« Prev Next »

Plants must invest energy and nutrients to grow stems, leaves, roots, and reproductive tissues. When herbivores eat these tissues, plants must cope with decreased abilities to convert nutrients and energy into offspring. Therefore, natural selection favors plant traits that limit the negative effects of herbivores. Some plants have evolved tolerance to herbivory, growing compensatory tissues so rapidly that reproduction can sometimes increase with light damage (Strauss & Agrawal 1999, Stowe et al. 2000). By comparison, other plants have evolved traits that reduce consumption by herbivores, or resistance. Because herbivores rely on plants for food, natural selection favors herbivores that overcome plant resistance, thus prompting plants to evolve new forms of resistance. This evolutionary race between plants and herbivores (i.e., ‘coevolution') has resulted in a wide array of resistance traits in terrestrial plants (Erlich & Raven 1964, Agrawal et al. 2009) including reduced apparency to herbivores and structural, chemical, and indirect defenses. These resistance traits are described in greater detail below.
Apparency
Some plants avoid herbivory by decreasing their apparency or ‘hiding' from herbivores in spatial or temporal refuges. Spatial refuges are areas physically inaccessible to or hidden from herbivores as when plants grow on cliff ledges and plateaus in the case of geologic refuges (Figure 1). Other spatial refuges can include areas above or below the reach of herbivores as when grazing stimulates plant growth near the ground for non-woody plants or above the browse line for trees and shrubs (Hayes & Holl 2003; Figure 1). Alternatively, some plants take advantage of temporal refuges by growing or flowering when herbivores are rare or inactive.
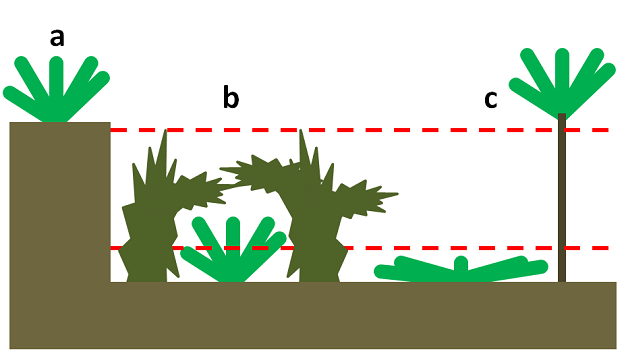
Biotic refuges, sometimes referred to as associational resistance, occur whenever a plant reduces the chances that herbivores find and consume neighboring plants of a different species. Associational resistance can occur as the result of several processes. First, some plants can release volatile organic chemicals (VOCs) that mask or overpower VOCs released by another plant that would normally attract herbivores (Jactel et al. 2011). Alternatively, plants may release VOCs that repel herbivores, though evidence of this effect is limited (Hambäck et al. 2000, Agrawal et al. 2006). Second, plants may provide an alternate food source that draws herbivores away from another plant (Russell & Louda 2005), an effect referred to as the alternate host hypothesis (Atsatt & O'Dowd 1976) in which associational resistance for one plant species is paired with associational susceptibility in another plant species. From the perspective of plants that serve as the alternate food source, association with the less preferred plant results in associational susceptibility rather than resistance. Finally, plants may alter the physical environment by changing the microclimate (Shepherd 1985) or increasing the structural complexity of an environment so that herbivores are unable to find or access their preferred host. Such complexity may occur when a poorly defended plant is growing under heavy brush (Milchunas & Noy-Meir 2002) or intermixed with similarly colored species (Finch & Collier 2000).
While refuges allow plants to escape some herbivores, small, mobile, or temporally flexible herbivores are not so easily deterred. As an alternative to refuges, some plants use crypsis (i.e., camouflage) to avoid herbivores. For example, the sensitive plant (Mimosa pudica) mimics the appearance of a dead or wilting plant by folding and drooping its leaves when touched (Figure 2a and b). Alternatively, stone plants (Lithops spp.) decrease apparency by, as their name implies, resembling stones (Figure 2c and d).

Structural defenses
Once herbivores find and access a plant, structural defenses can discourage consumption. These structures include spinescence, trichomes, thick leaves, and microscopic sand- and needle-like particles inside plant tissues (Figures 3 and 4).


Spinescence includes evolutionarily modified stems or leaves known as thorns or spines, respectively, or sharp extensions of the epidermis (i.e., the 'skin‘) known as prickles (Figure 3a, b and c). These sharp, pointed extensions can deter large herbivores but are generally less effective against smaller, more maneuverable herbivores like insects. To guard against herbivorous insects, some plants use a layer of plant hairs, or trichomes (Figure 3d). Trichomes are extensions of the epidermis that can prevent insect eggs from sticking to a plant, hinder movement by insects, and limit consumption by large herbivores due to their unpleasant texture. When combined with chemical defenses, trichomes can act as glands that secrete sticky resins or irritating chemicals to reduce grazing by large herbivores. For example, stinging nettle (Urtica dioica) produces trichomes that break easily when handled and inject painful chemicals, much like a syringe, to discourage grazing by large mammals (Lambers et al. 1998).
Plants may further limit herbivory by producing hard, rigid leaves (i.e., sclerophylly) and stems that are difficult to chew (Figure 3e). Leaf toughness and stem strength are bolstered by woody compounds such as cellulose and lignin. These compounds can only be digested with the aid of symbiotic bacteria, which occur, for example, in the guts of cows and termites, and have little to no dietary value. Structural compounds are therefore associated with poor nutritional values, sometimes expressed as large carbon-to-nutrient ratios, that diminish the benefits of eating a plant.
Some plants store non-toxic minerals from the soil, such as silica or calcium, as a form of physical defense. Silica released into the spaces between cells (extracellular spaces) forms stone-like phytoliths (from Greek phuton for plant, and lithos for stone) that increase wear on insect mouthparts or vertebrate teeth (Hanley et al. 2007). Calcium ions may be bound to an organic anion, oxalate, to form crystals in cell walls, vacuoles, and trichomes (Figure 4) that can pierce mouth tissues and cause swelling and irritation as in dumb cane (Dieffenbachia spp.; Mrvos et al. 1991). Both calcium oxalate crystals and silica phytoliths can also form painful, and potentially lethal, kidney stones, increasing the risks of feeding on plants with these structural defenses.
Chemical defenses
Chemical defenses include compounds with chemical properties that directly deter herbivores from feeding on a plant. Organic chemical defenses are produced by plants as secondary metabolites, compounds not directly related to basic metabolic pathways (e.g., photosynthesis or respiration), or by mutualistic fungi known as endophytes. By comparison, elemental defenses are inorganic chemical defenses that must be concentrated from the environment.
Organic chemical defenses include qualitative and quantitative defenses. Qualitative defenses are typically effective and, therefore, produced at low concentrations. Most qualitative defenses are used in tissues that are vulnerable over short time spans, such as young, tender leaves or seeds, and are often recycled when no longer needed. For example, some plants produce cyanide-containing compounds in their seeds that can kill herbivores when consumed in low doses. As the threat of herbivory declines, nitrogen stored in these compounds is recycled for plant growth (Poulton 1990). Although qualitative defenses are typically effective against generalist herbivores, specialists have adapted methods through coevolution to circumvent or even highjack these defenses. For instance, monarch butterfly (Danaus plexippus) caterpillars store qualitative chemical defenses from milkweed (Asclepias spp.) in their bodies to deter predators (Nishida 2002). As an important resource for some specialists, qualitative defenses can attract herbivores, turning the benefits of plant defense into a liability.
In comparison to qualitative defenses, quantitative defenses are generally effective against all herbivores but require larger doses. As a result, these compounds are typically mass produced and are rarely recycled. Condensed tannins are common quantitative defenses that bind to proteins, interfering with digestion and potentially leading to malnutrition (Ayres et al. 1997). Other quantitative defenses include chemicals that cause pain, inflammation, or swelling in the skin or mouth when touched as in the case of stinging nettle (see ‘Structural defenses' above) or poison ivy (Toxicodendron radicans).
Whether a plant relies more heavily on qualitative or quantitative defenses may be influenced by factors such as growth rate (Coley et al. 1985), nutrient availability (Stamp 2003), or how easily it can be found by herbivores (Feeny 1976). Regardless of their mode of operation, organic chemical defenses reduce the incentive for herbivores to feed on a plant and, consequently, the amount of damage sustained.
Humans use organic chemical defenses as common ingredients in food and medicine. For example, caffeine belongs to a class of qualitative defenses that block brain signals relaying drowsiness (Fisone et al. 2004). Uses for defensive and other secondary compounds are studied in the fields of ethnobotany and pharmacology (Fowler 2006, Soetan 2008).
Inorganic, elemental defenses likely evolved as a way for plants to cope with toxic elements inadvertently absorbed from the soil such as nickel, zinc, cadmium, and lead. Many plants avoid poisoning by storing these elements away from cell machinery in cell walls, vacuoles, or trichomes until they are released when a plant dies or is consumed. As these elements are also poisonous to most herbivores, plants that absorb and store toxic elements, referred to as ‘metal hyperaccumulators', benefit from reduced herbivory (Poschenrieder et al. 2006, Boyd 2007, Boyd 2009).
Chemical defenses are not always meant to deter all herbivores. Many plants benefit from interactions with mutualistic herbivores such as pollinators or seed dispersers and have evolved defenses that only target harmful herbivores. For example, chili seeds pass safely through the digestive systems of birds and are dispersed in the droppings but are destroyed when eaten by mammals. Not surprisingly, capsaicin, the compound that gives chilies their hot flavor, affects mammals but not birds (Tewksbury & Nabhan 2001).
Indirect defenses
Instead of directly defending against herbivores, indirect defenses reduce herbivory by increasing the likelihood that herbivores (usually insects) are attacked, removed, or harassed by predators like ants, wasps, and mites (Figure 5). Because indirect defenses rely on a third trophic, or feeding, level in the food web, they are sometimes referred to as tritrophic or biotic defenses. Plants increase predation of herbivores by luring and keeping predators on a plant with food rewards, shelters from harsh conditions, or chemicals signaling prey availability.
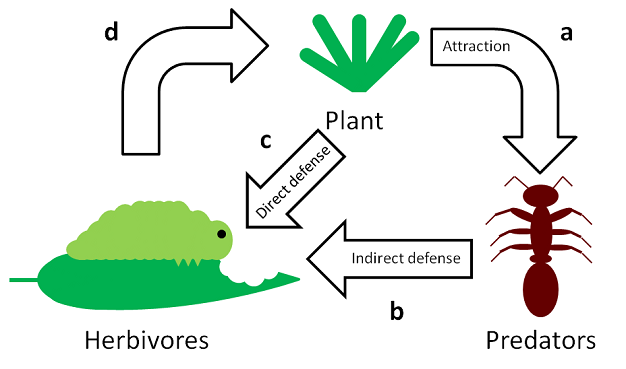
Many plants produce energy-rich food rewards to attract predators, decreasing production when predators or herbivores are absent or inactive (Heil et al. 1997, Wäckers & Bonifay 2004). Food rewards used in plant defense include nectar, produced by extrafloral nectaries (EFNs), and solid food bodies (Figure 6a). Unlike floral nectaries, the primary function of EFNs is to attract predators rather than pollinators. EFNs promote defensive mutualisms ranging from absolute requirements for survival in myrmecophytes (i.e., ‘ant plants') to beneficial but nonessential relationships in a number of other plants (Bentley 1977). Solid food bodies range in form from fruit-like appendages to soft layers of nutritious tissue. These structures contain lipids, carbohydrates, and proteins that represent a substantial investment by the plant. Consequently, food bodies have only been observed on myrmecophytes and are associated with other forms of indirect defense such as domatia (Heil 2008).
Domatia are structures that shelter predators from harsh environmental conditions or other predators. Domatia may range in complexity from shallow crevasses covered with trichomes as in some varieties of avocado (Persea americana; Agrawal 1997) to hollow tissues with multiple chambers and elaborate entrances as in many acacia (Acacia spp.; Figure 6b and c). Although domatia and food rewards may not directly attract predators, they can increase the likelihood that visiting predators will remain on a plant and reduce herbivory.

The only indirect defenses that actively attract predators are volatile organic chemicals (VOCs). These gaseous signals are often released from damaged plant tissues, advertising the presence of potential prey. VOCs can vary with time of attack (e.g., night vs. day) or herbivore identity to attract predators best adapted for a particular herbivore. For example, broad bean plants (Vicia faba) attacked by different species of aphid (Acyrthosiphon pisum and A. fabae) release different VOCs that attract different predators (Powell et al. 1998). Many VOCs also repel herbivores, including adult hawkmoths (Manduca quinquemaculata) that avoid laying eggs on tobacco plants (Nicotiana attenuata) emitting predator-attracting VOCs (Kessler & Baldwin 2001). VOCs, therefore, serve dual defensive purposes by indirectly reducing herbivory via predator attraction and directly deterring herbivores.
Resistance costs
The resistance traits discussed above can be costly in terms of energy, resource, and opportunity costs (Strauss et al. 2002). As plants have limited resources, an increase in either resistance or growth must be balanced by a decrease in the other (Coley et al. 1985), a relationship referred to as a tradeoff. Consequently, fast-growing plant species are typically less resistant to herbivory than slower growing species (Endara & Coley 2011). Tradeoffs may also exist among resistance traits so that a plant cannot maximize all forms of resistance against all possible herbivores. As a result, plants may express different resistance traits that minimize consumption by different herbivores in different places (e.g., Berenbaum & Zangerl 2006) or at different times (e.g., Wäckers et al. 2004).
To avoid negative effects of tradeoffs, many plants maintain low baseline, or constitutive, defensive levels until stimulated, or induced, by herbivore damage, VOCs, light availability, or day length (Zangerl 2003, Wäckers & Bonifay 2004, Conrath et al. 2006, Radhika et al. 2008). In fact, many direct (Chen 2008) and indirect (Heil 2008) resistance traits are only expressed following induction by some stimulus. In this way plants are able to avoid investing resource in unneeded resistance traits, thus allowing more resources for growth and reproduction.
Conclusion
The goal of this article is to briefly introduce various forms of plant resistance against herbivores; however, resistance traits against pathogens such as fungi, viruses, and nematodes also exist. Despite some overlap between herbivore and pathogen resistance, a number of uniquely anti-pathogenic defenses exist (Lambers et al. 1998, Anderson et al. 2010).
Natural selection from herbivory has prompted plants to evolve a wide array of resistance traits to reduce losses from herbivory. Plants avoid herbivory by hiding, building structural barriers, producing and acquiring chemical toxins, and recruiting predatory ‘bodyguards'. Thus, plants are not the helpless victims of herbivory but defend themselves against the loss of resources and energy, allowing for greater investment in reproduction and survival.
Glossary
apparency: Ease by which an object, in this case a plant, is found
associational resistance: Defensive benefits acquired by a plant due to close spatial association with a different plant species
associational susceptibility: Increased herbivory on a plant as a result of close spatial association with a different plant species
chemical defense: Compounds and elements with chemical properties that directly deter or limit herbivory
coevolution: Paired, reciprocal evolution between two closely interacting species in response to evolutionary change of either species
crypsis: Morphological adaptations used to specifically avoid herbivores/predators; a form of camouflage
domatia: Plant structures that shelter predators from environmental conditions or predation (from the Latin domus-‘house')
elemental defense: Toxic elements absorbed from the soil and stored in plant vacuoles, cell walls, and trichomes that reduce herbivory
endophytes: Fungi that live in the intracellular space of plant tissues, some of which produce chemical toxins used to deter herbivores
extrafloral nectaries (EFNS): Nectar-secreting structures that are not directly related to reproduction; typically used in indirect defense
food bodies: Dense multicellular structures, composed of carbohydrate, lipid, and protein, offered as food rewards to predatory mutualists
generalist: An organism with a wide resource base; as herbivores, generalists are not well adapted to defenses on any one plant
indirect defense: Traits that reduce herbivory without directly affecting herbivores, usually by attracting predators
inorganic chemical defense: Chemical defense not produced by a living organism that must be acquired from the environment (see elemental defense)
metal hyperaccumulators: Plants that actively acquire and store toxic elements in their tissues in lethal concentrations
microclimate: Climatic conditions that may vary at small geographical scales (typically <1 m), such as temperature, humidity, etc.
mutualism: Interaction between two organisms in which both organisms benefit
myrmecophytes: Plants that are nearly or completely reliant on predatory, mutualistic ants for survival (from the Greek murmēx-‘ant', phuton-‘plant')
organic chemical defense: Chemical defense produced by a living organism, in this case a plant or its mutualist
qualitative defense: Chemical defenses produced in low concentrations that have similar effects regardless of concentration
quantitative defense: Chemical defenses typically produced in high concentrations that increase in effect with concentration
refuge: A point in space (i.e., spatial refuge) or time (i.e., temporal refuge) where plants are inaccessible to herbivores
resistance: Plant strategy in which the negative effects of herbivores on plant fitness are reduced by preventing or limiting herbivory
sclerophylly: Hard or tough leaves; used as a form of structural defense (from the Greek sklēros-‘hard', phullon-‘leaf')
secondary metabolites: Chemical compounds produced by a plant that are not involved in basic metabolic processes
specialist: An organism with a narrow resource base. For herbivores, specialists are well adapted to defenses on one or a few plants.
spinescence: Collective term for pointed, prong-like extensions, including thorns, spines, and prickles
structural defense: Physical structures that reduce the amount of damage a plant receives from herbivores
tolerance: Plant strategy in which the negative effects of herbivores are reduced by increasing growth rates after herbivory
tradeoff: A negative relationship between two traits owing to limited resources so that one trait must decrease as the other increases
trichomes: Thin, hair-like extensions of the epidermis
volatile organica chemicals (VOCS): Chemical signals released from damaged plant tissues as a gas; VOCs can be used to attract predators or repel herbivores
References and Recommended Reading
Introduction/General
Agrawal, A. A et al. Evidence for adaptive radiation from a phylogenetic study of plant defenses. Proceedings of the National Academy of Sciences (USA) 106, 18067-18072 (2009).
Ehrlich, P. R. & Raven, P. H. Butterflies and plants: A study in coevolution. Evolution 18, 586-608 (1964).
Stowe, K. A. et al. 2000. The evolutionary ecology of tolerance to consumer damage. Annual Review of Ecology and Systematics 31, 565-595 (2000).
Strauss, S. Y. & Agrawal, A. A. 1999. The ecology and evolution of plant tolerance to herbivory. Trends in Ecology & Evolution 14, 179-185 (1999).
Apparency
Agrawal, A. A. et al. Community heterogeneity and the evolution of interactions between plants and insect herbivores. Quarterly Review of Biology 81, 349-376 (2006).
Atsatt, P. R. & O'Dowd, D. J. Plant defense guilds. Science 193, 24-29 (1976).
Finch, S. & Collier, R. H. Host-plant selection by insects - a theory based on 'appropriate/inappropriate landings' by pest insects of cruciferous plants. Entomologia Experimentalis et Applicata 96, 91-102 (2000).
Hämback, P. A. & Beckerman, A. P. Herbivory and plant resource competition: a review of two interacting interactions. Oikos 101, 26-37 (2003).
Hämback, P. A. et al. Associational resistance: Insect damage to purple loosestrife reduced in thickets of sweet gale. Ecology 81, 1784-1794 (2000).
Hayes, G. F. & Holl, K. D. Cattle grazing impacts on annual forbs and vegetation composition of mesic grasslands in California. Conservation Biology 17, 1694-1702 (2003).
Jactel, H. et al. Non-host volatiles mediate associational resistance to the pine processionary moth. Oecologia 166, 703-711 (2011).
Milchunas, D. G. & Noy-Meir, I. Grazing refuges, external avoidance of herbivory and plant diversity. Oikos 99, 113-130 (2002).
Russell, F. L. & Louda, S. M. Indirect interaction between two native thistles mediated by an invasive exotic floral herbivore. Oecologia 146, 373-384 (2005).
Shepherd, R. C.H. The present status of St. John's wort (Hypericum perforatum L.) and its biological control agents in Victoria, Australia. Agriculture, Ecosystems & Environment 12, 141-149 (1985).
Structural defenses
Hanley, M. E. et al. Plant structural traits and their role in anti-herbivore defence. Perspectives in Plant Ecology Evolution and Systematics 8, 157-178 (2007).
Kahn, S. R [ed.]. Calcium oxalate in biological systems., Boca Raton, FL: CRC Press 1995.
Lambers, H. et al. Plant physiological ecology. New York, NY: Springer Science, 1998.
Massey, F. P. & Hartley, S. E. Physical defences wear you down: progressive and irreversible impacts of silica on insect herbivores. Journal of Animal Ecology 78, 281-291 (2009).
Mrvos, R. et al. Philodendron/Dieffenbachia ingestions: Are they a problem? Clinical Toxicology 29, 485-491 (1991).
Turner, I. M. Sclerophylly: Primarily protective? Functional Ecology 8, 669-675 (1994).
Chemical defenses
Ayres, M. P. et al. Diversity of structure and antiherbivore activity in condensed tannins. Ecology 78, 1696-1712 (1997).
Boyd, R. S. The defense hypothesis of elemental hyperaccumulation: status, challenges and new directions. Plant and Soil 293, 153-176 (2007).
Boyd, R. S. High-nickel insects and nickel hyperaccumulator plants: A review. Insect Science 16, 19-31 (2009).
Coley, P. D. et al. Resource availability and plant antiherbivore defense. Science 230, 895-899 (1985).
Feeny, P. P. 1976. "Plant apparency and chemical defense," in Recent Advances in Phytochemistry, eds. by Wallace, J. W. & Mansell, R. L. (New York: Plenum Press, 1976) 1-40.
Fisone, G. et al. Caffeine as a psychomotor stimulant: mechanism of action. Cellular and Molecular Life Sciences 61, 857-872 (2004).
Hartley, S. E. & Gange, A. C. Impacts of plant symbiotic fungi on insect herbivores: mutualism in a multitrophic context. Annual Review of Entomology 54, 323-342 (2009).
Fowler, M. W. Plants, medicines and man. Journal of the Science of Food and Agriculture 86, 1797-1804 (2006).
Kramer, U. Metal Hyperaccumulation. Annual Review of Plant Biology 61, 517-534 (2010).
Nishida, R. Sequestration of defensive substances from plants by Lepidoptera. Annual Review of Entomology 47, 57-92 (2002).
Poschenrieder, C. et al. Can metals defend plants against biotic stress? Trends in Plant Science 11, 288-295 (2006).
Poulton, J. E. Cyanogenesis in plants. Plant Physiology 94, 401-405 (1990).
Tewksbury, J. J. & Nabhan, G. P. Directed deterrence by capsaicin in chillies. Nature 412, 403-404 (2001).
Soetan, K. O. Pharmacological and other beneficial effects of anti-nutritional factors in plants: A review. African Journal of Biotechnology 7, 4713-4721 (2008).
Stamp, N. Out of the quagmire of plant defense hypotheses. Quarterly Review of Biology 78, 23-55 (2003).
Indirect defenses
Agrawal, A. A. Do leaf domatia mediate a plant-mite mutualism? An experimental test of the effects on predators and herbivores. Ecological Entomology 22, 371-376 (1997).
Bentley, B. L. Extrafloral nectaries and protection by pugnacious bodyguards. Annual Review of Ecology & Systematics 8, 407-427 (1977).
Bronstein, J. L. et al. The evolution of plant-insect mutualisms. New Phytologist 172, 412-428 (2006).
Heil, M. Indirect defence via tritrophic interactions. New Phytologist 178, 41-61 (2008).
Heil, M. et al. Food body production in Macaranga triloba (Euphorbiaceae): a plant investment in anti-herbivore defence via symbiotic ant partners. Journal of Ecology 85, 847-861 (1997).
Kessler, A. & Baldwin, I. T. Defensive function of herbivore-induced plant volatile emissions in nature. Science 291, 2141-2144 (2001).
Powell, W. et al. Strategies involved in the location of hosts by the parasitoid Aphidius ervi Haliday (Hymenoptera : Braconidae : Aphidiinae). Biological Control 11, 104-112 (1998).
Romero, G. Q. & Benson, W. W. Biotic interactions of mites, plants and leaf domatia. Current Opinion in Plant Biology 8, 436-440 (2005).
Wäckers, F. L. & Bonifay, C. How to be sweet? Extrafloral nectar allocation by Gossypium hirsutum fits optimal defense theory predictions. Ecology 85, 1512-1518 (2004).
Resistance costs
Berenbaum, M. R. & Zangerl, A. R. . Parsnip webworms and host plants at home and abroad: Trophic complexity in a geographic mosaic. Ecology 87, 3070-3081 (2006).
Chen, M. S. Inducible direct plant defense against insect herbivores: A review. Insect Science 15, 101-114 (2008).
Coley, P. D. et al. Resource availability and plant antiherbivore defense. Science 230, 895-899 (1985).
Conrath, U. et al. 2006. Priming: Getting ready for battle. Molecular plant-microbe interactions 19, 1062-1071.
Endara, M. J. & Coley, P. D. The resource availability hypothesis revisited: a meta-analysis. Functional Ecology 25, 389-398 (2011).
Radhika, V. et al. Testing the optimal defence hypothesis for two indirect defences: extrafloral nectar and volatile organic compounds. Planta 228, 449-457 (2008).
Strauss, S. Y. et al. Direct and ecological costs of resistance to herbivory. Trends in Ecology & Evolution 17, 278-285 (2002).
Wäckers, F. L. & Bonifay, C. How to be sweet? Extrafloral nectar allocation by Gossypium hirsutum fits optimal defense theory predictions. Ecology 85, 1512-1518 (2004).
Zangerl, A. R. Evolution of induced plant responses to herbivores. Basic and Applied Ecology 4, 91-103 (2003).
Conclusion
Anderson, J. P. et al. Plants versus pathogens: An evolutionary arms race. Functional Plant Biology 37, 499-512 (2010).
Lambers, H. et al. Plant physiological ecology. New York, NY: Springer Science, 1998.






























