Abstract
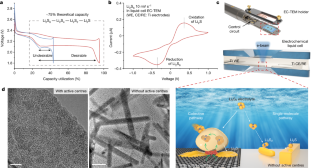
Benefiting from high energy density (2,600 Wh kg−1) and low cost, lithium–sulfur (Li–S) batteries are considered promising candidates for advanced energy-storage systems1,2,3,4. Despite tremendous efforts in suppressing the long-standing shuttle effect of lithium polysulfides5,6,7, understanding of the interfacial reactions of lithium polysulfides at the nanoscale remains elusive. This is mainly because of the limitations of in situ characterization tools in tracing the liquid–solid conversion of unstable lithium polysulfides at high temporal–spatial resolution8,9,10. There is an urgent need to understand the coupled phenomena inside Li–S batteries, specifically, the dynamic distribution, aggregation, deposition and dissolution of lithium polysulfides. Here, by using in situ liquid-cell electrochemical transmission electron microscopy, we directly visualized the transformation of lithium polysulfides over electrode surfaces at the atomic scale. Notably, an unexpected gathering-induced collective charge transfer of lithium polysulfides was captured on the nanocluster active-centre-immobilized surface. It further induced an instantaneous deposition of nonequilibrium Li2S nanocrystals from the dense liquid phase of lithium polysulfides. Without mediation of active centres, the reactions followed a classical single-molecule pathway, lithium polysulfides transforming into Li2S2 and Li2S step by step. Molecular dynamics simulations indicated that the long-range electrostatic interaction between active centres and lithium polysulfides promoted the formation of a dense phase consisting of Li+ and Sn2− (2 < n ≤ 6), and the collective charge transfer in the dense phase was further verified by ab initio molecular dynamics simulations. The collective interfacial reaction pathway unveils a new transformation mechanism and deepens the fundamental understanding of Li–S batteries.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors on reasonable request.
References
Ji, X., Lee, K. T. & Nazar, L. F. A highly ordered nanostructured carbon–sulphur cathode for lithium–sulphur batteries. Nat. Mater. 8, 500–506 (2009).
Zhao, C. et al. A high-energy and long-cycling lithium–sulfur pouch cell via a macroporous catalytic cathode with double-end binding sites. Nat. Nanotechnol. 16, 166–173 (2021).
Peng, L. et al. A fundamental look at electrocatalytic sulfur reduction reaction. Nat. Catal. 3, 762–770 (2020).
Shen, Z. et al. Cation-doped ZnS catalysts for polysulfide conversion in lithium–sulfur batteries. Nat. Catal. 5, 555–563 (2022).
Chen, H. et al. Catalytic materials for lithium-sulfur batteries: mechanisms, design strategies and future perspective. Mater. Today 52, 364–388 (2021).
Liu, Y. et al. Electrolyte solutions design for lithium-sulfur batteries. Joule 5, 2323–2364 (2021).
Liang, Z. et al. Advances in the development of single-atom catalysts for high-energy-density lithium–sulfur batteries. Adv. Mater. 34, e2200102 (2022).
Rehman, S., Pope, M., Tao, S. & McCalla, E. Evaluating the effectiveness of in situ characterization techniques in overcoming mechanistic limitations in lithium–sulfur batteries. Energy Environ. Sci. 15, 1423–1460 (2022).
Ross, F. M. Opportunities and challenges in liquid cell electron microscopy. Science 350, aaa9886 (2015).
Zeng, C. et al. Dynamic investigation of battery materials via advanced visualization: from particle, electrode to cell level. Adv. Mater. 34, e2200777 (2022).
Kong, L. et al. Current-density dependence of Li2S/Li2S2 growth in lithium–sulfur batteries. Energy Environ. Sci. 12, 2976–2982 (2019).
Zhao, G.-X., Ahmed, W. H. Z. & Zhu, F.-L. Nitrogen-sulfur co-doped porous carbon preparation and its application in lithium-sulfur batteries. J. Electrochem. 27, 614–623 (2021).
Pei, F. et al. A two-dimensional porous carbon-modified separator for high-energy-density Li-S batteries. Joule 2, 323–336 (2018).
Tsao, Y. et al. Designing a quinone-based redox mediator to facilitate Li2S oxidation in Li-S batteries. Joule 3, 872–884 (2019).
Nelson, J. et al. In operando X-ray diffraction and transmission X-ray microscopy of lithium sulfur batteries. J. Am. Chem. Soc. 134, 6337–6343 (2012).
Cuisinier, M. et al. Sulfur speciation in Li–S batteries determined by operando X-ray absorption spectroscopy. J. Phys. Chem. Lett. 4, 3227–3232 (2013).
Li, M. et al. Evidence of morphological change in sulfur cathodes upon irradiation by synchrotron X-rays. ACS Energy Lett. 7, 577–582 (2022).
See, K. A. et al. Ab initio structure search and in situ 7Li NMR studies of discharge products in the Li–S battery system. J. Am. Chem. Soc. 136, 16368–16377 (2014).
Chen, J.-J. et al. Conductive Lewis base matrix to recover the missing link of Li2S8 during the sulfur redox cycle in Li–S battery. Chem. Mater. 27, 2048–2055 (2015).
Sun, X. et al. Dislocation-induced stop-and-go kinetics of interfacial transformations. Nature 607, 708–713 (2022).
Frey, H. et al. Dynamic interplay between metal nanoparticles and oxide support under redox conditions. Science 376, 982–987 (2022).
Kim, H. et al. In situ TEM observation of electrochemical lithiation of sulfur confined within inner cylindrical pores of carbon nanotubes. Adv. Energy Mater. 5, 1501306 (2015).
Wang, Z. et al. In situ TEM observations of discharging/charging of solid-state lithium-sulfur batteries at high temperatures. Small 16, 2001899 (2020).
Xu, Z.-L. et al. Visualization of regulated nucleation and growth of lithium sulfides for high energy lithium sulfur batteries. Energy Environ. Sci. 12, 3144–3155 (2019).
Seo, H. K. et al. Direct visualization of lithium polysulfides and their suppression in liquid electrolyte. Nano Lett. 20, 2080–2086 (2020).
Zhang, L. et al. Revealing the electrochemical charging mechanism of nanosized Li2S by in situ and operando X-ray absorption spectroscopy. Nano Lett. 17, 5084–5091 (2017).
Conder, J. et al. Direct observation of lithium polysulfides in lithium–sulfur batteries using operando X-ray diffraction. Nat. Energy 2, 17069 (2017).
Yang, G., Shi, S., Yang, J. & Ma, Y. Insight into the role of Li2S2 in Li–S batteries: a first-principles study. J. Mater. Chem. A 3, 8865–8869 (2015).
Feng, Z. et al. Unravelling the role of Li2S2 in lithium–sulfur batteries: a first principles study of its energetic and electronic properties. J. Power Sources 272, 518–521 (2014).
Paolella, A. et al. Transient existence of crystalline lithium disulfide Li2S2 in a lithium-sulfur battery. J. Power Sources 325, 641–645 (2016).
Wagner, C. Theory of precipitate change by redissolution. Z. Elektrochem. 65, 581–591 (1961).
Lifshitz, I. M. & Slyozov, V. V. The kinetics of precipitation from supersaturated solid solutions. J. Phys. Chem. Solids 19, 35–50 (1961).
Viswanatha, R. & Sarma, D. D. in Nanomaterials Chemistry (eds Rao, C. N. R., Müller, A., Cheetham, A. K.) 139–170 (Wiley, 2007).
Gower, L. B. & Odom, D. J. Deposition of calcium carbonate films by a polymer-induced liquid-precursor (PILP) process. J. Cryst. Growth 210, 719–734 (2000).
Smeets, P. J. et al. Calcium carbonate nucleation driven by ion binding in a biomimetic matrix revealed by in situ electron microscopy. Nat. Mater. 14, 394–399 (2015).
Wallace, A. F. et al. Microscopic evidence for liquid-liquid separation in supersaturated CaCO3 solutions. Science 341, 885–889 (2013).
De Yoreo, J. J. et al. Crystallization by particle attachment in synthetic, biogenic, and geologic environments. Science 349, aaa6760 (2015).
Min, Y. J. et al. The role of interparticle and external forces in nanoparticle assembly. Nat. Mater. 7, 527–538 (2008).
Bishop, K. J., Wilmer, C. E., Soh, S. & Grzybowski, B. A. Nanoscale forces and their uses in self-assembly. Small 5, 1600–1630 (2009).
Müller, A. et al. Organizational forms of matter: an inorganic super fullerene and keplerate based on molybdenum oxide. Angew. Chem. Int. Ed. 37, 3360–3363 (1998).
Hummers, W. S. & Offeman, R. E. Preparation of graphitic oxide. J. Am. Chem. Soc. 80, 1339 (1958).
Cowley, J. M. & Moodie, A. F. The scattering of electrons by atoms and crystals. I. A new theoretical approach. Acta Crystallogr. 10, 609–619 (1957).
Goodman, P. & Moodie, A. F. Numerical evaluations of N-beam wave functions in electron scattering by the multi-slice method. Acta Crystallogr. A30, 280–290 (1974).
Liao, H.-G., Cui, L., Whitelam, S. & Zheng, H. Real-time imaging of Pt3Fe nanorod growth in solution. Science 336, 1011–1014 (2012).
Zheng, H. et al. Nanocrystal diffusion in a liquid thin film observed by in situ transmission electron microscopy. Nano Lett. 9, 2460–2465 (2009).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Grimme, S., Antony, J., Ehrlich, S. & Krieg, H. A consistent and accurate ab initio parametrization of density functional dispersion correction (DFT-D) for the 94 elements H-Pu. J. Chem. Phys. 132, 154104 (2010).
Le, J.-B., Fan, Q.-Y., Li, J.-Q. & Cheng, J. Molecular origin of negative component of Helmholtz capacitance at electrified Pt(111)/water interface. Sci. Adv. 6, eabb1219 (2020).
Hutter, J., Iannuzzi, M., Schiffmann, F. & VandeVondele, J. CP2K: atomistic simulations of condensed matter systems. Wiley Interdiscip. Rev. Comput. Mol. Sci. 4, 15–25 (2014).
Jain, A. et al. Commentary: The Materials Project: a materials genome approach to accelerating materials innovation. APL Mater. 1, 011002 (2013).
Kühne, T. D., Krack, M., Mohamed, F. R. & Parrinello, M. Efficient and accurate Car-Parrinello-like approach to Born-Oppenheimer molecular dynamics. Phys. Rev. Lett. 98, 066401 (2007).
Lan, J., Hutter, J. & Iannuzzi, M. First-principles simulations of an aqueous CO/Pt(111) interface. J. Phys. Chem. C 122, 24068–24076 (2018).
Dodda, L. S., Vaca, I. C. D., Tirado-Rives, J. & Jorgensen, W. L. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117, 1–19 (1995).
Dodda, L. S., Cabeza de Vaca, I., Tirado-Rives, J. & Jorgensen, W. L. LigParGen web server: an automatic OPLS-AA parameter generator for organic ligands. Nucleic Acids Res. 45, W331–W336 (2017).
Zhang, Y. et al. Water-in-salt LiTFSI aqueous electrolytes. 1. Liquid structure from combined molecular dynamics simulation and experimental studies. J. Phys. Chem. B 125, 4501–4513 (2021).
Hu, T. et al. Understanding structural and transport properties of dissolved Li2S8 in ionic liquid electrolytes through molecular dynamics simulations. ChemPhysChem 22, 419–429 (2021).
Abbaspour, M., Akbarzadeh, H. & Zaeifi, S. Thermodynamics, structure, and dynamic properties of nanostructured water confined into B-, N-, and Si-doped graphene surfaces and carbon nanotubes. Ind. Eng. Chem. Res. 59, 9642–9654 (2020).
Bernardes, C. E., Canongia Lopes, J. N. & Minas da Piedade, M. E. All-atom force field for molecular dynamics simulations on organotransition metal solids and liquids. Application to M(CO)n (M = Cr, Fe, Ni, Mo, Ru, or W) compounds. J. Phys. Chem. A 117, 11107–11113 (2013).
Jacobson, D. W. & Thompson, G. B. Revisting Lennard Jones, Morse, and N-M potentials for metals. Comp. Mater. Sci. 205, 111206 (2022).
Hoover, W. G. Canonical dynamics: equilibrium phase-space distributions. Phys. Rev. A 31, 1695–1697 (1985).
Nosé, S. A molecular dynamics method for simulations in the canonical ensemble. Mol. Phys. 52, 255–268 (2006).
Acknowledgements
This work was supported by the Fundamental Research Funds for the Central Universities (20720220009) and National Natural Science Foundation of China (grants nos. 22288102, 22021001, U22A20396, 32101217, 21991151, 2191150 and 91934303). Research at the Argonne National Laboratory was financed by the US Department of Energy (DOE), Vehicle Technologies Office. Support from T. Duong of the US DOE’s Vehicle Technologies Office programme is gratefully acknowledged. This research used resources of the Advanced Photon Source, a US Department of Energy (DOE) Office of Science user facility operated for the DOE Office of Science by Argonne National Laboratory under contract no. DE-AC02-06CH11357. G.-L.X. and K.A. also acknowledge the support from Clean Vehicles, US–China Clean Energy Research Center (CERC-CVC2). This research also employed the resources of the Shanghai Synchrotron Radiation Facility (SSRF, under contract no. 2021-SSRF-ZD-000730, 2022-SSRF-PT-019758 and 2022-SSRF-PT-021637), Hefei National Synchrotron Radiation Laboratory (NSRL-USTC, under contract no. 2021-HLS-PT-004529), and Beijing Synchrotron Radiation Laboratory (under contract no. 2021-BEPC-PT-005760 and 2022-BEPC-PT-006478). S.Z. is thankful for the support from Xiamen University Nanqiang Cultivation Program for Outstanding Doctoral Students. We thank Y. Cheng and M.-S. Wang (College of Materials, Xiamen University) and S. Li and C.-G. Shi (College of Chemistry and Chemical Engineering, Xiamen University) for helpful discussions and suggestions on in situ TEM, Raman and XRD characterizations.
Author information
Authors and Affiliations
Contributions
S.-G.S., H.-G.L., G.-L.X. and K.A. conceived and supervised this work. S.Z. designed the experiments, conducted TEM, materials synthesis, electrochemistry and wrote the paper. J.S. conducted the DFT calculations and MD simulations. J.-F.C., Y.Q., Y.L. and L.H. participated in discussions and data analysis. S.L., G.L. and Y.C. conducted the TEM characterization. F.P. and Q.Z. measured the electrochemical performance. C.Z., I.H. and C.-J.S. conducted XAS characterization. J.D. fabricated electrochemical liquid cells. J.L. and Y.D. conducted EELS characterization. All authors participated in the analysis of experimental data and discussion of the results, as well as in the writing and revision of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature thanks Hyun-Wook Lee, Yongfu Tang and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Configuration of the liquid-cell EC-TEM.
a, Schematic illustration of an electrochemical liquid cell from the top and side views, composed of top/bottom chips with 10-nm SiNx observation windows, 100-nm spacers and samples on electrodes. b, Digital photographs of EC-TEM holder and electrochemical liquid cell. c,d, Schematic process of liquid-cell preparation in an argon-filled glovebox. After liquid-cell assembly, the pre-prepared Li2S6 electrolyte with various concentrations was injected and filled by capillary force, before being sealed with epoxy. e, HRTEM image and corresponding FFT pattern of Li2S@graphene loaded on a single bottom chip with 11.2-nm SiNx layers. The thickness of the SiNx layers was measured using a spectroscopic ellipsometer with a spectral range of 193–1,000 nm. f, Time-series HRTEM images of Li2S@graphene (dashed red frame) showing the high spatial resolution and stable atomic structure at a dose rate of 18,200 e Å−2 s−1.
Extended Data Fig. 2 Crystal structure of the simulated Li2S2.
a, The fixed lattice parameters of tetragonal Li2S2 and hexagonal Li2S2 simulated on the basis of several hexagonal A2B2 structures. b, The corresponding energy comparison between tetragonal Li2S2 and hexagonal Li2S2. The hexagonal Li2S2 structures were constructed on the basis of Li2O2, Na2O2, Na2S2, Na2Se2, K2O2, K2S2 and Rb2S2, arranged from left to right in the dashed blue frame. c, The simulated crystal structures of hexagonal Li2S2 with P63/mmc and \({\rm{P}}\bar{6}{\rm{2m}}\) space groups. d, The crystal structures and simulated electron diffraction of tetragonal Li2S2 along the [001] and [111] directions, respectively. The abnormal systematic extinction of electron diffraction in Fig. 2d, that is, the appearance of (100) and (300), should be attributed to the lattice defect of Li2S2, which led to the diffraction deviating from the Bragg condition, and the secondary diffraction occurred. e, SAED pattern of granular Li2S2 (tetragonal, P42/mnm) along the [111] direction. f, SAED patterns of rod-like Li2S (cubic, \(Fm\bar{3}m\)) along the [100] and [011] directions. g, Elemental weight ratio according to EDS spectrum in Fig. 2f. The red and blue balls represent Li and S atoms, respectively.
Extended Data Fig. 3 Interfacial reactions of LiPSs at −0.1 V without mediation of active centres.
a, Comparison of output current during potentiostatic discharge at −0.1 and −0.5 V in electrochemical liquid cells. The current density was reduced by half at −0.1 V. b, The projection-area variation as a function of time of particles #5 and #6. Error bars represent standard deviations of the measurement. c, Time-series TEM images from Supplementary Video 3 showing two-step Li2S deposition in an electrochemical liquid cell during potentiostatic discharge at −0.1 V. 10 mM Li2S6 was used as the electrolyte. d, Statistics of rod and particle quantity at specific times corresponding to the dashed white frame area in the TEM image (0.6 s). e, Time-series TEM images of typical two-step deposition of Li2S by means of metastable Li2S2 (marked by dotted white lines). TEM images of I–IV correspond to the typical areas at 72.6 s.
Extended Data Fig. 4 In situ EC-TEM studies of Li2S deposition with mediation of active centres.
a, The projection-area variation as a function of time of Mo NCs/N-G and time-series TEM images from Supplementary Video 4. The electrochemical liquid cell was potentiostatically discharged at −0.5 V using 10 mM Li2S6 as the electrolyte. Error bars represent standard deviations of the measurement. b, Time-series TEM images to further observe the dashed red square area of Extended Data Fig. 4a (185 s) under a higher magnification (Supplementary Video 5). The electrochemical liquid cell was further potentiostatically discharged at −0.5 V. Many dark areas (marked with dotted red circles) and deposited particles (marked with white arrows) were observed. c, Time-series TEM images from Supplementary Video 6. The electrochemical liquid cell was potentiostatically discharged at −0.5 V using 100 mM Li2S6 as electrolyte. The imaging area was divided into electron inaccessible/accessible areas. Li2S deposition mainly proceeded on electron-accessible areas of Mo NCs/N-G, whereas no change was observed on the electron-inaccessible area and bare Ti electrode surface. This indicated the high reaction activity of Mo NCs/N-G for electrochemical deposition of Li2S.
Extended Data Fig. 5 Method of HRTEM observation in an electrochemical liquid cell.
a, Schematic illustration of the ‘e-beam bubble generation’ method for HRTEM observation of Li2S nanocrystalline deposited on Mo NCs/N-G nanosheet. Following the electrochemical deposition of Li2S through a potentiostatic discharge at −0.5 V, the e-beam was focused on the bulk electrolyte for the generation of bubbles. Once the bubble area stabilized, the atomic structure of Li2S nanocrystalline can be obtained, with the image resolution being mainly determined by the thickness of SiNx layers. b, TEM images showing different areas of the LiPSs electrolyte, Li2S nanocrystalline deposited on Mo NCs/N-G and the stabilized bubble area. c,d, HRTEM images before and after the removal of the wetting layer of electrolyte. e,f, Annular dark-field scanning TEM (ADF-STEM) image and the corresponding EELS image for relative thickness measurement in liquid cells. g, Typical low-loss EELS of Li2S nanocrystalline (I), Mo NC/N-G nanosheets (II) and SiNx layers (III) as marked in the EELS image. h, Average relative thickness obtained by the log ratio method in Digital Micrograph software. Error bars represent standard deviations from several EELS curves. i, Time-series HRTEM images, corresponding FFT patterns and partial enlarged details of Li2S nanocrystalline deposited on Mo NCs/N-G obtained in an electrochemical liquid cell.
Extended Data Fig. 6 Analysis of in situ EC-TEM studies of redox reactions of LiPSs with mediation of active centres.
a, Time-series TEM images from Supplementary Video 7 showing several droplets in different areas (highlighted in red). b, Motion trajectory of the mass centre of droplet #1 and the corresponding edge contour plots of droplet #1 in Fig. 4b. It included the typical processes of gathering, conversion and crystallization. c, Projection-area variation of droplets #2 and #3 (red) and Mo NCs/N-G (blue) as a function of time during potentiostatic discharge. A power function was used to fit the area variation of droplets #2 and #3 over time (area ∝ tn) according to the Lifshitz–Slyozov–Wagner model, in which n ≥ 1 represents reaction-limited growth. After a rapid decrease in the size of droplets of LiPSs, there was an obvious increase in the projection area of Mo NCs/N-G nanosheet, similar to that in Extended Data Fig. 4a,b, which should be attributed to the deposition of Li2S. Error bars represent standard deviations of the measurement. d, MSD analysis of droplet #1 as a function of time. The two-dimensional diffusion coefficient (D) was calculated to be 4.71 nm2 s−1. The initial point of MSD corresponds to 34.4 s of droplet #1 in Fig. 4b.
Extended Data Fig. 7 Structure characterization of droplet-like dense phase.
a,b, Comparative characterization of the dense phase of LiPSs and Li2S nanocrystalline, including HRTEM images, the corresponding FFT and SAED patterns, HAADF-STEM images, EDS mappings and the corresponding linear elemental analysis. c, TEM and HAADF-STEM images of Mo NCs/N-G nanosheet after the electrochemical deposition in a liquid cell, showing the dense phase of LiPSs and Li2S nanocrystalline from a wider observation region. d, Raw and post-processed HRTEM images using a Gaussian filter. To solve the scattering and artefacts in HRTEM images resulting from amorphous SiNx windows, disordered electrolyte and the contrast transfer function of the electron microscope, a Gaussian filter was applied to suppress high-spatial-frequency noise and enhance the boundary of dense phase areas. e,f, Partial enlarged details of the HAADF-STEM image and the corresponding linear HAADF intensity analysis. The areas of the dense phase of LiPSs are marked by the dotted white lines. The linear HAADF intensity analysis is marked by the white arrow. The contrast contribution of HAADF intensity can be attributed to Li2S nanocrystalline, dense phase of LiPSs, electrolyte and background, respectively. g, Schematic illustration of the path of scattered electrons.
Extended Data Fig. 8 MD simulations and DFT calculations at different interfaces.
a, Snapshots of MD simulations with different Li2S6 concentrations of 5.28 wt% and 17.93 wt%. b, Statistics of S atoms in aggregated clusters with different electrolyte concentration. The five largest clusters were used to calculate the average number of contained S atoms. c, Snapshots of MD simulations on different surfaces of Mo NCs/N-G and Ti with Li2S6 concentration of 5.28 wt%. The right row is the enlarged view of the interface details and the initial S62− and Li+ ions were randomly dispersed in the electrolyte. After the NVT relaxation of 2 ns, the sampling results indicate that Mo NCs/N-G had a strong long-range attraction with S62− ions. d, Statistics of S atoms attracted to the electrode surfaces of Mo NCs/N-G and Ti with different electrolyte concentration. e, Comparison of z-axis position variation (Å) of dense ion-complex phase at different interfaces as a function of time corresponding to MD simulations in Fig. 4e with Li2S6 concentration of 17.93 wt%. The z-axis distance is defined from droplets of LiPSs to the electrode surfaces (Ti or Mo NCs/N-G). f, MSD analysis of Li2S6 movement along the z-axis direction. The diffusion coefficients of S atoms in Li2S6 molecules over Mo NCs/N-G (D1) and Ti (D2) surfaces were 2.73 × 10−7 cm2 s−1 and 4.88 × 10−9 cm2 s−1, respectively. The initial point of MSD corresponds to 42 ns of the simulation. g, AIMD simulation of charge variation of polysulfide ions around Mo active centres from a top view and a side view. Considering the dynamic charge distribution, the polysulfide ions in the dense phase are defined as S6 ions in AIMD simulations. S6 ions in the dense phase are divided into internal, middle and external layers according to the distances from Mo active centres when viewed from the top. The charge-distribution diagrams, shown from left to right, include pristine structure relaxed for 20 ps before applying the electrode potential (I) and simulations at 0 ps (II) and 8 ps (III) after applying the electrode potential.
Extended Data Fig. 9 Evaluation of electrochemical behaviour using Li2S6 electrolyte (100 mM) in coin-type cells.
a, Potentiostatic discharge profiles of Li2S deposition at 2.05 V with/without active centres. The scale of the y axis is indicated by the bar. b, Nyquist profiles of EIS after potentiostatic discharge. Re, Rs and Rct indicate the resistance of electrolyte, surface layer and charge transfer, respectively. The inset is the local magnification profiles. c, Potentiostatic charge profiles of Li2S dissolution at 2.4 V with/without active centres. The scale of the y axis is indicated by the bar. d, Arrhenius plots of the linear relationship between logarithmic values of the reciprocal of Rct and the reciprocal of absolute temperatures for 1.7, 1.8, 1.9 and 2.0 V, with (red)/without (blue) active centres. e, Comparison of the corresponding Ea calculated on the basis of the slope of linear fitting. The Ea values with/without active centres are 0.27/0.5, 0.23/0.5, 0.17/0.49 and 0.19/0.49 eV, corresponding to the discharge potentials of 2.0, 1.9, 1.8 and 1.7 V, respectively. f, CV profiles of symmetric cells with/without active centres at a scan rate of 0.1 mV s−1. g, Comparison of cycling performance with/without mediation of active centres at 16 μA cm−2. h,i, Corresponding charge/discharge profiles at specific cycles, showing the energy barriers of Li2S nucleation and dissolution.
Extended Data Fig. 10 Evaluation of electrochemical performance in Li–S batteries.
a,b, Cycling performance (1 C, 100 cycles) and rate performance (0.2, 0.5, 1, 2, 3, 4 and 0.2 C) of Mo NCs/N-G/S, with the sulfur areal loading of 2 mg cm−2. A battery activation was performed at 0.2 C for two cycles (cycling performance) and 0.1 C for one cycle (rate performance), respectively. c, Charge/discharge profiles from rate performance of Mo NCs/N-G/S at 0.2, 0.5, 1, 2, 3 and 4 C. d, Cycling performance of Mo NCs/N-G/S at 0.2 C for 40 cycles with different sulfur areal loading of 2, 3.5, 5.4 and 7 mg cm−2. A battery activation was performed at 0.05 C for two cycles. e, Long-term and high-rate cycling performance at 2 and 3 C for 500 cycles. A battery activation was performed at 0.2 C for two cycles. The capacity value is selected every five cycles.
Supplementary information
Supplementary Video 1
Two-step deposition of rod-like Li2S by means of metastable Li2S2 and its following dissolution without mediation of active centres. For the two-step pathway, the reactions on bare electrode surface involved the initial nucleation of metastable granular Li2S2 and further growth of rod-like Li2S. (Shown at a speed 25 times the real-time process.)
Supplementary Video 2
Single-step deposition of rod-like Li2S without mediation of active centres. For the single-step pathway, rod-like Li2S was deposited directly on the bare electrode surface. (Shown at a speed 25 times the real-time process.)
Supplementary Video 3
Two-step deposition of rod-like/plate-like Li2S without mediation of active centres at −0.1 V. When reducing the discharge potential to −0.1 V, solid discharge products primarily nucleated in granular structures during the early stage of deposition, which gradually transformed into rod-like/plate-like Li2S as the deposition continued. (Shown at a speed 25 times the real-time process.)
Supplementary Video 4
Deposition of Li2S with mediation of active centres. During potentiostatic discharge, Mo NCs/N-G nanosheet continuously spread with an increase in projection area, possibly because of the deposition of Li2S. By contrast, no nucleation growth of rod-like/plate-like Li2S was observed on the Ti electrode. (Shown at a speed 50 times the real-time process.)
Supplementary Video 5
Deposition of Li2S following Supplementary Video 4 with mediation of active centres. Further observation at a higher magnification during discharge revealed that the spreading of Mo NCs/N-G nanosheet in Supplementary Video 4 should be attributed to the deposition of nanocrystalline Li2S on the edge and surface. (Shown at a speed 10 times the real-time process.)
Supplementary Video 6
Deposition of Li2S from 100 mM Li2S6 electrolyte with mediation of active centres. In Li2S6 electrolyte with a higher concentration, distinct contrast variation was observed on the electron-accessible areas of Mo NCs/N-G nanosheet (bottom right) owing to the deposition of Li2S, in comparison with the electron-inaccessible Mo NCs/N-G nanosheet (bottom left) and bare Ti electrode surface (top right). (Shown at a speed 50 times the real-time process.)
Supplementary Video 7
Reversible deposition of Li2S by means of droplet-like dense phase with mediation of active centres. With mediation of active centres, several droplets (1, 2 and 3) were observed on the surface of Mo NCs/N-G nanosheet before the instantaneous deposition of Li2S. Droplet 1 is marked with a red square and locally zoomed in. It indicated a gathering-induced crystallization pathway, involving the collective reaction behaviour of gathering, conversion and crystallization. Active centres also enabled the reversible solid–liquid phase transformation of LiPSs, shown by the surface of Mo NCs/N-G nanosheet periodically getting clear and blurry. (Shown at a speed 10 times the real-time process in the first deposition and 20 times the real-time process in the following dissolution/deposition.)
Supplementary Video 8
MD simulation of diffusion of LiPSs at the Mo NCs/N-G surface with different electrolyte concentrations. Mo NCs/N-G gathered the S62− and Li+ ions with different concentrations (5.28 wt%, 9.68 wt% and 17.93 wt%) from bulk solution to the electrode surface. The pink, orange, purple, blue and green balls represent Li, S, Mo, N and C atoms, respectively.
Supplementary Video 9
MD simulation of diffusion of LiPSs at the Mo NCs/N-G surface. S62− and Li+ ion complex was continuously attracted and gathered by the Mo NCs/N-G surface to form a droplet-like dense phase. The pink, orange, purple, blue and green balls represent Li, S, Mo, N and C atoms, respectively.
Supplementary Video 10
MD simulation of diffusion of LiPSs at the Ti surface. The Ti surface showed no interaction with LiPSs. The pink, orange, purple, blue and green balls represent Li, S, Mo, N and C atoms, respectively.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, S., Shi, J., Liu, S. et al. Visualizing interfacial collective reaction behaviour of Li–S batteries. Nature 621, 75–81 (2023). https://doi.org/10.1038/s41586-023-06326-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-023-06326-8
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.



