasis.
- Yasunori Saheki
- , Xin Bian
- & Pietro De Camilli
-
Article |
 IRE1α is an endogenous substrate of endoplasmic-reticulum-associated degradation
IRE1α is an endogenous substrate of endoplasmic-reticulum-associated degradation
Through a proteomics approach, Qi and colleagues and Long and colleagues identify the sensor of the unfolded protein response IRE1α as an endogenous substrate of the E3 ubiquitin ligase involved in ER-associated degradation, Hrd1.
- Shengyi Sun
- , Guojun Shi
- & Ling Qi
-
News & Views |
 STIMATE reveals a STIM1 transitional state
STIMATE reveals a STIM1 transitional state
Decreases in endoplasmic reticulum calcium content are sensed by resident STIM proteins, which can activate plasma membrane Orai channels to facilitate Ca2+ entry. The role of STIMATE, a previously unknown component of the store-operated calcium entry complex, has now been identified and defined.
- Robert Hooper
- & Jonathan Soboloff
-
Letter |
 Proteomic mapping of ER–PM junctions identifies STIMATE as a regulator of Ca2+ influx
Proteomic mapping of ER–PM junctions identifies STIMATE as a regulator of Ca2+ influx
Through a proteomic analysis of ER–PM junctions, Zhou and colleagues and Wang and colleagues discover that the transmembrane protein STIMATE is a positive regulator of STIM localization and function, thereby stimulating Ca2+ influx.
- Ji Jing
- , Lian He
- & Yubin Zhou
-
News & Views |
 How cells coordinate waste removal through their major proteolytic pathways
How cells coordinate waste removal through their major proteolytic pathways
The eukaryotic cell uses two complex machineries to degrade unwanted proteins. The first is the ubiquitin–proteasome system and the second is autophagy. A new study contributes to our understanding of how the two systems interconnect to coordinate protein degradation.
- Sascha Martens
- & Andreas Bachmair
-
Review Article |
 Proteostasis control by the unfolded protein response
Proteostasis control by the unfolded protein response
- Claudio Hetz
- , Eric Chevet
- & Scott A. Oakes
-
Article |
 Amino-terminal arginylation targets endoplasmic reticulum chaperone BiP for autophagy through p62 binding
Amino-terminal arginylation targets endoplasmic reticulum chaperone BiP for autophagy through p62 binding
Kim and colleagues and Kwon and colleagues reveal that amino-terminal arginylation of BiP promotes its targeting to autophagy adaptor p62 and subsequent lysosomal degradation of BiP, p62 and associated cargo.
- Hyunjoo Cha-Molstad
- , Ki Sa Sung
- & Yong Tae Kwon
-
Article |
 Der1 promotes movement of misfolded proteins through the endoplasmic reticulum membrane
Der1 promotes movement of misfolded proteins through the endoplasmic reticulum membrane
How misfolded proteins are extracted from the endoplasmic reticulum (ER) for degradation remains unclear. Sommer and colleagues demonstrate that following assembly into the HRD ligase complex, Der1 forms oligomers in the ER membrane and enables extraction of proteins from the ER lumen.
- Martin Mehnert
- , Thomas Sommer
- & Ernst Jarosch
-
Article |
 PtdIns(3)P-bound UVRAG coordinates Golgi–ER retrograde and Atg9 transport by differential interactions with the ER tether and the beclin 1 complex
PtdIns(3)P-bound UVRAG coordinates Golgi–ER retrograde and Atg9 transport by differential interactions with the ER tether and the beclin 1 complex
ER–Golgi transport and autophagy are tightly connected. Liang and colleagues find that UVRAG binds to PtdIns(3)P to localize it to the ER, from where, under normal conditions, it regulates the transport of COPI cargo transfer to the ER and Golgi integrity, but from where, following autophagy induction, it dissociates to modulate ATG9 transfer to autophagosomes.
- Shanshan He
- , Duojiao Ni
- & Chengyu Liang
-
Article |
 ER-stress-induced transcriptional regulation increases protein synthesis leading to cell death
ER-stress-induced transcriptional regulation increases protein synthesis leading to cell death
In the presence of stress stimuli, the endoplasmic reticulum either adapts the protein synthesis or triggers an apoptotic response, but the mechanisms underlying this decision are unknown. Kaufman and colleagues show that the ER stress response factors ATF4 and CHOP increase protein synthesis, which in turn induces oxidative stress and increased ATP consumption, leading to cell death during chronic ER stress.
- Jaeseok Han
- , Sung Hoon Back
- & Randal J. Kaufman
-
News & Views |
 Rab10 joins the ER social network
Rab10 joins the ER social network
The endoplasmic reticulum (ER) is a heterogeneous organelle with distinct morphologies of sheets and an interconnected network of tubules sharing a common lumen. An ER domain marked by the Rab10 GTPase and several lipid-synthesizing enzymes is implicated in dynamic ER tubule formation and fusion events in cells.
- Jaerak Chang
- & Craig Blackstone
-
Article |
 Rab10 GTPase regulates ER dynamics and morphology
Rab10 GTPase regulates ER dynamics and morphology
ER tubules grow and fuse to give the ER its characteristic shape. English and Voeltz show that the small GTPase Rab10 is crucial for ER tubule growth and fusion. Rab10 localizes to the leading edge of new ER tubules with enzymes that promote phospholipid synthesis, suggesting that ER tubule growth, fusion and phospholipid synthesis might be coupled.
- Amber R. English
- & Gia K. Voeltz
-
Letter |
 PARP16 is a tail-anchored endoplasmic reticulum protein required for the PERK- and IRE1α-mediated unfolded protein response
PARP16 is a tail-anchored endoplasmic reticulum protein required for the PERK- and IRE1α-mediated unfolded protein response
Chang and colleagues reveal that the poly(ADP-ribose) polymerase PARP16 participates in the endoplasmic reticulum (ER) stress response. They report that PARP16 is a transmembrane ER protein that PARsylates and activates PERK and IRE1α in response to ER stress.
- Miri Jwa
- & Paul Chang
-
Article |
 ER network formation requires a balance of the dynamin-like GTPase Sey1p and the Lunapark family member Lnp1p
ER network formation requires a balance of the dynamin-like GTPase Sey1p and the Lunapark family member Lnp1p
The endoplasmic reticulum (ER) forms an intricate network of interconnected tubules. Sey1 is known to govern tubule formation, but the proteins that counteract tubule fusion remained unclear. Chen, Novick and Ferro-Novick propose that Lnp1 antagonizes the activity of Sey1 to modulate ER network formation.
- Shuliang Chen
- , Peter Novick
- & Susan Ferro-Novick
-
-
Article |
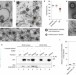 BAP31 and BiP are essential for dislocation of SV40 from the endoplasmic reticulum to the cytosol
BAP31 and BiP are essential for dislocation of SV40 from the endoplasmic reticulum to the cytosol
Non-enveloped viruses such as SV40 are transported from the extracellular space into the host cell nucleus through a pathway involving endocytosis, trafficking to the endoplasmic reticulum (ER) lumen, transport across the ER membrane to the cytoplasm, and subsequent nuclear import. Helenius and colleagues provide insight into how SV40 escapes from the ER by showing that viral proteins interact with components of the host ER-associated degradation machinery (ERAD). These interactions are crucial for translocation of SV40 into the cytoplasm and infectivity.
- Roger Geiger
- , Daniel Andritschke
- & Ari Helenius
-
Review Article |
 Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress
Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress
- Ira Tabas
- & David Ron
-
News & Views |
 Neuronal transport: myosins pull the ER
Neuronal transport: myosins pull the ER
Whether class V myosins can work as point-to-point transporters in animal cells is highly debated. Myosin-Va is now shown to function as a point-to-point transporter that pulls the endoplasmic reticulum (ER) into dendritic spines, with important consequences for dendritic development and cerebellar motor learning.
- Michael Stiess
- & Frank Bradke
-
Article |
 Myosin-Va transports the endoplasmic reticulum into the dendritic spines of Purkinje neurons
Myosin-Va transports the endoplasmic reticulum into the dendritic spines of Purkinje neurons
The role of actin-based motors and the mode of endoplasmic reticulum (ER) transport into spines had remained unclear. Myosin-Va is now shown to act as a point-to-point ER transporter into dendritic spines.
- Wolfgang Wagner
- , Stephan D. Brenowitz
- & John A. Hammer III

 Unfolding emergency calls stress granules to the ER
Unfolding emergency calls stress granules to the ER Mitochondrial fission: ER marks the spot
Mitochondrial fission: ER marks the spot