Abstract
The molecular biology revolution coupled with the development of monoclonal antibody technology enabled remarkable progress in rheumatology therapy, comprising an array of highly effective biologic agents. With advances in understanding of the molecular nature of immune cell receptors came elucidation of intracellular signalling pathways downstream of these receptors. These discoveries raise the question of whether selective targeting of key intracellular factors with small molecules would add to the rheumatologic armamentarium. In this Review, we discuss several examples of this therapeutic strategy that seem to be successful, and consider their implications for the future of immune-targeted treatments. We focus on kinase inhibitors, primarily those targeting Janus kinase family members and spleen tyrosine kinase, given their advanced status in clinical development and application. We also summarize other targets involved in signalling pathways that might offer promise for therapeutic intervention in the future.
Key Points
-
Biologic agents have revolutionized the therapy of rheumatoid arthritis (RA); nevertheless, not all patients achieve remission and many exhibit only partial responses
-
Advances in our understanding of signal transduction by key immunological receptors offer numerous opportunities for devising new oral, targeted therapies
-
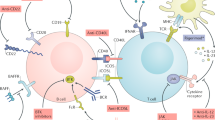
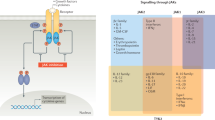
>60 cytokines signal via the Janus kinase (JAK) and signal transducer and activator of transcription (STAT) pathways; genetic evidence has established that these factors are essential to cytokine signalling
-
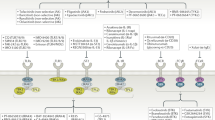
JAK inhibitors (jakinibs) have now been approved for the treatment of RA and other diseases, and various jakinibs are being developed and tested in a range of autoimmune diseases
-
Spleen tyrosine kinase (SYK) and Bruton tyrosine kinase (BTK) are critical kinases downstream of key immunological receptors; inhibitors of these proteins are being tested in RA and other diseases
-
The human kinome comprises 518 kinases; therefore, no shortage of potential new therapeutic targets exists
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Leonard, W. J. & O'Shea, J. J. JAKs and STATs: biological implications. Ann. Rev. Immunol. 16, 293–322 (1998).
O'Shea, J. J., Holland, S. M. & Staudt, L. M. JAKs and STATs in immunity, immunodeficiency and cancer. N. Engl. J. Med. 368, 161–170 (2013).
Ghoreschi, K. et al. Modulation of innate and adaptive immune responses by tofacitinib (CP-690,550). J. Immunol. 186, 4234–4243 (2011).
Karaman, M. W. et al. A quantitative analysis of kinase inhibitor selectivity. Nat. Biotechnol. 26, 127–132 (2008).
Changelian, P. S. et al. Prevention of organ allograft rejection by a specific Janus kinase 3 inhibitor. Science 302, 875–878 (2003).
Fleischmann, R. et al. Placebo-controlled trial of tofacitinib monotherapy in rheumatoid arthritis. N. Engl. J. Med. 367, 495–507 (2012).
van Vollenhoven, R. F. et al. Tofacitinib or adalimumab versus placebo in rheumatoid arthritis. N. Engl. J. Med. 367, 508–519 (2012).
Kremer, J. M. et al. A phase 2B dose-ranging study of the oral JAK inhibitor tofacitinib (CP-690,550) versus placebo in combination with background methotrexate in patients with active rheumatoid arthritis and inadequate response to methotrexate alone. Arthritis Rheum. 64, 970–981 (2012).
Kremer, J. M. et al. The safety and efficacy of a JAK inhibitor in patients with active rheumatoid arthritis: results of a double-blind, placebo-controlled phase IIa trial of three dosage levels of CP-690,550 versus placebo. Arthritis Rheum. 60, 1895–1905 (2009).
Tanaka, Y., Suzuki, M., Nakamura, H., Toyoizumi, S. & Zwillich, S. H. Phase II study of tofacitinib (CP-690,550) combined with methotrexate in patients with rheumatoid arthritis and an inadequate response to methotrexate. Arthritis Care Res. (Hoboken) 63, 1150–1158 (2011).
Burmester, G. R. et al. Tofacitinib (CP-690,550) in combination with methotrexate in patients with active rheumatoid arthritis with an inadequate response to tumour necrosis factor inhibitors: a randomised phase 3 trial. Lancet http://dx.doi.org/10.1016/S0140–6736(12)61424-X.
Sandborn, W. J. et al. Tofacitinib, an oral Janus kinase inhibitor, in active ulcerative colitis. N. Engl. J. Med. 367, 616–624 (2012).
Papp, K. A. et al. Efficacy and safety of tofacitinib, an oral Janus kinase inhibitor, in the treatment of psoriasis: a phase 2b randomized placebo-controlled dose-ranging study. Br. J. Dermatol. 167, 668–677 (2012).
Vincenti, F. et al. Randomized phase 2b trial of tofacitinib (CP-690,550) in de novo kidney transplant patients: efficacy, renal function and safety at 1 year. Am. J. Transplant. 12, 2446–2456 (2012).
Fleischmann, R. et al. Phase IIb dose-ranging study of the oral JAK inhibitor tofacitinib (CP-690,550) or adalimumab monotherapy versus placebo in patients with active rheumatoid arthritis with an inadequate response to disease-modifying antirheumatic drugs. Arthritis Rheum. 64, 617–629 (2012).
van der Heijde, D. et al. Tofacitinib, an oral Janus kinase inhibitor, in combination with methotrexate reduced the progression of structural damage in patients with rheumatoid arthritis: year 2 efficacy and safety results from a 24-month phase 3 study [abstract 1277]. Arthritis Rheum. 64 (Suppl.), S546 (2012).
Tefferi, A. JAK inhibitors for myeloproliferative neoplasms: clarifying facts from myths. Blood 119, 2721–2730 (2012).
Verstovsek, S. Ruxolitinib: the first agent approved for myelofibrosis. Clin. Adv. Hematol. Oncol. 10, 111–113 (2012).
Harrison, C. et al. JAK inhibition with ruxolitinib versus best available therapy for myelofibrosis. N. Engl. J. Med. 366, 787–798 (2012).
Greenwald, M. W. et al. A randomized dose-ranging, placebo-controlled study of INCB028050, a selective JAK1 and JAK2 inhibitor in subjects with active rheumatoid arthritis [abstract 2172]. Arthritis Rheum. 62 (Suppl.), S911 (2010).
Smolen, J. S. et al. 12- and 24-week patient-reported outcomes from a phase 2b dose-ranging study of baricitinib, an oral Janus kinase 1/Janus kinase 2 inhibitor, in combination with traditional disease-modifying antirheumatic drugs in patients with rheumatoid arthritis [abstract 490]. Arthritis Rheum. 64 (Suppl.), S214 (2012).
Genovese, M. C. et al. 24-week results of a blinded phase 2b dose-ranging study of baricitinib, an oral Janus kinase 1/Januse kinase 2 inhibitor, in combination with traditional disease modifying antirheumatic drugs in patients with rheumatoid arthritis [abstract 2487]. Arthritis Rheum. 64 (Suppl.), S1049–S1050 (2012).
Peterfy, C. G. et al. Magnetic resonance imaging substudy in a phase 2b dose-ranging study of baricitinib, an oral Janus kinase 1/Janus kinase 2 inhibitor, in combination with traditional disease-modifying antirheumatic drugs in patients with rheumatoid arthritis [abstract 2488]. Arthritis Rheum. 64 (Suppl.), S1050–S1051 (2012).
O'Shea, J. J. & Plenge, R. JAK and STAT signaling molecules in immunoregulation and immune-mediated disease. Immunity 36, 542–550 (2012).
Conklyn, M., Andresen, C., Changelian, P. & Kudlacz, E. The JAK3 inhibitor CP-690550 selectively reduces NK and CD8+ cell numbers in cynomolgus monkey blood following chronic oral dosing. J. Leukoc. Biol. 76, 1248–1255 (2004).
Paniagua, R. et al. Effects of JAK3 inhibition with CP-690,550 on immune cell populations and their functions in nonhuman primate recipients of kidney allografts. Transplantation 80, 1283–1292 (2005).
Maeshima, K. et al. A JAK inhibitor tofacitinib regulates synovitis through inhibition of IFN-γ and IL-17 production by human CD4+ T cells. Arthritis Rheum. 64, 1790–1798 (2012).
Labranche, T. P. et al. JAK inhibition with tofacitinib suppresses arthritic joint structural damage through decreased RANKL production. Arthritis Rheum. 64, 3531–3542 (2012).
Thomis, D. C., Gurniak, C. B., Tivol, E., Sharpe, A. H. & Berg, L. J. Defects in B lymphocyte maturation and T lymphocyte activation in mice lacking JAK3. Science 270, 794–797 (1995).
Macchi, P. et al. Mutations of JAK-3 gene in patients with autosomal severe combined immune deficiency (SCID). Nature 377, 65–68 (1995).
Russell, S. M. et al. Mutation of JAK3 in a patient with SCID: essential role of JAK3 in lymphoid development. Science 270, 797–800 (1995).
Nosaka, T. et al. Defective lymphoid development in mice lacking Jak3. Science 270, 800–802 (1995).
Rodig, S. J. et al. Disruption of the JAK1 gene demonstrates obligatory and nonredundant roles of the JAKs in cytokine-induced biologic responses. Cell 93, 373–383 (1998).
US National Library of Medicine. A study to evaluate both the efficacy and safety profile of CP-690,550 in patients with moderately to severely active ulcerative colitis (OCTAVE). ClinicalTrials.gov[online], (2012).
US National Library of Medicine. A study evaluating the efficacy and safety of CP-690,550 in patients with moderate to severe ulcerative colitis (OCTAVE). ClinicalTrials.gov[online], (2012).
US National Library of Medicine. A study of oral CP-690550 as a maintenance therapy for ulcerative colitis (OCTAVE). ClinicalTrials.gov[online], (2012).
US National Library of Medicine. Study of the mechanism of action of CP-690,550 in the skin of subjects with moderate to severe chronic plaque psoriasis. ClinicalTrials.gov[online], (2012).
US National Library of Medicine. Pharmacokinetics of CP-690,550 in pediatric patients with juvenile idiopathic arthritis (JIA). ClinicalTrials.gov[online], (2012).
US National Library of Medicine. Long-term safety study of CP-690,550 in patients with juvenile idiopathic arthritis. ClinicalTrials.gov[online], (2013).
US National Library of Medicine. A one-year study to evaluate the effects and safety of CP-690,550 in patients with moderate to severe chronic plaque psoriasis. ClinicalTrials.gov[online], (2012).
Stump, K. L. et al. A highly selective, orally active inhibitor of Janus kinase 2, CEP-33779, ablates disease in two mouse models of rheumatoid arthritis. Arthritis Res. Ther. 13, R68 (2011).
Kawasaki, M. et al. Possible role of the JAK/STAT pathways in the regulation of T cell-interferon related genes in systemic lupus erythematosus. Lupus 20, 1231–1239 (2011).
Wang, S. et al. Jak/STAT signaling is involved in the inflammatory infiltration of the kidneys in MRL/lpr mice. Lupus 19, 1171–1180 (2010).
Kudlacz, E., Conklyn, M., Andresen, C., Whitney-Pickett, C. & Changelian, P. The JAK-3 inhibitor CP-690550 is a potent anti-inflammatory agent in a murine model of pulmonary eosinophilia. Eur. J. Pharmacol. 582, 154–161 (2008).
Fleischmann, R. Novel small-molecular therapeutics for rheumatoid arthritis. Curr. Opin. Rheumatol. 24, 335–341 (2012).
US National Library of Medicine. A 12-week study of 4 doses of VX-509 in subjects with active rheumatoid arthritis. ClinicalTrials.gov[online], (2012).
US National Library of Medicine. A magnetic resonance imaging study and arthroscopic biopsy substudy in subjects with active rheumatoid arthritis receiving VX-509, an oral JAK3 inhibitor. ClinicalTrials.gov[online], (2013).
Vanhoutte, F., Mazur, M., Van der Aa, A., Wigerinck, P. & van 't Klooster, G. Selective JAK1 inhibition in the treatment of rheumatoid arthritis: proof of concept with GLPG0634 [abstract 2489]. Arthritis Rheum. 64 (Suppl.), S1051 (2012).
Namour, F. et al. Once daily high dose regimens of GLPG0634 in healthy volunteers are safe and provide continuous inhibition of JAK1 but not JAK2 [abstract 1331]. Arthritis Rheum. 64 (Suppl.), S573 (2012).
Samelson, L. E. Signal transduction mediated by the T cell antigen receptor: the role of adapter proteins. Annu. Rev. Immunol. 20, 371–394 (2002).
Chu, D. H., Morita, C. T. & Weiss, A. The Syk family of protein tyrosine kinases in T-cell activation and development. Immunol. Rev. 165, 167–180 (1998).
Smith-Garvin, J. E., Koretzky, G. A. & Jordan, M. S. T cell activation. Annu. Rev. Immunol. 27, 591–619 (2009).
Kurosaki, T. Genetic analysis of B cell antigen receptor signaling. Annu. Rev. Immunol. 17, 555–592 (1999).
Gilfillan, A. M. & Rivera, J. The tyrosine kinase network regulating mast cell activation. Immunol. Rev. 228, 149–169 (2009).
Chan, A. C., Irving, B. A. & Weiss, A. New insights into T-cell antigen receptor structure and signal transduction. Curr. Opin. Immunol. 4, 246–251 (1992).
Chan, A. C. et al. ZAP-70 deficiency in an autosomal recessive form of severe combined immunodeficiency. Science 264, 1599–1601 (1994).
Cambier, J. C., Pleiman, C. M. & Clark, M. R. Signal transduction by the B cell antigen receptor and its coreceptors. Annu. Rev. Immunol. 12, 457–486, (1994).
Mócsai, A., Ruland, J. & Tybulewicz, V. L. The SYK tyrosine kinase: a crucial player in diverse biological functions. Nat. Rev. Immunol. 10, 387–402 (2010).
Turner, M. et al. Perinatal lethality and blocked B-cell development in mice lacking the tyrosine kinase Syk. Nature 378, 298–302 (1995).
Abtahian, F. et al. Regulation of blood and lymphatic vascular separation by signaling proteins SLP-76 and Syk. Science 299, 247–251 (2003).
Pine, P. R. et al. Inflammation and bone erosion are suppressed in models of rheumatoid arthritis following treatment with a novel Syk inhibitor. Clin. Immunol. 124, 244–257 (2007).
Weinblatt, M. E. et al. Treatment of rheumatoid arthritis with a SYK kinase inhibitor: a twelve-week, randomized, placebo-controlled trial. Arthritis Rheum. 58, 3309–3318 (2008).
Genovese, M. C. et al. An oral SYK kinase inhibitor in the treatment of rheumatoid arthritis: a three-month randomized, placebo-controlled, phase II study in patients with active rheumatoid arthritis that did not respond to biologic agents. Arthritis Rheum. 63, 337–345 (2011).
Barr, P. M. et al. Syk inhibition with fostamatinib leads to transitional B lymphocyte depletion. Clin. Immunol. 142, 237–242 (2012).
US National Library of Medicine. Evaluation of long-term safety and effectiveness of fostamatinib in the treatment of rheumatoid arthritis (RA) (OSKIRA-X). ClinicalTrials.gov[online], (2013).
Bahjat, F. R. et al. An orally bioavailable spleen tyrosine kinase inhibitor delays disease progression and prolongs survival in murine lupus. Arthritis Rheum. 58, 1433–1444 (2008).
Deng, G. M., Liu, L., Bahjat, F. R., Pine, P. R. & Tsokos, G. C. Suppression of skin and kidney disease by inhibition of spleen tyrosine kinase in lupus-prone mice. Arthritis Rheum. 62, 2086–2092 (2010).
Friedberg, J. W. et al. Inhibition of SYK with fostamatinib disodium has significant clinical activity in non-Hodgkin lymphoma and chronic lymphocytic leukemia. Blood 115, 2578–2585 (2010).
Gomez-Rodriguez, J., Kraus, Z. J. & Schwartzberg, P. L. Tec family kinases Itk and Rlk/Txk in T lymphocytes: cross-regulation of cytokine production and T-cell fates. FEBS J. 278, 1980–1989 (2011).
Rawlings, D. J. & Witte, O. N. Bruton's tyrosine kinase is a key regulator in B-cell development. Immunol. Rev. 138, 105–119 (1994).
Thomas, J. D. et al. Colocalization of X-linked agammaglobulinemia and X-linked immunodeficiency genes. Science 261, 355–358 (1993).
Horwood, N. J., Urbaniak, A. M. & Danks, L. Tec family kinases in inflammation and disease. Int. Rev. Immunol. 31, 87–103 (2012).
Ellmeier, W., Abramova, A. & Schebesta, A. Tec family kinases: regulation of FcεRI-mediated mast-cell activation. FEBS J. 278, 1990–2000 (2011).
Chang, B. Y. et al. The Bruton tyrosine kinase inhibitor PCI-32765 ameliorates autoimmune arthritis by inhibition of multiple effector cells. Arthritis Res. Ther. 13, R115 (2011).
Honigberg, L. A. et al. The Bruton tyrosine kinase inhibitor PCI-32765 blocks B-cell activation and is efficacious in models of autoimmune disease and B-cell malignancy. Proc. Natl Acad. Sci. USA 107, 13075–13080 (2010).
Weiss, A., Irving, B. A., Tan, L. K. & Koretzky, G. A. Signal transduction by the T cell antigen receptor. Semin. Immunol. 3, 313–324 (1991).
Yablonski, D. & Weiss, A. Mechanisms of signaling by the hematopoietic-specific adaptor proteins, SLP-76 and LAT and their B cell counterpart, BLNK/SLP-65. Adv. Immunol. 79, 93–128 (2001).
Lewis, R. S. Calcium signaling mechanisms in T lymphocytes. Annu. Rev. Immunol. 19, 497–521 (2001).
Altman, A. & Villalba, M. Protein kinase C-θ (PKCθ): it's all about location, location, location. Immunol. Rev. 192, 53–63 (2003).
Altman, A. & Kong, K. F. PKCθ: a new target for selective immunosuppression. Expert Rev. Clin. Immunol. 8, 205–208 (2012).
Fuller, T. F. et al. Protein kinase C inhibition ameliorates posttransplantation preservation injury in rat renal transplants. Transplantation 94, 679–686 (2012).
Wu, X., Li, J., Zhu, M., Fletcher, J. A. & Hodi, F. S. Protein kinase C inhibitor AEB071 targets ocular melanoma harboring GNAQ mutations via effects on the PKC/Erk1/2 and PKC/NF-κB pathways. Mol. Cancer Ther. 11, 1905–1914 (2012).
Pagès, F. et al. Binding of phosphatidylinositol-3-OH kinase to CD28 is required for T-cell signalling. Nature 369, 327–329 (1994).
Jones, R. G. et al. CD28-dependent activation of protein kinase B/Akt blocks Fas-mediated apoptosis by preventing death-inducing signaling complex assembly. J. Exp. Med. 196, 335–348 (2002).
Leslie, N. R., Biondi, R. M. & Alessi, D. R. Phosphoinositide-regulated kinases and phosphoinositide phosphatases. Chem. Rev. 101, 2365–2380 (2001).
Edinger, A. L. & Thompson, C. B. Antigen-presenting cells control T cell proliferation by regulating amino acid availability. Proc. Natl Acad. Sci. USA 99, 1107–1109 (2002).
Frauwirth, K. A. et al. The CD28 signaling pathway regulates glucose metabolism. Immunity 16, 769–777 (2002).
Rathmell, J. C., Farkash, E. A., Gao, W. & Thompson, C. B. IL-7 enhances the survival and maintains the size of naive T cells. J. Immunol. 167, 6869–6876 (2001).
Cantley, L. C. & Neel, B. G. New insights into tumor suppression: PTEN suppresses tumor formation by restraining the phosphoinositide 3-kinase/AKT pathway. Proc. Natl Acad. Sci. USA 96, 4240–4245 (1999).
Beitz, L. O., Fruman, D. A., Kurosaki, T., Cantley, L. C. & Scharenberg, A. M. SYK is upstream of phosphoinositide 3-kinase in B cell receptor signaling. J. Biol. Chem. 274, 32662–32666 (1999).
Edinger, A. L. & Thompson, C. B. Akt maintains cell size and survival by increasing mTOR-dependent nutrient uptake. Mol. Biol. Cell 13, 2276–2288 (2002).
Murgia, M. G., Jordan, S. & Kahan, B. D. The side effect profile of sirolimus: a phase I study in quiescent cyclosporine–prednisone-treated renal transplant patients. Kidney Int. 49, 209–216 (1996).
Kappos, L. et al. The effect of oral temsirolimus on new magnetic resonance imaging scan lesions, brain atrophy, and the number of relapses in multiple sclerosis: results from a randomised, controlled clinical trial [abstract]. J. Neurol. 252 (Suppl. 2), S46 (2005).
Dong, C., Davis, R. J. & Flavell, R. A. MAP kinases in the immune response. Annu. Rev. Immunol. 20, 55–72 (2002).
Rincon, M. MAP-kinase signaling pathways in T cells. Curr. Opin. Immunol. 13, 339–345 (2001).
Josephs, D. H. & Ross, P. J. Sorafenib in hepatocellular carcinoma. Br. J. Hosp. Med. (Lond.) 71, 451–456 (2010).
Govindarajan, R., Adusumilli, J., Baxter, D. L., El-Khoueiry, A. & Harik, S. I. Reversible posterior leukoencephalopathy syndrome induced by RAF kinase inhibitor BAY 43–9006. J. Clin. Oncol. 24, e48 (2006).
Gijtenbeek, J. M., van den Bent, M. J. & Vecht, C. J. Cyclosporine neurotoxicity: a review. J. Neurol. 246, 339–346 (1999).
Ohori, M., Takeuchi, M., Maruki, R., Nakajima, H. & Miyake, H. FR180204, a novel and selective inhibitor of extracellular signal-regulated kinase, ameliorates collagen-induced arthritis in mice. Naunyn Schmiedebergs Arch. Pharmacol. 374, 311–316 (2007).
Su, B. et al. JNK is involved in signal integration during costimulation of T lymphocytes. Cell 77, 727–736 (1994).
Dong, C. et al. Defective T cell differentiation in the absence of Jnk1. Science 282, 2092–2095 (1998).
Yang, D. D. et al. Differentiation of CD4+ T cells to TH1 cells requires MAP kinase JNK2. Immunity 9, 575–585 (1998).
Arbour, N. et al. c-Jun NH2-terminal kinase (JNK)1 and JNK2 signaling pathways have divergent roles in CD8+ T cell-mediated antiviral immunity. J. Exp. Med. 195, 801–810 (2002).
Conze, D. et al. c-Jun NH2-terminal kinase (JNK)1 and JNK2 have distinct roles in CD8+ T cell activation. J. Exp. Med. 195, 811–823 (2002).
Ip, Y. T. & Davis, R. J. Signal transduction by the c-Jun N-terminal kinase (JNK)—from inflammation to development. Curr. Opin. Cell Biol. 10, 205–219 (1998).
Swantek, J. L., Cobb, M. H. & Geppert, T. D. Jun N-terminal kinase/stress-activated protein kinase (JNK/SAPK) is required for lipopolysaccharide stimulation of tumor necrosis factor α (TNF-α) translation: glucocorticoids inhibit TNF-α translation by blocking JNK/SAPK. Mol. Cell. Biol. 17, 6274–6282 (1997).
Ishizuka, T. et al. Mast cell tumor necrosis factor α production is regulated by MEK kinases. Proc. Natl Acad. Sci. USA 94, 6358–6363 (1997).
Manning, A. M. & Davis, R. J. Targeting JNK for therapeutic benefit: from junk to gold? Nat. Rev. Drug Discov. 2, 554–565 (2003).
Lee, J. C. et al. A protein kinase involved in the regulation of inflammatory cytokine biosynthesis. Nature 372, 739–746 (1994).
Dominguez, C., Powers, D. A. & Tamayo, N. p38 MAP kinase inhibitors: many are made, but few are chosen. Curr. Opin. Drug Discov. Devel. 8, 421–430 (2005).
Dyckman, A. J. et al. Discovery of pyrrolo[2,1-f][1,2,4]triazine C6-ketones as potent, orally active p38α MAP kinase inhibitors. Bioorg. Med. Chem. Lett. 21, 4633–4637 (2011).
Cohen, S. & Fleischmann, R. Kinase inhibitors: a new approach to rheumatoid arthritis treatment. Curr. Opin. Rheumatol. 22, 330–335 (2010).
Author information
Authors and Affiliations
Contributions
All authors contributed equally to each stage of the preparation of this manuscript for publication.
Corresponding author
Ethics declarations
Competing interests
J. J. O'Shea and the NIH hold a US patent related to therapeutic targeting of Janus kinases, and has received research support through a longstanding Collaborative Research And Development Agreement with Pfizer. I. B. McInnes has acted as a consultant and received grant/research support from AstraZeneca and Pfizer, and has also acted as a consultant for Vertex. A. Laurence declares no competing interests.
Rights and permissions
About this article
Cite this article
O'Shea, J., Laurence, A. & McInnes, I. Back to the future: oral targeted therapy for RA and other autoimmune diseases. Nat Rev Rheumatol 9, 173–182 (2013). https://doi.org/10.1038/nrrheum.2013.7
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2013.7
This article is cited by
-
Association analysis of Vascular Endothelial Growth Factor-A (VEGF-A) polymorphism in rheumatoid arthritis using computational approaches
Scientific Reports (2023)
-
Arzneitherapie der rheumatoiden Arthritis – wo stehen Biologika und neue synthetische Basistherapeutika heute?
Die Innere Medizin (2023)
-
Cancer vs. SARS-CoV-2 induced inflammation, overlapping functions, and pharmacological targeting
Inflammopharmacology (2021)
-
IgE autoantibodies and autoreactive T cells and their role in children and adults with atopic dermatitis
Clinical and Translational Allergy (2020)
-
Development of JAK inhibitors for the treatment of immune-mediated diseases: kinase-targeted inhibitors and pseudokinase-targeted inhibitors
Archives of Pharmacal Research (2020)