Abstract
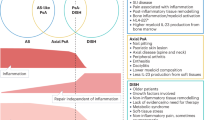
Ankylosing spondylitis (AS) was first identified in the late 17th century. 250 years later, inflammatory spine disease was recognized to be one of the patterns of psoriatic arthritis (PsA). Isolated spondylitis is rare among patients with PsA, occurring in less than 5% of patients; however, many patients with PsA have axial disease that is concurrent with peripheral arthritis. At the other end of the spondyloarthritis spectrum, psoriasis is observed in 10% of patients with AS. Although axial involvement in PsA can be indistinguishable from axial disease in AS, it can also differ in several respects, raising the question of whether axial PsA and AS (with or without psoriasis) are different clinical presentations of the same disease, or whether they are separate diseases that have overlapping features. In this Review, the clinical presentation, metrology, radiographic characteristics, genetic factors, treatment options and axial prognosis of the two diseases are addressed. The aim of this Review is to capture all available comparisons made to date, to highlight the similarities and differences between AS and axial PsA and to propose a research agenda.
Key points
-
Ankylosing spondylitis and axial psoriatic arthritis are both part of the spectrum of spondyloarthritis and have overlapping features but also differ in their genetic, clinical, radiographic and prognostic characteristics.
-
HLA-B*27 occurs less frequently in axial psoriatic arthritis than in ankylosing spondylitis but is a genetic risk factor for both diseases.
-
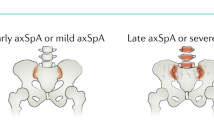
Axial psoriatic arthritis develops at an older age, is less symptomatic and is associated with distinct radiographic features compared with ankylosing spondylitis.
-
The majority of comparative studies to date have had a cross-sectional design, which captures patients at different stages of disease and hampers the true comparison of these two diseases.
-
The lack of a universally accepted definition of axial psoriatic arthritis needs to be addressed.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Rudwaleit, M. et al. The assessment of SpondyloArthritis International Society classification criteria for peripheral spondyloarthritis and for spondyloarthritis in general. Ann. Rheum. Dis. 70, 25–31 (2011).
van der Linden, S. M., Valkenburg, H. A., de Jongh, B. M. & Cats, A. The risk of developing ankylosing spondylitis in HLA-B27 positive individuals. A comparison of relatives of spondylitis patients with the general population. Arthritis Rheum. 27, 241–249 (1984).
Taurog, J. D., Chhabra, A. & Colbert, R. A. Ankylosing spondylitis and axial spondyloarthritis. N. Engl. J. Med. 374, 2563–2574 (2016).
Moll, J. M. & Wright, V. Psoriatic arthritis. Semin. Arthritis Rheum. 3, 55–78 (1973).
Ritchlin, C. T. et al. Treatment recommendations for psoriatic arthritis. Ann. Rheum. Dis. 68, 1387–1394 (2009).
Taylor, W. J., Zmierczak, H. G. & Helliwell, P. S. Problems with the definition of axial and peripheral disease patterns in psoriatic arthritis. J. Rheumatol. 32, 974–977 (2005).
Gladman, D. D. Axial disease in psoriatic arthritis. Curr. Rheumatol. Rep. 9, 455–460 (2007).
Chandran, V., Tolusso, D. C., Cook, R. J. & Gladman, D. D. Risk factors for axial inflammatory arthritis in patients with psoriatic arthritis. J. Rheumatol. 37, 809–815 (2010).
Baraliakos, X., Coates, L. C. & Braun, J. The involvement of the spine in psoriatic arthritis. Clin. Exp. Rheumatol. 33, S31–35 (2015).
Torre Alonso, J. C. et al. Psoriatic arthritis (PA): a clinical, immunological and radiological study of 180 patients. Br. J. Rheumatol. 30, 245–250 (1991).
Yang, Q. et al. Prevalence and characteristics of psoriatic arthritis in Chinese patients with psoriasis. J. Eur. Acad. Dermatol. Venereol. 25, 1409–1414 (2011).
Moghaddassi, M., Shahram, F., Chams-Davatchi, C., Najafizadeh, S. R. & Davatchi, F. Different aspects of psoriasis: analysis of 150 Iranian patients. Arch. Iran. Med. 12, 279–283 (2009).
Coates, L. C. et al. Sensitivity and specificity of the classification of psoriatic arthritis criteria in early psoriatic arthritis. Arthritis Rheum. 64, 3150–3155 (2012).
Niccoli, L. et al. Frequency of iridocyclitis in patients with early psoriatic arthritis: a prospective, follow up study. Int. J. Rheum. Dis. 15, 414–418 (2012).
Nossent, J. C. & Gran, J. T. Epidemiological and clinical characteristics of psoriatic arthritis in northern Norway. Scand. J. Rheumatol. 38, 251–255 (2009).
van der Linden, S., Valkenburg, H. A. & Cats, A. Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum. 27, 361–368 (1984).
Chandran, V., Barrett, J., Schentag, C. T., Farewell, V. T. & Gladman, D. D. Axial psoriatic arthritis: update on a longterm prospective study. J. Rheumatol. 36, 2744–2750 (2009).
Hanly, J. G., Russell, M. L. & Gladman, D. D. Psoriatic spondyloarthropathy: a long term prospective study. Ann. Rheum. Dis. 47, 386–393 (1988).
Queiro, R. et al. Clinically asymptomatic axial disease in psoriatic spondyloarthropathy. A retrospective study. Clin. Rheumatol. 21, 10–13 (2002).
Gladman, D. D., Brubacher, B., Buskila, D., Langevitz, P. & Farewell, V. T. Differences in the expression of spondyloarthropathy: a comparison between ankylosing spondylitis and psoriatic arthritis. Clin. Invest. Med. 16, 1–7 (1993).
Lubrano, E. et al. The definition and measurement of axial psoriatic arthritis. J. Rheumatol. Suppl. 93, 40–42 (2015).
Lubrano, E. & Spadaro, A. Axial psoriatic arthritis: an intriguing clinical entity or a subset of an intriguing disease? Clin. Rheumatol. 31, 1027–1032 (2012).
Jadon, D. R. et al. Axial disease in psoriatic arthritis study: defining the clinical and radiographic phenotype of psoriatic spondyloarthritis. Ann. Rheum. Dis. 76, 701–707 (2017).
Exarchou, S. et al. The prevalence of clinically diagnosed ankylosing spondylitis and its clinical manifestations: a nationwide register study. Arthritis Res. Ther. 17, 118 (2015).
Cortes, A. et al. Identification of multiple risk variants for ankylosing spondylitis through high-density genotyping of immune-related loci. Nat. Genet. 45, 730–738 (2013).
O’Rielly, D. D., Uddin, M. & Rahman, P. Ankylosing spondylitis: beyond genome-wide association studies. Curr. Opin. Rheumatol. 28, 337–345 (2016).
Brown, M. A., Kenna, T. & Wordsworth, B. P. Genetics of ankylosing spondylitis — insights into pathogenesis. Nat. Rev. Rheumatol. 12, 81–91 (2016).
Wordsworth, P. Genes in the spondyloarthropathies. Rheum. Dis. Clin. North Am. 24, 845–863 (1998).
Uddin, M. et al. Integrated genomics identifies convergence of ankylosing spondylitis with global immune mediated disease pathways. Sci. Rep. 5, 10314 (2015).
Essers, I. et al. Characteristics associated with the presence and development of extra-articular manifestations in ankylosing spondylitis: 12-year results from OASIS. Rheumatology 54, 633–640 (2015).
Machado, P. et al. Ankylosing spondylitis patients with and without psoriasis do not differ in disease phenotype. Ann. Rheum. Dis. 72, 1104–1107 (2013).
Parkes, M., Cortes, A., van Heel, D. A. & Brown, M. A. Genetic insights into common pathways and complex relationships among immune-mediated diseases. Nat. Rev. Genet. 14, 661–673 (2013).
Chandran, V. & Raychaudhuri, S. P. Geoepidemiology and environmental factors of psoriasis and psoriatic arthritis. J. Autoimmun. 34, J314–321 (2010).
Ritchlin, C. T., Colbert, R. A. & Gladman, D. D. Psoriatic Arthritis. N. Engl. J. Med. 376, 957–970 (2017).
Ananthakrishnan, R., Eckes, L. & Walter, H. On the genetics of psoriasis. An analysis of Hellgren’s data for a model of multifactorial inheritance. Arch. Dermatol. Forsch. 247, 53–58 (1973).
Brandrup, F., Holm, N., Grunnet, N., Henningsen, K. & Hansen, H. E. Psoriasis in monozygotic twins: variations in expression in individuals with identical genetic constitution. Acta Derm. Venereol. 62, 229–236 (1982).
Lonnberg, A. S. et al. Heritability of psoriasis in a large twin sample. Br. J. Dermatol. 169, 412–416 (2013).
Myers, A., Kay, L. J., Lynch, S. A. & Walker, D. J. Recurrence risk for psoriasis and psoriatic arthritis within sibships. Rheumatology 44, 773–776 (2005).
Chandran, V. et al. Familial aggregation of psoriatic arthritis. Ann. Rheum. Dis. 68, 664–667 (2009).
Karason, A., Love, T. J. & Gudbjornsson, B. A strong heritability of psoriatic arthritis over four generations — the Reykjavik Psoriatic Arthritis Study. Rheumatology 48, 1424–1428 (2009).
Winchester, R. et al. HLA associations reveal genetic heterogeneity in psoriatic arthritis and in the psoriasis phenotype. Arthritis Rheum. 64, 1134–1144 (2012).
Eder, L., Chandran, V. & Gladman, D. D. What have we learned about genetic susceptibility in psoriasis and psoriatic arthritis? Curr. Opin. Rheumatol. 27, 91–98 (2015).
Chandran, V. et al. Human leukocyte antigen alleles and susceptibility to psoriatic arthritis. Hum. Immunol. 74, 1333–1338 (2013).
Eder, L. et al. Human leucocyte antigen risk alleles for psoriatic arthritis among patients with psoriasis. Ann. Rheum. Dis. 71, 50–55 (2012).
Queiro, R. et al. HLA-C locus alleles may modulate the clinical expression of psoriatic arthritis. Arthritis Res. Ther. 8, R185 (2006).
FitzGerald, O., Haroon, M., Giles, J. T. & Winchester, R. Concepts of pathogenesis in psoriatic arthritis: genotype determines clinical phenotype. Arthritis Res. Ther. 17, 115 (2015).
Tang, H. et al. A large-scale screen for coding variants predisposing to psoriasis. Nat. Genet. 46, 45–50 (2014).
Ellinghaus, E. et al. Genome-wide association study identifies a psoriasis susceptibility locus at TRAF3IP2. Nat. Genet. 42, 991–995 (2010).
Cargill, M. et al. A large-scale genetic association study confirms IL12B and leads to the identification of IL23R as psoriasis-risk genes. Am. J. Hum. Genet. 80, 273–290 (2007).
Liu, Y. et al. A genome-wide association study of psoriasis and psoriatic arthritis identifies new disease loci. PLoS Genet. 4, e1000041 (2008).
Huffmeier, U. et al. Common variants at TRAF3IP2 are associated with susceptibility to psoriatic arthritis and psoriasis. Nat. Genet. 42, 996–999 (2010).
Ellinghaus, E. et al. Genome-wide meta-analysis of psoriatic arthritis identifies susceptibility locus at REL. J. Invest. Dermatol. 132, 1133–1140 (2012).
Stuart, P. E. et al. Genome-wide association analysis of psoriatic arthritis and cutaneous psoriasis reveals differences in their genetic architecture. Am. J. Hum. Genet. 97, 816–836 (2015).
Lanier, L. L. NK cell recognition. Annu. Rev. Immunol. 23, 225–274 (2005).
Chandran, V. et al. Killer-cell immunoglobulin-like receptor gene polymorphisms and susceptibility to psoriatic arthritis. Rheumatology 53, 233–239 (2014).
Weisman, M. H., Witter, J. P. & Reveille, J. D. The prevalence of inflammatory back pain: population-based estimates from the US National Health and Nutrition Examination Survey, 2009–2010. Ann. Rheum. Dis. 72, 369–373 (2013).
Mease, P. J., Garg, A., Helliwell, P. S., Park, J. J. & Gladman, D. D. Development of criteria to distinguish inflammatory from noninflammatory arthritis, enthesitis, dactylitis, and spondylitis: a report from the GRAPPA 2013 annual meeting. J. Rheumatol. 41, 1249–1251 (2014).
Queiro, R. et al. Clinical expression, but not disease outcome, may vary according to age at disease onset in psoriatic spondylitis. Joint Bone Spine 75, 544–547 (2008).
Helliwell, P. S., Hickling, P. & Wright, V. Do the radiological changes of classic ankylosing spondylitis differ from the changes found in the spondylitis associated with inflammatory bowel disease, psoriasis, and reactive arthritis? Ann. Rheum. Dis. 57, 135–140 (1998).
Lindstrom, U. et al. Back pain and health status in patients with clinically diagnosed ankylosing spondylitis, psoriatic arthritis and other spondyloarthritis: a cross-sectional population-based study. BMC Musculoskelet. Disord. 17, 106 (2016).
Perez Alamino, R. et al. Differential features between primary ankylosing spondylitis and spondylitis associated with psoriasis and inflammatory bowel disease. J. Rheumatol. 38, 1656–1660 (2011).
Gladman, D. D., Brubacher, B., Buskila, D., Langevitz, P. & Farewell, V. T. Psoriatic spondyloarthropathy in men and women: a clinical, radiographic, and HLA study. Clin. Invest. Med. 15, 371–375 (1992).
Landi, M. et al. Gender differences among patients with primary ankylosing spondylitis and spondylitis associated with psoriasis and inflammatory bowel disease in an iberoamerican spondyloarthritis cohort. Medicine 95, e5652 (2016).
Gladman, D. D. et al. International spondyloarthritis interobserver reliability exercise — the INSPIRE study: I. Assessment of spinal measures. J. Rheumatol. 34, 1733–1739 (2007).
Helliwell, P. S. Assessment of disease activity in psoriatic arthritis. Clin. Exp. Rheumatol. 33, S44–S47 (2015).
Taylor, W. J. & Harrison, A. A. Could the Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) be a valid measure of disease activity in patients with psoriatic arthritis? Arthritis Rheum. 51, 311–315 (2004).
Fernandez-Sueiro, J. L. et al. Validity of the Bath Ankylosing Spondylitis Disease Activity Index for the evaluation of disease activity in axial psoriatic arthritis. Arthritis Care Res. 62, 78–85 (2010).
Orbai, A. M. et al. International patient and physician consensus on a psoriatic arthritis core outcome set for clinical trials. Ann. Rheum. Dis. 76, 673–680 (2017).
Eder, L., Chandran, V., Shen, H., Cook, R. J. & Gladman, D. D. Is ASDAS better than BASDAI as a measure of disease activity in axial psoriatic arthritis? Ann. Rheum. Dis. 69, 2160–2164 (2010).
Turkiewicz, A. M. & Moreland, L. W. Psoriatic arthritis: current concepts on pathogenesis-oriented therapeutic options. Arthritis Rheum. 56, 1051–1066 (2007).
MacKay, K., Mack, C., Brophy, S. & Calin, A. The Bath Ankylosing Spondylitis Radiology Index (BASRI): a new, validated approach to disease assessment. Arthritis Rheum. 41, 2263–2270 (1998).
Averns, H. L. et al. Radiological outcome in ankylosing spondylitis: use of the Stoke Ankylosing Spondylitis Spine Score (SASSS). Br. J. Rheumatol. 35, 373–376 (1996).
Creemers, M. C. et al. Assessment of outcome in ankylosing spondylitis: an extended radiographic scoring system. Ann. Rheum. Dis. 64, 127–129 (2005).
Lubrano, E. et al. Psoriatic Arthritis Spondylitis Radiology Index: a modified index for radiologic assessment of axial involvement in psoriatic arthritis. J. Rheumatol. 36, 1006–1011 (2009).
Baraliakos, X., Listing, J., Rudwaleit, M., Sieper, J. & Braun, J. Development of a radiographic scoring tool for ankylosing spondylitis only based on bone formation: addition of the thoracic spine improves sensitivity to change. Arthritis Rheum. 61, 764–771 (2009).
Biagioni, B. J. et al. Reliability of radiographic scoring methods in axial psoriatic arthritis. Arthritis Care Res. 66, 1417–1422 (2014).
Lubrano, E. et al. The radiological assessment of axial involvement in psoriatic arthritis: a validation study of the BASRI total and the modified SASSS scoring methods. Clin. Exp. Rheumatol. 27, 977–980 (2009).
Rudwaleit, M. et al. The development of Assessment of SpondyloArthritis International Society classification criteria for axial spondyloarthritis (part I): classification of paper patients by expert opinion including uncertainty appraisal. Ann. Rheum. Dis. 68, 770–776 (2009).
Rudwaleit, M. et al. The development of Assessment of SpondyloArthritis International Society classification criteria for axial spondyloarthritis (part II): validation and final selection. Ann. Rheum. Dis. 68, 777–783 (2009).
Richette, P. et al. Psoriasis and phenotype of patients with early inflammatory back pain. Ann. Rheum. Dis. 72, 566–571 (2013).
Poggenborg, R. P., Sorensen, I. J., Pedersen, S. J. & Ostergaard, M. Magnetic resonance imaging for diagnosing, monitoring and prognostication in psoriatic arthritis. Clin. Exp. Rheumatol. 33, S66–S69 (2015).
Williamson, L. et al. Clinical assessment of sacroiliitis and HLA-B27 are poor predictors of sacroiliitis diagnosed by magnetic resonance imaging in psoriatic arthritis. Rheumatology 43, 85–88 (2004).
Ostergaard, M., Poggenborg, R. P., Axelsen, M. B. & Pedersen, S. J. Magnetic resonance imaging in spondyloarthritis — how to quantify findings and measure response. Best Pract. Res. Clin. Rheumatol. 24, 637–657 (2010).
Maldonado-Ficco, H., Sheane, B. J., Thavaneswaran, A., Chandran, V. & Gladman, D. D. Magnetic resonance imaging in psoriatic arthritis: a descriptive study of indications, features and effect on treatment change. J. Clin. Rheumatol. 23, 243–245 (2017).
Queiro, R., Tejon, P., Alonso, S., Alperi, M. & Ballina, J. Erosive discovertebral lesion (Andersson lesion) as the first sign of disease in axial psoriatic arthritis. Scand. J. Rheumatol. 42, 220–225 (2013).
Castillo-Gallego, C., Aydin, S. Z., Emery, P., McGonagle, D. G. & Marzo-Ortega, H. Magnetic resonance imaging assessment of axial psoriatic arthritis: extent of disease relates to HLA-B27. Arthritis Rheum. 65, 2274–2278 (2013).
Poggenborg, R. P. et al. Head-to-toe whole-body MRI in psoriatic arthritis, axial spondyloarthritis and healthy subjects: first steps towards global inflammation and damage scores of peripheral and axial joints. Rheumatology 54, 1039–1049 (2015).
Braun, J. et al. 2010 update of the ASAS/EULAR recommendations for the management of ankylosing spondylitis. Ann. Rheum. Dis. 70, 896–904 (2011).
Rohekar, S. et al. 2014 update of the Canadian Rheumatology Association/Spondyloarthritis Research Consortium of Canada treatment recommendations for the management of spondyloarthritis. Part I: principles of the management of spondyloarthritis in Canada. J. Rheumatol. 42, 654–664 (2015).
Ward, M. M. et al. American College of Rheumatology/Spondylitis Association of America/Spondyloarthritis Research and Treatment Network 2015 recommendations for the treatment of ankylosing spondylitis and nonradiographic axial spondyloarthritis. Arthritis Care Res. 68, 151–166 (2016).
Sieper, J. et al. Secukinumab efficacy in anti-TNF-naive and anti-TNF-experienced subjects with active ankylosing spondylitis: results from the MEASURE 2 Study. Ann. Rheum. Dis. 76, 571–592 (2017).
Mease, P. J. et al. Secukinumab inhibition of interleukin-17A in patients with psoriatic arthritis. N. Engl. J. Med. 373, 1329–1339 (2015).
Strand, V. et al. Secukinumab improves patient-reported outcomes in subjects with active psoriatic arthritis: results from a randomised phase III trial (FUTURE 1). Ann. Rheum. Dis. 76, 203–207 (2017).
Mease, P. & McInnes, I. B. Secukinumab: a new treatment option for psoriatic arthritis. Rheumatol. Ther. 3, 5–29 (2016).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT02721966 (2017).
Poddubnyy, D., Hermann, K. G., Callhoff, J., Listing, J. & Sieper, J. Ustekinumab for the treatment of patients with active ankylosing spondylitis: results of a 28-week, prospective, open-label, proof-of-concept study (TOPAS). Ann. Rheum. Dis. 73, 817–823 (2014).
Yeremenko, N., Paramarta, J. E. & Baeten, D. The interleukin-23/interleukin-17 immune axis as a promising new target in the treatment of spondyloarthritis. Curr. Opin. Rheumatol. 26, 361–370 (2014).
Baeten, D. et al. Secukinumab, an interleukin-17A inhibitor, in ankylosing spondylitis. N. Engl. J. Med. 373, 2534–2548 (2015).
Pathan, E. et al. Efficacy and safety of apremilast, an oral phosphodiesterase 4 inhibitor, in ankylosing spondylitis. Ann. Rheum. Dis. 72, 1475–1480 (2013).
US National Library of Medicine. ClinicalTrials.gov https://clinicaltrials.gov/ct2/show/NCT01583374 (2018).
van der Heijde, D. et al. Tofacitinib in patients with ankylosing spondylitis: a phase II, 16-week, randomised, placebo-controlled, dose-ranging study. Ann. Rheum. Dis. 76, 1340–1347 (2017).
Coates, L. C. et al. Group for Research and Assessment of Psoriasis and Psoriatic Arthritis 2015 treatment recommendations for psoriatic arthritis. Arthritis Rheumatol. 68, 1060–1071 (2016).
Gossec, L. et al. European League Against Rheumatism (EULAR) recommendations for the management of psoriatic arthritis with pharmacological therapies: 2015 update. Ann. Rheum. Dis. 75, 499–510 (2016).
Nash, P. et al. Updated guidelines for the management of axial disease in psoriatic arthritis. J. Rheumatol. 41, 2286–2289 (2014).
Mease, P. J. et al. Etanercept treatment of psoriatic arthritis: safety, efficacy, and effect on disease progression. Arthritis Rheum. 50, 2264–2272 (2004).
Mease, P. J. et al. Adalimumab for the treatment of patients with moderately to severely active psoriatic arthritis: results of a double-blind, randomized, placebo-controlled trial. Arthritis Rheum. 52, 3279–3289 (2005).
Mease, P. J. et al. Brodalumab, an anti-IL17RA monoclonal antibody, in psoriatic arthritis. N. Engl. J. Med. 370, 2295–2306 (2014).
Antoni, C. E. et al. Sustained benefits of infliximab therapy for dermatologic and articular manifestations of psoriatic arthritis: results from the infliximab multinational psoriatic arthritis controlled trial (IMPACT). Arthritis Rheum. 52, 1227–1236 (2005).
Kavanaugh, A. et al. Clinical efficacy, radiographic and safety findings through 5 years of subcutaneous golimumab treatment in patients with active psoriatic arthritis: results from a long-term extension of a randomised, placebo-controlled trial (the GO-REVEAL study). Ann. Rheum. Dis. 73, 1689–1694 (2014).
Kavanaugh, A. et al. Radiographic progression of patients with psoriatic arthritis who achieve minimal disease activity in response to golimumab therapy: results through 5 years of a randomized, placebo-controlled study. Arthritis Care Res. 68, 267–274 (2016).
Gladman, D. D. et al. Adalimumab for long-term treatment of psoriatic arthritis: forty-eight week data from the adalimumab effectiveness in psoriatic arthritis trial. Arthritis Rheum. 56, 476–488 (2007).
McInnes, I. B. et al. Efficacy and safety of secukinumab, a fully human anti-interleukin-17A monoclonal antibody, in patients with moderate-to-severe psoriatic arthritis: a 24-week, randomised, double-blind, placebo-controlled, phase II proof-of-concept trial. Ann. Rheum. Dis. 73, 349–356 (2014).
Lubrano, E., Parsons, W. J. & Perrotta, F. M. Assessment of response to treatment, remission, and minimal disease activity in axial psoriatic arthritis treated with tumor necrosis factor inhibitors. J. Rheumatol. 43, 918–923 (2016).
Lubrano, E. et al. The effectiveness of a biologic agent on axial manifestations of psoriatic arthritis. A twelve months observational study in a group of patients treated with etanercept. Clin. Exp. Rheumatol. 29, 80–84 (2011).
Maharaj, A. B. & Chandran, V. Treatment of psoriatic arthritis with traditional DMARD’s and novel therapies: approaches and recommendations. Expert Rev. Clin. Immunol. 13, 319–331 (2017).
Raychaudhuri, S. P., Wilken, R., Sukhov, A. C., Raychaudhuri, S. K. & Maverakis, E. Management of psoriatic arthritis: early diagnosis, monitoring of disease severity and cutting edge therapies. J. Autoimmun. 76, 21–37 (2017).
Gladman, D. et al. Tofacitinib for psoriatic arthritis in patients with an inadequate response to TNF inhibitors. N. Engl. J. Med. 377, 1525–1536 (2017).
Carette, S., Graham, D., Little, H., Rubenstein, J. & Rosen, P. The natural disease course of ankylosing spondylitis. Arthritis Rheum. 26, 186–190 (1983).
Kaprove, R. E., Little, A. H., Graham, D. C. & Rosen, P. S. Ankylosing spondylitis: survival in men with and without radiotherapy. Arthritis Rheum. 23, 57–61 (1980).
Machado, P. et al. Both structural damage and inflammation of the spine contribute to impairment of spinal mobility in patients with ankylosing spondylitis. Ann. Rheum. Dis. 69, 1465–1470 (2010).
Poddubnyy, D. & Sieper, J. Radiographic progression in ankylosing spondylitis/axial spondyloarthritis: how fast and how clinically meaningful? Curr. Opin. Rheumatol. 24, 363–369 (2012).
van der Heijde, D. et al. Radiographic findings following two years of infliximab therapy in patients with ankylosing spondylitis. Arthritis Rheum. 58, 3063–3070 (2008).
van der Heijde, D. et al. Radiographic progression of ankylosing spondylitis after up to two years of treatment with etanercept. Arthritis Rheum. 58, 1324–1331 (2008).
van der Heijde, D. et al. Adalimumab effectiveness for the treatment of ankylosing spondylitis is maintained for up to 2 years: long-term results from the ATLAS trial. Ann. Rheum. Dis. 68, 922–929 (2009).
Haroon, N. et al. The impact of tumor necrosis factor alpha inhibitors on radiographic progression in ankylosing spondylitis. Arthritis Rheum. 65, 2645–2654 (2013).
Molnar, C. et al. TNF blockers inhibit spinal radiographic progression in ankylosing spondylitis by reducing disease activity: results from the Swiss Clinical Quality Management cohort. Ann. Rheum. Dis. 77, 63–69 (2018).
Acknowledgements
The authors acknowledge the role of the Krembil Foundation in financially supporting the Psoriatic Arthritis Research Program at the University of Toronto. The work of J.F. was supported financially by a fellowship grant from Novartis.
Author information
Authors and Affiliations
Contributions
All authors researched data for the article, provided substantial contributions to discussions of the content, wrote the article and reviewed and/or edited the article before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Feld, J., Chandran, V., Haroon, N. et al. Axial disease in psoriatic arthritis and ankylosing spondylitis: a critical comparison. Nat Rev Rheumatol 14, 363–371 (2018). https://doi.org/10.1038/s41584-018-0006-8
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41584-018-0006-8
This article is cited by
-
Serum proteome analysis identifies a potential biomarker for axial psoriatic arthritis
European Journal of Medical Research (2024)
-
Effectiveness and safety of secukinumab in ankylosing spondylitis and psoriatic arthritis: a 52-week real-life study in an Italian cohort
Advances in Rheumatology (2023)
-
Risk of incident autoimmune diseases in patients with newly diagnosed psoriatic disease: a nationwide population-based study
Scientific Reports (2023)
-
Genetic and Molecular Distinctions Between Axial Psoriatic Arthritis and Radiographic Axial Spondyloarthritis: Post Hoc Analyses from Four Phase 3 Clinical Trials
Advances in Therapy (2023)
-
18F-sodium fluoride PET-CT visualizes both axial and peripheral new bone formation in psoriatic arthritis patients
European Journal of Nuclear Medicine and Molecular Imaging (2023)