Abstract
Hepatocellular carcinoma (HCC) is one of the most common primary liver malignancies and is the third leading cause of tumor-related mortality worldwide. Despite advances in HCC treatment, diagnosis at the later stages, and the complex mechanisms relating to the cause and pathogenesis, results in less than 40% of HCC patients being eligible for potential therapy. Prolonged inflammation and resulting immunosuppression are major hallmarks of HCC; however, the mechanisms responsible for these processes have not been clearly elucidated. In this study, we identified SOCS-7, an inhibitor of cytokine signaling, as a novel regulator of immunosuppression in HCC. We found that SOCS-7 mediated E3 ubiquitin ligase activity on a signaling adaptor molecule, Shc1, in Huh-7 cells. Overexpression of SOCS-7 reduced the induction of immunosuppressive factors, TGF-β, Versican, and Arginase-1, and further reduced STAT3 activation. Furthermore, using an in vivo tumor model, we confirmed that SOCS-7 negatively regulates immunosuppression and inhibits tumor growth by targeting Shc1 degradation. Together, our study identified SOCS-7 as a possible therapeutic target to reverse immunosuppression in HCC.
Similar content being viewed by others
Introduction
Hepatocellular carcinoma (HCC) is one of the primary forms of liver malignancy that affects more than 800,000 people worldwide every year1. Depending on the stage of the disease HCC patients have a 5-year survival rate of 10–30%2. Chronic viral infections with hepatitis B or C virus predispose a fraction of patients for development of HCC3. Other predispositions include alcohol consumption, diabetes, smoking, and body weight4. Recent treatment strategies in HCC include immunotherapy, which involves induction of the immune system to eradicate tumor cells. For example, treatment with programmed cell death-1 (PD-1) antibodies had a promising effect in a small fraction of HCC patients5,6. However, there is still no broad-spectrum treatment that effectively targets HCC, which can be attributed to a poor understanding of immune deregulation and molecular pathogenesis of the disease. Hence, to identify promising therapeutic strategies, a holistic understanding of the pathways involved in promoting immunosuppression and driving HCC pathogenesis is required.
The suppressor of cytokine signaling (SOCS) family of proteins are intracellular proteins capable of regulating various signaling pathways and altering the response of cells to different cytokines7,8. It consists of eight proteins including SOCS 1–7 and cytokine-inducible SH2 domain-containing protein (CIS)9. SOCS proteins have been shown to be associated with signal transducer and activator of transcription 3 (STAT3) hyperactivation, regulating tumorigenesis and vascular invasion10. A key member of this family, SOCS-7, alters signaling pathways by ubiquitination of targets. It negatively correlates with STAT3 activation in renal fibrosis11,12. Proteasomal degradation of SOCS-7 promoted inflammation resulting in renal failure13, and knockdown of SOCS-7 favored tumor growth in in vivo models of breast cancer14. A comprehensive analysis of methylation patterns of socs genes in HCC patients revealed hypermethylation of SOCS1 in HBV infection-related hepatocarcinogenesis but not SOCS-7. Hence, the role of SOCS-7 in HCC pathogenesis has not been described so far.
The Src homology 2 domain containing (SHC)-transforming protein 1 (Shc1) is an adaptor protein that functions in coupling growth factor receptors to intracellular signaling pathways. It has three different isoforms: p46 and p52 (activation of Ras signaling), and p66 (pro-apoptotic). The p52 isoform has been implicated in promoting breast cancer, whereas the p66 isoform has been shown to promote autophagy and disrupt mitochondrial function in B cells15,16. In breast cancer, Shc1 specifically activates STAT3-mediated immunosuppressive signals and inhibits STAT1 mediated immune responses to drive tumorigenesis17. Furthermore, it has been shown that inhibition of STAT3 activation or STAT3 deletion can improve immune evasion to suppress tumor progression18. In our previous experiments, it was shown that high expression of Shc1 was associated with poor survival of HCC patients, and knockdown of p66 subtype significantly reduced tumor growth, in vitro and in vivo, by inhibiting the activation of STAT319. However, the regulation of Shc1 expression in the context of HCC is still unknown.
In this study, we identified a novel mechanism that involved SOCS-7-mediated regulation of the Shc1/STAT3 pathway in regulating tumor immunosuppression to inhibit tumorigenesis in HCC, by using both in vitro and in vivo HCC models.
Materials and methods
Tissues and cell culture
Tissues was obtained from patients recruited from Zhongshan Hospital, Fudan University, Shanghai, China. Informed consent was obtained from all patients and the study was approved by, and conformed to all criteria set, by the local Ethics Committee. Human hepatoma carcinoma cell lines, Huh-7 and HepG2 cells, were purchased from Procell Life Science (Wuhan, China), and cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) (Gibco, Grand Island, NY, USA) supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin and 100 mg/mL streptomycin at 5% CO2 at 37 °C.
Lentivirus construction and infection
The SOCS-7 gene was cloned into the lentiviral vector pENTR1A (Miaoling, Wuhan, China) and the overexpression vector pENTR1A-SOCS-7 was constructed. Transfection of the constructed plasmid into 293 T cells by Lipofectamine 3000® transfection reagent (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s protocol. After 48 h, the culture lentiviral supernatant was collected, the virus titer was determined, and Huh-7 cells were infected with the virus to establish Huh-7 cells stably overexpressing SOCS-7. The following are the primer sequences for plasmid generation: SOCS-7 (forward: 5′-ATGCAGGAGGCCGAGCTCCGG-3′, reverse: 5′-CTACGTGGAGGG TTCCACCTCTTGC-3′). Overexpression vectors used empty vectors as controls. Similarly, to silence SOCS-7 expression, shRNA expression vectors pGCSIL-GFP-shSOCS-7 (Genechem, Shanghai, China) will be constructed. It was also transfected into 293 T cells by Lipofectamine 3000® transfection reagent for stable expression. The virus infects Huh-7 cells, thereby establishing Huh-7 cells that stably silence SOCS-7, while silencing vectors used scrambled oligomer as controls. shSOCS-7 (5′-GACGUUUAUAGUAGAUAAG-3′). A scrambled oligomer (5′-UUCUUCGAACGUGUCACGUTT-3′) was used as the control for shRNA experiments.
RNA extraction and qRT-PCR
Total RNA was extracted from Huh-7 cells or tumor tissues using TRIzol reagent, according to the manufacturer’s instructions (Invitrogen). First strand cDNA synthesis was performed using a ReverTra Ace qPCR RT Kit (Toyobo, Osaka, Japan) and expression of Shc1, SOCS-7, and β-actin were evaluated using the following gene specific primers: Shc1 forward: 5′-TGGGATGAGGAGGAGGAAGA-3′, reverse: 5′-CAATGTAGCTCCCAAGTGGC-3′; SOCS-7 forward: 5′-TCAAATCCCTCCAGCACCTT-3′, reverse 5′-GCTGCGCTTCCTTTAG AGAC-3′; TGF-β forward: 5′-GCCTGAGTGGCTGTCTTTTG-3′, reverse: 5′-CTGTATTCC GTCTCCTTGGTTC-3′; Veriscan forward: 5′-TCGTTTTGAGAACCAGACAGG-3′, reverse: 5′-GTTCATTTTGCAGCGATCAG-3′; Arginase-1 forward: 5′-CATGAGCTCCAAGCCAAA GT-3′, reverse: 5′-TTTTTCCAGCAGACCAGCTT-3′; β-actin forward 5′-CCCTGGAGAA GAGCTACGAG-3′, reverse 5′-CGTACAGGTCTTTGCGGATG-3′. Gene expression was calculated as 2−∆∆Ct relative to β-actin.
Immunoprecipitation
For immunoprecipitation using magnetic beads, the Pierce™ MS-Compatible Magnetic IP Kit (Pierce, Waltham, MA, USA) was used according to the manufacturer’s protocol. Briefly, the cells were harvested and washed with ice-cold phosphate-buffered saline (PBS), and lysed directly in lysis buffer containing 25 mM Tris-HCl (pH 7.4), 150 mM NaCl, 1% Nonidet P-40, 1 mM EDTA, and 5% glycerol with freshly added protease inhibitors (Sigma-Aldrich, St. Louis, MO, USA). After incubating for 30 min on ice, the lysates were spun at full-speed to remove cell debris and the clear supernatant was used with antibody-coupled magnetic beads for immunoprecipitation. After magnetic separation, the immunoprecipitated fraction was eluted and subjected to Western blot analysis.
Western blotting
To generate whole-cell lysates for analysis of protein expression, the cells were harvested and lysed in RIPA lysis buffer (Abcam, Cambridge, UK) for 30 min, on ice, by periodic vortexing. Cell lysates were centrifuged at 13,000 rpm for 10 min to separate cell debris. The supernatants were carefully transferred to a new tube and total protein estimation was performed using a BCA protein assay kit (Pierce). Twenty µg of total protein per sample was loaded onto a 10% sodium dodecyl sulfate (SDS)-acrylamide gel and subjected to electrophoresis. Separated proteins were then blotted onto a 0.45 µm nitrocellulose membrane (Abcam) and blocked in 5% defatted milk (in PBS or Tris-buffered saline) containing 0.1% Tween 20, for 2 h at room temperature. Membranes were then incubated with primary antibodies against SOCS-7 (1:200), Shc1 (1:500), STAT3 (1:500) and p-STAT3 (1:500) (all from Santa Cruz Biotechnology, Santa Cruz, CA, USA) overnight at 4 °C. After washing the membranes three times with PBS-T, they were incubated with the respective horseradish peroxidase (HRP)-conjugated secondary antibody (Santa Cruz Biotechnology) for 1 h at room temperature. The membranes were developed using a peroxidase substrate (Thermo Fisher Scientific, Waltham, MA, USA) and visualized by chemiluminescence. Quantification of protein expression was performed using ImageJ (National Institutes of Health, Bethesda, MD, USA) by comparing densities between the proteins of interest with that of β-actin, which was used as a loading control.
Immunohistochemistry
Liver tissue sections from HCC patients were processed by standard immunohistochemistry procedures [18] including deparaffinization, rehydration, and antigen retrieval. The sections were stained with primary antibody against SOCS-7 (1:100) for 2 h followed by HRP-conjugated secondary antibody for 1 h. Liquid DAB Plus Substrate Kit (Thermo Fisher Scientific) was used to visualize the proteins of interest. H-Score (H-SCORE = ∑(PI × I) = (percentage of cells of weak intensity × 1) + (percentage of cells of moderate intensity × 2) + percentage of cells of strong intensity × 3)20,21 of the destination area of each image was quantified using Densito quant module in Quant Center 2.1 analysis software, the higher the value of H-score, the stronger the positive SOCS-7 intensity.
Protein half-life and ubiquitination assays
To determine the half-life of Shc1, cycloheximide (100 µg/mL) (MedChemExpress, Monmouth, NJ, USA) was added to transfected cells and the cells were harvested after incubation for 0, 2, 4, 6 and 8 h. Harvested cells were lysed as described above and evaluated for Shc1 expression by western blotting. To evaluate the ubiquitination levels of proteins, transfected cells were treated with a proteasomal inhibitor (10 µM MG-132) (MedChemExpress) for 6 h prior to harvest. Harvested cells were analyzed for protein expression, as previously described.
TUNEL assay
Cell apoptosis was evaluated using a TUNEL assay kit (Sigma–Aldrich), according to the manufacturer’s instructions. Briefly, paraffin-embedded tissue sections were fixed using 4% paraformaldehyde and counterstained with 4′,6-diamidino-2-phenylindole (DAPI) for nuclear localization, without permeabilization. The cells were visualized by fluorescence microscopy and apoptotic cells were marked in both green (fragmented DNA) and blue (nuclear DNA).
Migration and scratch assays
Cell migration of Huh-7 cells was evaluated using a Transwell assay and scratch test. A total of 2.5 × 104 cells/well were seeded on 6-well plates overnight or starved on Transwell chambers (Corning, Corning, NY, USA) in serum-free medium. For the scratch assay, an even scratch wound was made using a disposable pipette, followed by washing with PBS. The cells were then incubated in supplemented medium and immediately imaged for the first time point (0 time point) and then subsequently imaged at 24 and 48 h. Cell migration was calculated by the scratch distance. For Transwell assays, Transwell chambers containing starved cells were incubated in wells containing media supplemented with bovine serum albumin (BSA) (10 g/L) as a chemoattractant for 0, 24, and 48 h at 37 °C/5% CO2. The chambers were removed and migrated cells in the lower well were visualized after washing with PBS, fixing with methanol at 4 °C, and staining with 0.1% Crystal Violet. Images were taken from five different fields of view using a 200× objective.
In vivo HCC tumor mouse model
Six weeks old BALB/c nude mice (Shanghai Slac Laboratory Animal Co., China) housed under sterile pathogen-free conditions were used for the study. 1 × 106 Huh-7 cells either stably expressing or not overexpressing SOCS-7 were subcutaneously xenografted into mice in the flank region, respectively, and normal Huh-7 cells were used as control. These mice were sacrificed 5 weeks after transfer, and the tumors were harvested. Tumor volume was evaluated using the formula: ½ (length × width2). Tissue sections were prepared and stained with hematoxylin and eosin followed by Ki-67 (Abcam, 1:500) staining to assess cell proliferation. All animal experimentation was approved by Institutional Animal Care and Use Committee of the Fudan University, Shanghai, China.
Statistical analysis
All data are expressed as the mean ± standard deviation (SD). The Kaplan–Meier method was used to evaluate the survival of patients. Student’s t test was used for sample comparison between two groups, and one-way analysis of variance (ANOVA) with Bonferroni’s post-hoc test was used for sample comparison among multiple groups. All differences with p < 0.05 were considered statistically significant. All statistical analysis was performed using SPSS statistical software for Windows, version 21.0 (SPSS, Chicago, IL, USA).
Results
Expression of SOCS-7 is downregulated in HCC
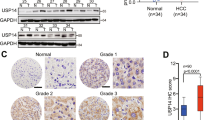
SOCS-7 is a regulator of various signaling pathways including the JAK/STAT pathway that controls cell proliferation12. To analyze its role in HCC, tumor tissues along with adjacent healthy tissues were obtained from 10 patients, and SOCS-7 expression was evaluated by Western blotting. Compared to healthy control tissues, HCC samples were significantly lower in SOCS-7 protein expression (Fig. 1A). Similarly, the mRNA level of SOCS-7 was higher in adjacent tissues than in cancer tissues by qRT-PCR (Fig. 1B). However, as shown in Table 1, of the 77 patients studied, 36 exhibited low-SOCS-7 (H-score < 76.96), while 41 exhibited high-SOCS-7 (H-score ≥ 76.96). Data analysis showed that tumor differentiation was correlated with SOCS-7 expression (p = 0.0365), but for other clinical data, such as age, gender, size, recurrence, death, cirrhosis, there was no correlation. Through the H-Score and histochemical analysis of SOCS-7 in adjacent tissues and tumor tissues (Figs. 1C, 1D), we found that the content of SOCS-7 in tumor tissues was significantly lower than that in adjacent tissues, which makes us think that SOCS-7 had a significant impact on the tumorigenesis of HCC. Taken together, these results indicated that SOCS-7 was downregulated in HCC and might play an important role in HCC pathogenesis.
A Representative western blot and densitometric quantification of HCC tissue samples and adjacent healthy control samples from 10 patients. B Relative expression of SOCS-7 mRNA in tumor tissues and adjacent tissues of 10 patients was detected by qRT-PCR. C Representative immunohistochemical (IHC) images of SOCS-7 expression in tumor tissues and adjacent tissues. D Relative IHC staining of SOCS-7 in paired tumor tissues and adjacent tissues samples are shown. Statistical significance was determined by student’s t tests. Statistical significance was calculated by the Student’s t test. *p < 0.05; **p < 0.01 and ***p < 0.001.
Increased SOCS-7 expression can inhibit the growth of HCC in vitro
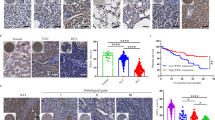
To further characterize the role of SOCS-7 in HCC, we quantitatively analyzed the mRNA expression of SOCS-7 in different hepatoma cell lines (Huh-7, HepG2). The Huh-7 cells showed a more significant reduction compared with normal human hepatocytes LO2 (Fig. 2A); therefore, Huh-7 cells were selected for subsequent experiments. We constructed vector-SOCS-7, an overexpression vector of SOCS-7, and shSOCS-7, a small interfering RNA that impedes the expression of SOCS-7. These plasmids were transfected into Huh-7 cells by liposome transfection. After 48 h, the protein level of SOCS-7 was detected by WB to evaluate the expression and effect of the vector plasmid, as shown in Fig. 2B. Next, we used siRNA knockdown and overexpression vectors in Huh-7 cells. The cells were transfected with the respective expression constructs; they were harvested and analyzed for cell apoptosis, proliferation, migration, and viability. MTT assay was used to detect the cell viability. After 72 h of infection, compared with the control group, the cell viability decreased when SOCS-7 was overexpressed, and increased significantly after knockout (Fig. 2C). The TUNEL assay revealed an increase in apoptosis upon SOCS-7 overexpression, when compared to siRNA depletion of SOCS-7 expression (Fig. 2D). The wound healing assay and Transwell migration assay showed that SOCS-7 overexpression reduced the proliferative and migratory capacities of cells, respectively, when compared to cells expressing shSOCS-7 after 24 h (Figs. 2E, F). In vitro experiments showed that SOCS-7 could inhibit the growth and migration ability of HCC cells.
A Relative expression of SOCS-7 mRNA was determined by qRT-PCR in different human HCC cell lines. B The constructed overexpression vector and empty vector, scrambled oligomer and small interfering RNA were transfected into Huh-7 cells. Cells were collected after 48 h and the protein level of SOCS 7 was detected by WB. C Cell viability measured by the MTT assay in Huh-7 cells transfected with SOCS-7 or shSOCS-7. Shown are the means ± standard deviation (SD) from at least three independent experiments. D Cell apoptosis measured by the TUNEL assay. Huh-7 cells expressing a control vector, SOCS-7 overexpression vector, or shSOCS-7 were stained for fragmented (TUNEL) and nuclear DNA (4′,6-diamidino-2-phenylindole) 48 h post-transfection. Cells undergoing apoptosis were double positive for both fragmented (green) and nuclear (blue) DNA signals. Representative micrographs are shown. E Cell migration using the scratch assay. Scratch area closure of Huh-7 cells expressing the respective lentiviral vectors were evaluated 0, 24, and 48 h post-transfection. Representative images were taken from five different fields using a 200× objective. F Cell invasion measured by the Transwell migration assay. Huh-7 cells were seeded on the upper chamber of a Transwell chamber and invasion was measured 48 h after transfection with the respective lentiviral construct, in the lower chamber. Representative images were taken from five different fields using a 200× objective. Statistical significance was calculated using the unpaired Student’s t test. *p < 0.05; **p < 0.01.
SOCS-7 inhibits tumor growth through immunoregulatory factors in vivo
To evaluate the relevance of SOCS-7 expression in tumor growth in vivo, we used six-week-old BALB/c nude mice and xenografted Huh-7 cells either overexpressing SOCS-7 or shSOCS-7 that reduced SOCS-7 expression. Five weeks later, tumors were harvested and characterized for weight and volume. As shown in Fig. 3A, B, SOCS-7 significantly reduced the tumor volume in mice, and as shown in Fig. 3C, its weight was also significantly decreased compared with the control group, while transfection of shSOCS-7 resulted in an increase in volume and weight. In order to examine cell proliferation in the tumor tissues, IHC staining of Ki67 was performed. As shown in Fig. 3D, up/down regulated SOCS-7 in these mouse tumors resulted in decreased/increased cell proliferation, as visualized by Ki67 staining. Next, we examined the protein and mRNA levels of immunosuppressive factors such as TGF-β, integrin-like molecule Versican, and Arginase-1 in tumor tissues (Figs. 3E, F). The protein and mRNA levels of TGF-β, Versican, and Arginase-1 were significantly decreased in tumor tissues from mice, xenografted with SOCS-7 overexpression cells when compared to controls. In the serum of mice overexpressing SOCS-7, the content of TGF-β was significantly decreased, whereas in the serum of mice knocking out SOCS-7, it was significantly increased (Fig. 3G). In addition, overexpression of SOCS-7 upregulated the production of INF-γ, whereas knockdown of SOCS-7 significantly decreased INF-γ (Fig. 3H). These results suggested that SOCS-7 may regulate tumor growth by modulating tumor immunity in vivo. Also, expression of programmed death-ligand 1 (PD-L1) and beta-2- macroglobulin (B2M), which are critical factors expressed in response to inhibition or activation of immune responses, respectively, were also altered in response to SOCS-7 expression. PD-L1 was downregulated upon SOCS-7 overexpression, whereas it was upregulated in tissues from shSOCS-7-expressing mice (Fig. 3I). In contrast, B2M was upregulated upon SOCS-7 overexpression and downregulated upon knockdown (Fig. 3I). These results indicated a significant correlation between the expression of SOCS-7 and immunosuppressive factors that could evade host immune responses and promote tumorigenesis.
A–C In vivo analysis of tumor growth. 1 × 106 Huh-7 cells stably expressing control, SOCS-7 overexpression, or siRNA vectors were xenografted into mice, and tumor weight/volume were evaluated 5 weeks post-injection. B Evaluation of tumor weight. A, C Tumor volume calculated using the formula: ½ (length × width2) and representative images of the harvested tumors are shown in comparison (n = 5 mice/group). D The representative images of H&E stain and IHC of Ki67 of tumor tissues are shown. E The expression of immunosuppressive factors TGF-β, Versican and Arginase-1 in tumor tissues was detected by WB. F Expression of immunosuppressive factors, TGF-β, Versican, and Arginase-1 in tumor tissues quantified by qRT-PCR. G TGF-β was measured in mouse serum by the ELISA method (H) Detection of INF-γ released in the fluid of tumor tissues, quantified by ELISA. I B2M and PD-L1 protein expressions evaluated by western blots of tumor tissues. Representative western blot images and corresponding densitometric quantitation are shown. Data are represented as the means ± SD. Statistical significance was calculated using the unpaired Student’s t test. *p < 0.05; **p < 0.01; ***p < 0.001.
SOCS-7 targets Shc1 degradation by ubiquitination
The SOCS family of proteins is known to promote ubiquitination and degradation of targets22. To further characterize the mechanism behind the regulation of immunosuppression by SOCS-7, we used UbiBROWSER, a specific ubiquitin ligase target prediction software23, and identified Shc1 as a downstream target of SOCS-7. To functionally verify this relationship, we evaluated the expression of Shc1 isoforms upon overexpression or knockdown of SOCS-7 in Huh-7 cells. Western blot analysis revealed a reduction in Shc1 isoforms p46 and p66 upon SOCS-7 overexpression, and an increase upon knockdown (Fig. 4A). In contrast, qPCR analysis revealed no significant differences in mRNA levels of shc1 expression (Fig. 4B). We next evaluated ubiquitination levels of Shc1 using an anti-ubiquitin antibody. Shc1 was highly ubiquitinated upon SOCS-7 overexpression and reduced upon its knockdown (Fig. 4C). We further analyzed Shc1 protein half-life by treating Huh-7 cells with cycloheximide for 0, 2, 4, 6 h. A gradual decrease in Shc1 protein levels was observed in Huh-7 cells from 0 to 6 h; however, compared with the control group, cells that overexpressed SOCS-7 decreased more rapidly, while cells that interfered with SOCS-7 decreased more slowly (Fig. 4D). Addition of the proteasomal inhibitor, MG-132, prevented degradation of Shc1 in the presence of SOCS-7 (Fig. 4E). Taken together, these results showed that in the cell line of HCC, Shc1 was ubiquitinated and targeted degradation by the E3 ubiquitin ligase, SOCS-7.
A After 48 h of transfection, the cells were collected and the protein level of shc1 was detected by WB. B The mRNA levels of shc1 in different samples were detected by qRT-PCR. Shown are the means ± SD. C Representative western blot images of Shc1 immunoprecipitated and analyzed by anti-ubiquitin antibody. D SOCS-7, or shSOCS-7-expressing Huh-7 cells were treated with cycloheximide post-transfection for 0 h, 2 h, 4 h, 6 h, and Shc-1 expression was evaluated by western blotting. E At 42 h after transfection, Huh-7 cells were treated with MG132 for 6 h, and the expression of SOCS-7 and Shc1 was detected by WB. Representative western blot images are shown. Data are represented as the means ± SD.
SOCS-7 inhibits tumorigenesis in HCC via regulation of Shc1 in vivo
To further verify whether the regulation of SOCS-7 on tumorigenesis is regulated by Shc1 in vivo, we xenografted Huh-7 cells transfected with shSOCS-7, sh-Shc1 and shSOCS-7+sh-Shc1 into 6-week-old mice to observe the effect of Shc1 on tumor growth in mice when interfering with or overexpressing SOCS-7. As shown in Fig. 5A–C, after interfering with the expression of SOCS-7, inhibiting the expression of Shc1 at the same time has a more significant effect on the growth of tumors in mice, while overexpressing Shc1 could promote the growth of tumors. In the mouse model, co-expressing SOCS-7 and Shc1, the growth of tumors also showed a slow trend due to the inhibition of SOCS-7 on Shc1. Similarly, the tumor immune microenvironment of mice in each group was also examined by tumor immunosuppressive factors TGF-β, Veriscan, Arginase-1 detection, as shown in Figs. 5D, E, we found that Shc1 inhibits the immunosuppress regulation of SOCS-7, which indicates that Shc1 participates in the regulation of tumor immune evasion by SOCS-7. Through intra-tissue western blot, we detected that SOCS-7 had a significant inhibitory effect on the phosphorylation modification of STAT3 in tumor tissues, as shown in Fig. 5F. Previous studies found that Shc1 promotes the phosphorylation of STAT319, which indicated that SOCS-7 can regulate the phosphorylation modification of STAT3 by regulating Shc1. Overall, we have demonstrated experimentally that in hepatoma cells, SOCS-7 modifies Shc1 by ubiquitination, affecting the phosphorylation modification of intracellular STAT3, thereby regulating tumor immunosuppressive factors in the tumor microenvironment, and inhibiting tumorigenesis.
A Tumor weight evaluated from mice from different groups: Huh-7 cells expressing sh-Shc1, shSOCS-7, shSOCS-7+sh-Shc1, or control. B Quantification of tumor volume. C Representative images of mouse tumors evaluated 5 weeks after xenograft of Huh-7 cells. D, E The protein and mRNA expression of immunosuppressive factors, TGF-β, Versican, and Arginase-1 by qRT-PCR in cells which overexpressing Shc1 or sh-Shc1. F Western blot images of lysates from tissues indicating expressions of STAT3 and phosphor-STAT3. Representative western blot images are shown. Data are represented as the means ± SD. Statistical significance was calculated using the unpaired Student’s t test. *p < 0.05; **p < 0.01; ***p < 0.001.
Discussion
Regulation of cytokine-mediated signaling pathways and their contribution to the pathogenesis of HCC has not been clearly elucidated. Important pathways triggered during tumorigenesis, such as growth factor and interferon signaling, are regulated by the SOCS family of proteins. They are capable of interacting with each other to target proteins proteasomal degradation24. In the context of HCC, SOCS-1 and SOCS-3 have been shown to negatively regulate Janus kinase (JAK)/STAT signaling and suppressing tumorigenesis25,26. In this study, we highlighted the clinical significance of SOCS-7 in HCC pathogenesis. We identified Shc1 as a downstream target of SOCS-7, which was ubiquitinated and targeted for degradation.
Constitutive activation of the JAK/STAT signaling pathway leads to aberrant cell growth and proliferation, as observed in several leukemias and in vitro in transformed cell lines27,28. SOCS-1 and SOCS-3 have been previously identified as negative regulators of this pathway. In our analysis from patient samples, we identified SOCS-7 to be significantly downregulated in HCC tissue samples. This downregulation in expression can be associated with epigenetic silencing, which includes methylation of CpG islands, as seen for SOCS-1, instability of mRNA, and/or protein expression25. Aberrant cell proliferation and migration, along with increased tumor growth seen upon reduction of SOCS-7, can be explained by constitutive activation of the JAK/STAT signaling pathway.
The consequence of reduced SOCS-7 expression extends from promoting cell growth to favoring immunosuppressive pathways, including interferon signaling for tumor growth29. Reduction in SOCS-7 levels promoted expression of immunosuppressive and tumor-inducing factors such as TGF-β, Versican, and Arginase-1. The role of TGF-β in promoting tumor invasion is well-described and in the context of HCC; it acts as a master regulator of immune activation and tolerance. In T cells, it upregulates expression of PD-1, and in association with its ligand PD-L1 can trigger immunosuppressive signals. A large genomic study on HCC tissues also revealed an increase in expression of PD-1, PD-L1, and other immunosuppressive markers30. We also observed a decrease in PD-L1 expression upon SOCS-7 overexpression in tumor tissues. Additionally, transactivation of Versican, a proteoglycan, is also associated with increased tumor invasion and promoted progression of HCC31,32.
Through in silico analysis of downstream ubiquitin E3 ligase targets, we identified Shc1 as a target of SOCS-7. Shc1 expression was regulated at the protein level but not at the mRNA level, by selective ubiquitination by SOCS-7. Although SOCS-7 is known to directly affect STAT3 activation by sequestering STAT3 in the cytoplasm to prevent its translocation into the nucleus33. We observed that overexpression of Shc1, along with SOCS-7 knockdown, was capable of restoring tumor growth, indicating that the mechanism was regulated via Shc1. In previous experiments, we also found that the longest proapoptotic isomer (Shcp66) preferentially phosphorylated STAT3 at serine 727 position to promote tumorigenesis in HCC19. It also promotes autophagy in lung adenocarcinoma cells, and in B cells by affecting mitochondrial integrity15,34. Hence, in addition to its effect on STAT3 activation, Shc1 can also induce autophagy in HCC to balance cellular stress35. Therefore, Shc1 can reduce the progress of HCC, by affecting tumor progression through several mechanisms.
In summary, we identified a novel role for SOCS-7 in HCC pathogenesis, which reduces STAT3 activation through ubiquitination modification of Shc1, enhances the sensitivity of HCC cells to NK cell-mediated immune attack, thereby inhibiting tumorigenesis. This newly identified mechanism identified a possible immunotherapeutic target by utilizing SOCS-7 and/or Shc1 to manipulate the balance established between favoring cell growth and promoting immunosuppression in HCC.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
WHO. International Agency for Research on Cancer. Liver. https://gco.iarc.fr/today/data/factsheets/cancers/11-Liver-fact-sheet.pdf (2018).
Golabi, P. et al. Mortality assessment of patients with hepatocellular carcinoma according to underlying disease and treatment modalities. Medicine (Baltimore). 96, e5904 (2017).
Levrero, M. & Zucman-Rossi, J. Mechanisms of HBV-induced hepatocellular carcinoma. J. Hepatol. 64, S84–S101 (2016).
Balogh, J. et al. Hepatocellular carcinoma: a review. J. Hepatocell Carcinoma. 3, 41–53 (2016).
El-Khoueiry, A. B. et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet. 389, 2492–2502 (2017).
Zhu, A. X. et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol. 19, 940–952 (2018).
Starr, R. et al. A family of cytokine-inducible inhibitors of signalling. Nature. 387, 917–921 (1997).
Ilangumaran, S., Ramanathan, S. & Rottapel, R. Regulation of the immune system by SOCS family adaptor proteins. Semin Immunol. 16, 351–365 (2004).
Hilton, D. J. et al. Twenty proteins containing a C-terminal SOCS box form five structural classes. Proc Natl Acad Sci USA. 95, 114–119 (1998).
Huang, Y. et al. Defective hepatic response to interferon and activation of suppressor of cytokine signaling 3 in chronic hepatitis C. Gastroenterology 132, 733–744 (2007).
Yang, R. et al. p53 induces miR199a-3p to suppress SOCS7 for STAT3 activation and renal fibrosis in UUO. Sci. Rep. 7, 43409 (2017).
Martens, N. et al. Suppressor of cytokine signaling 7 inhibits prolactin, growth hormone, and leptin signaling by interacting with STAT5 or STAT3 and attenuating their nuclear translocation. J. Biol. Chem. 280, 13817–13823 (2005).
Fu, B. et al. PTPN14 aggravates inflammation through promoting proteasomal degradation of SOCS7 in acute liver failure. Cell Death Dis. 11, 803 (2020).
Sasi, W., Ye, L., Jiang, W. G., Sharma, A. K. & Mokbel, K. In vitro and in vivo effects of suppressor of cytokine signalling 7 knockdown in breast cancer: the influence on cellular response to hepatocyte growth factor. Biomed Res. Int. 2014, 648040 (2014).
Onnis, A. et al. The pro-oxidant adaptor p66SHC promotes B cell mitophagy by disrupting mitochondrial integrity and recruiting LC3-II. Autophagy. 14, 2117–2138 (2018).
Wright, K. D. et al. The p52 isoform of SHC1 is a key driver of breast cancer initiation. Breast Cancer Res. 21, 74 (2019).
Ahn, R. et al. The Shc1 adaptor simultaneously balances Stat1 and Stat3 activity to promote breast cancer immune suppression. Nat Commun. 8, 14638 (2017).
Moreira, D. et al. STAT3 inhibition combined with CpG immunostimulation activates antitumor immunity to eradicate genetically distinct castration-resistant prostate cancers. Clin. Cancer Res. 24, 5948–5962 (2018).
Huang, P. et al. p66Shc promotes HCC progression in the tumor microenvironment via STAT3 signaling. Exp. Cell Res. 383, 111550 (2019).
Guo, R. et al. MET IHC is a poor screen for MET amplification or MET Exon 14 mutations in lung adenocarcinomas: Data from a Tri-Institutional Cohort of the Lung Cancer Mutation Consortium. J. Thorac. Oncol. 14, 1666–1671 (2019).
Paschalis, A. et al. Prostate-specific membrane antigen heterogeneity and DNA repair defects in prostate cancer. Eur. Urol. 76, 469–478 (2019).
Krebs, D. L. & Hilton, D. J. A new role for SOCS in insulin action. Suppressor of cytokine signaling. Sci. STKE. 2003, PE6 (2003).
Li, Y. et al. An integrated bioinformatics platform for investigating the human E3 ubiquitin ligase-substrate interaction network. Nat. Commun. 8, 347 (2017).
Piessevaux, J. et al. Functional cross-modulation between SOCS proteins can stimulate cytokine signaling. J. Biol. Chem. 281, 32953–32966 (2006).
Yoshikawa, H. et al. SOCS-1, a negative regulator of the JAK/STAT pathway, is silenced by methylation in human hepatocellular carcinoma and shows growth-suppression activity. Nat. Genet. 28, 29–35 (2001).
Niwa, Y. et al. Methylation silencing of SOCS-3 promotes cell growth and migration by enhancing JAK/STAT and FAK signalings in human hepatocellular carcinoma. Oncogene 24, 6406–6417 (2005).
Weber-Nordt, R. M. et al. Constitutive activation of STAT proteins in primary lymphoid and myeloid leukemia cells and in Epstein-Barr virus (EBV)-related lymphoma cell lines. Blood 88, 809–816 (1996).
Danial, N. N., Pernis, A. & Rothman, P. B. Jak-STAT signaling induced by the v-abl oncogene. Science 269, 1875–1877 (1995).
Lu, C. et al. Current perspectives on the immunosuppressive tumor microenvironment in hepatocellular carcinoma: challenges and opportunities. Mol Cancer 18, 130 (2019).
Chen, J. et al. Analysis of genomes and transcriptomes of hepatocellular carcinomas identifies mutations and gene expression changes in the transforming growth factor-beta pathway. Gastroenterology 154, 195–210 (2018).
Tanaka, Y. et al. Sharpin promotes hepatocellular carcinoma progression via transactivation of Versican expression. Oncogenesis 5, e277 (2016).
Zhangyuan, G. et al. VersicanV1 promotes proliferation and metastasis of hepatocellular carcinoma through the activation of EGFR-PI3K-AKT pathway. Oncogene 39, 1213–1230 (2020).
Kershaw, N. J., Murphy, J. M., Lucet, I. S., Nicola, N. A. & Babon, J. J. Regulation of Janus kinases by SOCS proteins. Biochem. Soc. Trans. 41, 1042–1047 (2013).
Matsuda, S., Saito, H. & Nishiyama, N. Basic fibroblast growth factor suppressed the enhancement of choline acetyltransferase activity induced by nerve growth factor. Neurosci. Lett. 114, 69–74 (1990).
Yu, S. et al. Autophagy in the “inflammation-carcinogenesis” pathway of liver and HCC immunotherapy. Cancer Lett. 411, 82–89 (2017).
Funding
The research is supported by National Natural Science Foundation of China (NSFC), No. 81772590, 81972233.
Author information
Authors and Affiliations
Contributions
P.H. participated in the design of the study, animal model construction and the tissue collection, cultured cells and drafted the manuscript. Z.Z. and Y.C. participated in cultured cells and performed the data analysis. B.Y. assisted with the animal model construction and drafted the manuscript. J.X. conceived and participated in the design of the study, and final approval of the version to be submitted. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval and consent to participate
All animal experimentation was approved by Institutional Animal Care and Use Committee of the Fudan University, Shanghai, China. All experiments were conducted according to guidelines approved by the Zhongshan Hospital affiliated with Fudan University.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Huang, P., Zhao, Z., Chen, Y. et al. The E3 ubiquitin ligase SOCS-7 reverses immunosuppression via Shc1 signaling in hepatocellular carcinoma. Lab Invest 102, 613–620 (2022). https://doi.org/10.1038/s41374-022-00727-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41374-022-00727-5