Abstract
We previously reported that osteopontin (OPN), a matrix structural glycophosphoprotein, is upregulated in the injured mouse lens prior to the epithelial–mesenchymal transition (EMT). Here, we investigated the role of this protein in EMT of the lens epithelium during wound healing. The crystalline lens was injured by needle puncture in OPN-null (KO, n=40) and wild-type (WT, n=40) mice. The animals were killed at day 1, 2, 5, and 10 postinjury. Immunohistochemistry was employed to detect α-smooth muscle action (αSMA), a marker of EMT, collagen type I, transforming growth factor β1 (TGFβ1), TGFβ2, and phospho-Smad2/3. Cell proliferation was assayed by examining uptake of bromodeoxyuridine (BrdU). The results showed that injury-induced EMT of mouse lens epithelium, as evaluated by histology, expression pattern of αSMA and collagen I, was altered in the absence of OPN with reduced phospho-Smad2/3 signaling. Upregulation of TGFβ1 and TGFβ2 in the epithelium was also inhibited. Cell proliferation was more active in KO mice as compared with WT mice at day 1 and 2, but not at day 5 and 10. An in vitro experiment shows OPN facilitates cell adhesion of lens epithelial cell line. OPN is required for activation of Smad2/3 signal in an injured lens epithelium and lens cell EMT.
Similar content being viewed by others
Main
The lens epithelial cell undergoes an epithelial-mesenchymal transition (EMT)1, 2 and transdifferentiates into fibroblasrtic cells that are capable of expression of fibrous extracellular matrix (ECM) components upon various stimuli, that is, during the healing process in a healing lens. However, this healing process of EMT often leads to formation of scar tissue rather than to regeneration of normal functional lens tissue.3, 4 Lens cell EMT is a major cause of development of fibrous type postcataract surgery capsular opacification (PCO) that impairs patients' postoperative vision.5, 6, 7 Although expression of α-smooth muscle actin (αSMA) and collagen I is characteristic of myofibroblasts,8, 9, 10 these molecules are also the established markers of EMT in lens epithelial cells in that lens epithelial cells are positive for vimentin even in a quiescent condition.3, 4 Certainly human lens epithelial cells up-regulate αSMA and that the PCO tissue is positive for collagen type I, indicating that human lens epithelial cells undergo EMT in vivo postoperatively.3, 11 Although EMT of in vivo lens epithelial cells or other cell types is mediated by transforming growth factor β (TGFβ)/Smad signal,2, 7, 12, 13, 14, 15, 16, 17, 18 some ECM molecules, that is, fibronectin or lumican are known to further support the process.19, 20, 21, 22 Nevertheless the whole molecular and cellular mechanisms underlying the EMT in lens cells during responses to injury are still not fully characterized.
Osteopontin (OPN) is a matrix structural glycophosphoprotein that is also abundantly expressed in tissues during inflammation and repair.23, 24, 25, 26, 27, 28, 29, 30 Its molecular weight deduced from SDS-polyacrylamide gel electrophoresis mobility ranges from 55 to 80 kDa. It functions as a cytokine that regulates activities of macrophages, other immune cells, and resident tissue cells (epithelial cell types and mesenchymal cells) at sites of injury.23, 24, 25, 26, 27, 28, 29, 30 OPN contains RGD-dependent and -independent (SVVYGLR) adhesion motifs and binds via αv integrins, CD44 and some β1 integrins.23, 24, 25, 26, 27, 28, 29, 30 Such cell adhesive protein might affect EMT via modulation of cell adhesion and adhesion-dependent cytoplasmic signaling in various cell types. However, the role of this molecule in the regulation of EMT is to be uncovered. We previously reported that OPN is newly expressed in injured lens epithelial cells in association with fibrotic scar formation in mice and humans.31 Lens epithelium is a suitable model for investigation of injury-induced EMT because behaviors of lens epithelial cells can be observed with minimal contamination of other cell types from outside the lens. To address this question, we took advantage of the availability of OPN-null (KO) mice. In the present study, we showed that injury-induced EMT of mouse lens epithelium was perturbed in KO mice with inhibition of Smad2/3 signal.
Materials and methods
Experiments were approved by the DNA Recombination Experiment Committee and the Animal Care and Use Committee of Wakayama Medical University and conducted in accordance with the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research.
Lens Injury in Mouse Eyes
Lens injury was performed as reported previously.4, 17, 31 We employed the protocol of mouse lens capsular injury approved by National Cancer Institute/National Institutes of Health, MD (Laboratory of Cell Regulation and Carcinogenesis). KO of C57Bl/6 background (n=20) and wild-type (+/+, WT, n=20) mice were generally anesthetized with an i.p. pentobarbital sodium (70 mg/kg body weight) and also topically by oxybuprocaine eyedrops.4, 17, 31 Our preliminary investigation showed the occurrence of EMT in lens epithelium postinjury was similar between WT and heterozygous mice (data not shown). A small incision was made in the central anterior capsule with a 26 G hypodermic needle through a corneal incision in one eye (right or left) after topical application of mydriatics as previously reported. In brief, the central anterior lens capsule was pierced one time by the blade part of the 26 G needle apex. The depth of puncture from the corneal surface was approximately 300 μm, which was about one-forth of the length of the blade part of the needle. The animals with an accidentally deeper injury in the lens were not included in the experiment. After instillation of ofloxacin ointment, the mice were allowed to heal for 1, 2, 5, and 10 days. The number of eye samples were 6/6, 6/6, 4/4, and 4/4 for each of WT/KO mice at 1, 2, 5, and 10 days, respectively. Mice were killed 2 h after i.p. bromodeoxyuridine (BrdU) injection by CO2 asphyxia and cervical dislocation.4, 17, 31 Enucleated globes were fixed and embedded in paraffin.4, 17, 31
Immunohistochemistry
Deparaffinized sections and fixed cultured cells were processed for immunohistochemistry as reported previously.4, 17, 31 Antibodies used were; mouse monoclonal anti-αSMA antibody (Neomarker); rabbit polyclonal anti-phospho-Smad2 (Ser465/467) antibody (1:100 in phosphate-buffered saline (PBS), Chemicon, Temecula, CA, USA); rabbit polyclonal anti-phospho-Smad3 (Ser423/425) antibody (1:200 in PBS, Biosource, Camarillo, CA, USA); goat polyclonal anti-collagen type I antibody (1:100 in PBS, Southern Biotechnology, Birmingham, AL, USA); goat polyclonal anti-TGFβ1 antibody (1: 100 in PBS, Santa Cruz Biotechnology, Santa Cruz, CA, USA); rabbit polyclonal anti-TGFβ2 antibody and mouse monoclonal anti-BrdU antibody (1:10 in PBS, Rosch-Boehringer-Mannheim, Germany). For BrdU immuohistochemistry the tissue sections were treated with 2 N HCl for 2 h at 37°C and then processed for blocking and primary antibody reaction as reported previously. After secondary peroxidase-conjugated antibody reaction and washing in PBS, the antibody complex was visualized with 3,3′-diaminobenzidine reaction. After nuclear counterstaining with methylgreen the specimens were observed under regular light microscopy.
Adhesion of Lens Epithelial Cell Line to OPN-Coated Glass
Lab-Tec Glass chamber slides (Nunc, NY, USA) were coated with human recombinant OPN (0.25 μg/ml, Chemicon, Catalog # CC1074, Temecula, CA, USA) in PBS for 24 h at 4°C. α-TN4 mouse lens epithelial cell line (1.8 × 104/ml/well) were seeded in the wells of chamber slides. After specific intervals of incubation, the slides were fixed with formalin and the cells were stained with HE. The number of cells that had adhered to and spread on the chamber slides was counted. Three wells were prepared and the mean value of the cells from three independent areas was determined. Data were analyzed by unpaired t-test. P<0.05 was considered significant.
Results
Histology of a Mouse Injured Lens
We examined the effects of endogenous OPN on injury-induced EMT of lens epithelium by using HE stain for histology and immunohistochemistry for expression of αSMA and collagen type I.
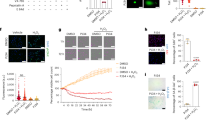
Histology of HE-stained tissues showed a remarkable alteration of the morphology of cells accumulated at the site of injury in WT mice (Figure 1). Lens epithelial cells of the uninjured lens (not shown) and those at the capsular break at until day 2 of injury maintained an epithelial cell-like morphology in both WT (Figure 1a, a′) and KO mice (Figure 1d, d′). Thereafter, the WT cells assumed an elongated, fibroblast-like morphology, implicating the process of EMT at days 5 (Figure 1b, b′) and 10 (Figure 1c, c′). On the other hand, the KO lens epithelial cells were of epithelial shape even at day 5 (Figure 1e, e′). Finally, the KO cells exhibited a fibroblastic, elongated morphology at day 10 (Figure 1f, f′).
Histology of the edge of the break in the anterior lens capsule of WT and osteopontin-null mice. At day 2 lens epithelial cells around to the capsular break exhibit epithelial-like morphology in both a WT (a, a′, arrow) and KO (d, d′, arrow) mice. At day 5 the epithelial cells form a multilayer at the edge of the broken capsule in both WT (b) and KO (e) mice. At a higher magnification the WT cells of (b′) show an elongated fibroblast-like shape (arrows), while those of an KO (e′) lens exhibit an epithelial-like morphology (arrows). At day 10 a multicellular layer forms beneath the anterior capsule in both WT (c) and KO (f) mice. At a higher magnification the cells in both groups (c′, f′) exhibit an elongated fibroblast-like shape. Frames a′–f′ indicate the higher magnification pictures of the boxed areas in frames a–f. AC, anterior lens capsule, bar, 20 μm.
Immunohistochemistry
HE histology suggested that loss of OPN might attenuate the EMT process of the lens epithelium. Therefore, we conducted immunodetection of αSMA to confirm this hypothesis. Both WT and KO lens epithelial cells of the injured lenses were negative for αSMA at days 1 and 2 (data not shown). The multilayered fibroblast-like lens cells at the capsular break of a WT mouse from day 5 began to express αSMA (Figure 2a), whereas the epithelial-shaped lens cells outside the multicell layer were negative (not shown). At day 10 the majority of the cells beneath the broken capsule were labeled with anti-αSMA antibody in a WT lens (Figure 2b). In KO mice the whole lens cells were not labeled for aSMA in two specimens and the majority of the cells adjacent to the capsular break were not labeled for αSMA in the other two samples (Figure 2c), and even at day 10 the cells were negative except for the cells just beneath the broken anterior lens capsule (Figure 2d).
Expression pattern of α-SMA in lens epithelial cells of injured lenses. The multicelllular layer of epithelial cells at the edge of the broken capsule is strongly labeled with antiα-SMA antibody in a WT mouse at days 5 (a) and 10 (b). On the other hand, in KO mice almost all the epithelial cells in the multicellular layer do not stain for α-SMA and even at day 10 the majority of the cells are not labeled. Arrows, α-SMA-expressing lens cells; stars, immunoreactivity for α-SMA in iris sphincter muscle. AC, anterior capsule, bar, 20 μm.
Type I collagen is the major ECM component seen in lens capsule fibrosis postinjury or surgery and is secreted by fibroblast-like lens cells post-EMT. The single layered epithelial cells of injured lens 1 and 2 days after injury were not labeled by anti-collagen I antibody (data not shown), whereas weak immunoreactivity for type I collagen could be seen in the multilayered lens epithelial cells around the capsular break 5 (Figure 3a) and 10 (Figure 3b) days of injury. In KO mice, the majority of the cells in cell multilayer adjacent to the capsular break were not labeled for collagen I (Figure 3c); at day 10 the collagen I expression level in the KO lens was similar to that in the WT lens (Figure 3d).
Expression pattern of type I collagen in injured lenses. At days 5 (a, c) and 10 (b, d) immunoreactivity for type I collagen is detected in the multicellular layer formed at the site of capsular injury of a WT mouse (a, b). Whereas such immunoreactivity is not observed in the multicellular layer at day 5 in an injured KO lens (c, d), it is readily detected at day 10. Arrows, type I collagen immunoreactivity; AC, anterior capsule, bar, 20 μm.
As for the TGFβ/Smad signal, we immunostained for phosphorylated Smad2 and phosphorylated Smad3 in tissue sections. At day 1 (Figure 4Aa) and 2 (Figure 4Ab) lens epithelial cells adjacent to the break in the anterior capsule were labeled for phospho-Smad2 in their nuclei in WT mice, whereas epithelial cells in KO mice were not labeled (day 1, Figure 4Ae; day 2, Figure 4Af). At day 5 the majority of the lens epithelial cells in cell multilayer formed adjacent the break in a KO mouse were negative for phospho-Smad2 (Figure 4Ag) except for a few cells at the leading edge of the cell multi-later (arrow). In a WT lens many positive cells were observed at day 5 (Figure 4Ac). At day 10 many phospho-Smad2-positive nuclei were still detected in cell multilayer at the injury site in a WT mouse (Figure 4Ad). On the other hand phospho-Smad2 was faintly detected mainly in the cell cytoplasm, not in the nuclei, of lens epithelial cells beneath the waved anterior capsule (Figure 4Ah). Expression pattern of phospho-Smad3 was quite similar to that of phospho-Smad2 (Figure 4B). At day 5 many cells were labeled for phospho-Smad3 in a WT lens (Figure 4Ba), while almost none of them were positive in a KO lens (Figure 4Bb).
Expression patterns of phospho-Smad2/3 and TGF β in lens epithelia of injured lenses. (A) At day 1 (a, e) and day 2 (b, f) the cells at the edge of the broken capsule are positive for phospho-Smad2 in the nuclei in WT mice (a, b), while the cells are negative in the KO mice (e, f). Insets in frames a and b show higher magnification pictures of the cells at the edge of the capsular break indicated by arrows. At day 5 the nuclei of many epithelial cells in the multicellular layer are labeled for phospho-Smad2 in an injured WT lens (C, arrows). On the other hand, in an KO injured lens the nuclei of the epithelial cells at the leading edge of the cellular multi-layer are labeled (arrows), but the majority of the nuclei are not labeled, for phospho-Smad2. At day 10, there are many epithelial cells of which nuclei are positive for phospho-Smad2 in the multicellular layer at the capsular break site in a WT mouse (arrows). Whereas phospho-Smad2 is detected in the cytoplasm, but not in cell nuclei, of the majority of the epithelial cells there in an injured KO lens. (B) The expression pattern of phospho-Smad3 was similar to that of phospho-Smad2. Marked phospho-Smad3 expression is detected in the cells in the multicellular layer formed at the capsular break site in a WT mouse (a), while the majority of the epithelial cells there is negative in an KO mouse (b) at day 5. (C) At days 5 (a) and 10 (b) immunoreactivity for TGFβ1 (arrows) is detected in the multicellular layer formed at the site of capsular injury of a WT mouse. Whereas such immunoreactivity is not observed in the multicellular ayer at day 5 (c) in an injured KO lens. At day 10 (d), however, its immunoreactivity is observed (arrow) in both groups of specimens. (D) Marked immunoreactivity for TGFβ2 is detected the multicellular layer (asterisk) of a WT mouse at day 5 (a), while faint immunoreactivity for TGFβ2 is seen in cells (asterisk) in an KO mouse (b). Arrows, TGFb1 immunoreactivity, AC, anterior capsule, bar, 20 μm.
Although we previously reported that the main component of TGFβ that activates Smad signal in epithelium of an injured mouse lens is TGFβ2 derived from aqueous humor, there might be a possibility that autocrine TGFβs also affect the cells. TGFβ1 and TGFβ2 are known to be upregulated during EMT in mouse lens epithelium. The present immunohistochemistry did not detect TGFβ1 in whole epithelium in an uninjured lens epithelium as reported previously17 (data not shown). TGFβ2 protein expression was observed in the epithelium in the lens equator, but not in the anterior epithelium of the uninjured lens (data not shown). Both TGFβ1 (Figure 4C) and TGFβ2 (Figure 4D) were upregulated in EMT lens epithelium in WT mice, while such upregulation was quite minimal in KO mice.
Cell Proliferation as Examined by BrdU Labeling
Cell proliferation in the epithelium of an injured lens was evaluated by BrdU uptake (Figure 5). The epithelium of the WT lens begins to proliferate at day 2 postinjury (Figure 5A), while the cells in a KO lens around the capsular break contained a significant number of cells with BrdU-immunoreactivity beneath the anterior capsule even at day 1 (Figure 5A and B). At day 2 the number of BrdU-labeled cells was significantly higher in a KO lens as compared with a WT lens (Figure 5A). At day 5 proliferating cells were still detected in a KO lens, but statistically not significantly different from the WT lens (Figure 5A and B). At day 10 there were no cells labeled with anti-BrdU antibody in either genotype (Figure 5A).
Immunohistochemical detection of incorporated BrdU in injured lenses. Cell proliferation in the epithelia of injured lenses was evaluated by BrdU uptake. (A) The number of anterior epithelial cells that are labeled with anti-BrdU antibody at each time point. BrdU-labeled cells are not seen in the WT epithelium until day 2 postinjury whereas they are seen at day 1 in the KO epitheliuim around the capsular break. At day 2 the number of BrdU-labeled cells was significantly higher in the KO lens as compared with a WT lens. At day 10 there were no cells labeled with the anti-BrdU antibody. *P<0.05, **P<0.01. (B) Immunohistology of BrdU-labeled cells. No BrdU-positive nuclei are seen in a WT (a), while BrdU-labeled nuclei are observed (arrows) in cell multilater at this timepoint. At day 5 no BrdU-labeled cells are seen in both WT and osteoponitn-null mice at day 5. Solid lines, anterior lens capsule, dotted lines, the border between lens cortex and lens epithelium, bar, 20 μm.
Adhesion of Lens Epithelial Cell Line to OPN-Coated Glass
Coating the glass slides with exogenous OPN markedly facilitated adhesion of α-TN4 cells (Figure 6).
Discussion
Our previous study revealed that lens epithelial cells upregulate OPN in response to injury prior to EMT,31 although we failed to uncover its biological role. The present study was undertaken to answer this question by using OPN-deficient mice. Here, we show that the absence of OPN disrupts the injury-induced EMT in mice in vivo. As HE histology suggested the impairment of injury-induced EMT in lens epithelium, we conducted immunohistochemistry and discovered reduced (relative to WT lens) expression of αSMA, collagen I and TGFβ1/2 in lens epithelial cells in KO mice at day 5 postinjury. The exact mechanism that explains the phenomenon remains to be investigated. However, we speculate that the Smad2/3 signal might be affected by the absence of OPN, because this signal is critical for the EMT of the lens epithelium. We therefore examined activation of Smad2/3 signals to evaluate the effect of OPN on the TGFβ/activin-Smad signal by employing immunohiostochemistry. Eye aqueous humor contains abundant TGFβ, especially TGFβ232, 33, 34 and the Smad signal is activated 12 h following a puncture injury in the mouse lens.4 We found that loss of OPN suppressed the Smad2/3 signal in the injured lens epithelium. Phospho-Smad2/3 was not detected in the cells even at day 2 in KO mice, while it was readily seen at day 1 in a WT mouse injured lens. At days 5 and 10 the expression level of phospho-Smad2 was reduced in KO lens epithelium as compared with WT epithelium. This is consistent with our previous report that a Smad3 deficiency or inhibition of Smad signaling by Smad7 gene transfer suppresses injury-induced EMT in mouse lens epithelium.17, 18, 35 Although multiple signaling cascades are required for EMT and expression of collagen and TGFβ1, the TGFβ/Smad signal is considered to be essential. However, the mechanism by which OPN affects TGFβ/Smad signal is to be uncovered. Impaired adhesive nature of the cells might impair activation of cells upon injury. As OPN is an integrin-binding protein, loss of this molecule might affect integrin-mediated cytoplasmic signaling. Alternatively, we propose that OPN might interact TGFβ receptors on the cell surface, for example the TGFβ type III receptor. Our unpublished data show that cultured ocular fibroblasts derived from KO mice show attenuated Smad3 signal upon addition of TGFβ1, supporting the notion (Saika et al, unpublished data 2006).
As for cell proliferation, histological studies revealed greater numbers of cells in the cell multilayer around the capsular break in a KO lens as compared with a WT lens at day 1 and day 2, suggesting that in the absence of OPN there was increased cell proliferation in the lens epithelium upon injury. Indeed, BrdU immunostaining showed more BrdU-labeled epithelial cells in KO mice at day 1 and day 2 postinjury. Attenuation of Smad2/3 signaling in the absence of OPN is considered to counteract inhibition of cell proliferation by endogenous TGFβ2, because we previously reported that neutralization of endogenous TGFβ2 by an antibody or Smad7 gene transfer to lens epithelium induces more cell proliferation in response to lens injury in mice.35
There are other reports that a deficiency in OPN results in an attenuation of the TGFβ-related fibrogenic reaction. For example, the process of the development of bleomycin-induced lung fibrosis, which is characterized by cystic dilated air spaces, decreased type I collagen expression, and a reduction of active TGFβ1 and MMP-2, is reduced in the absence of OPN.36 Similarly, OPN reportedly affects the process of wound healing or tissue fibrosis in many other tissues and in the majority of cases a deficiency in OPN attenuates the fibrogenic reaction as in the lung.37, 38, 39, 40, 41, 42, 43, 44 To uncover the role of OPN in lens epithelium EMT is considered to contribute not only to developing new strategies for suppression of PCO, but also to understand its role in EMT in other pathological conditions, that is, cancer progression and fibrogenic reaction by renal, lung or biliary epithelial cells, or retinal pigment epithelium.2, 15, 45
Recently, the concept of ‘matrikine’ has been proposed.46, 47, 48, 49 This term designates ECM-derived peptides that are able to regulate cell activity proliferation, migration, differentiation, and dedifferentiation via binding to their specific receptors. Our previous studies revealed loss of lumican, a core protein of keratan sulfate proteoglycan, perturbs EMT in lens epithelium upon injury,21 and also that lumican, promotes corneal epithelial healing,50 and also up-regulates expression of keratocan, another corneal keratan sulfate proteoglycan.51 These findings suggest that lumican not only forms ECM structure but also modulates cell behavior as a ‘matrikine’. It has been proposed that soluble OPN delivers signals to cells like those delivered by the insoluble ECM.26
References
Hay ED, Zuk A . Transformations between epithelium and mesenchyme: normal, pathological, and experimentally induced. Am J Kidney Dis 1995;26:678–690.
Roberts AB, Tian F, Byfield SD, et al. Smad3 is key to TGF-β-mediated epithelial-to-mesenchymal transition, fibrosis, tumor suppression and metastasis. Cytokine Growth Factor Rev 2006;17:19–27.
Saika S, Kawashima Y, Miyamoto T, et al. Immunolocalization of prolyl 4-hydroxylase subunits, a-smooth muscle actin, and extracellular matrix components in human lens capsules with lens implants. Exp Eye Res 1998;66:283–294.
Saika S, Okada Y, Miyamoto T, et al. Smad translocation and growth suppression in lens epithelial cells by endogenous TGFβ2 during wound repair. Exp Eye Res 2001;72:679–686.
Meacock WR, Spalton DJ, Stanford MR . Role of cytokines in the pathogenesis of posterior capsule opacification. Br J Ophthalmol 2000;84:332–336.
Wormstone IM . Posterior capsular opacification: a cell biological perspective. Exp Eye Res 2002;74:337–347.
Saika S . Relationship between posterior capsule opacification and intraocular lens biocompatibility. Prog Retin Eye Res 2004;23:283–305.
Serini G, Bochaton-Piallat M-L, Ropraz P, et al. The fibronectin domain ED-A is crucial for myofibroblastic phenotype induction by transforming growth factor-β1. J Cell Biol 1998;142:873–881.
Serini G, Gabbiani G . Mechanism of myofibroblast activity and phenotypic modulation. Exp Cell Res 1999;250:273–283.
Tomasek JJ, Gabbiani G, Hinz B, et al. Myofibroblasts and mechano-regulation of connective tissue remodelling. Nat Rev Mol Cell Biol 2002;3:349–363.
Saika S, Miyamoto T, Tanaka T, et al. Latent TGFβ binding protein-1 and fibrillin-1 in human capsular opacification and in cultured lens epithelial cells. Br J Ophthalmol 2001;85:1362–1366.
Miettinen PJ, Ebner R, Lopez AR, et al. TGF-β induced transdifferenttiation of mammary epithelial cells to mesenchymal cells: involvement of type I receptors. J Cell Biol 1994;127:2021–2036.
Srinivasan Y, Lovicu FJ, Overbeek PA . Lens specific expression of transforming growth factor β1 in transgenic mice causes anterior subcapsular cataracts. J Clin Invest 1998;101:625–634.
Lee EH, Joo C-K . Role of transforming growth factor-β in transdifferentiation and fibrosis of lens epithelial cells. Invest Ophthalmol Vis Sci 1999;40:2025–2032.
Oft M, Akhurst RJ, Balmain A . Metastasis is driven by sequential elevation of H-ras and Smad2 levels. Nature Cell Biol 2002;4:487–494.
Wormstone IM, Tamiya S, Andersone I, et al. TGF-β2-induced matrix modification and cell transdifferentiation in the human lens capsular bag. Invest Ophthalmol Vis Sci 2002;43:2301–2308.
Saika S, Kono-Saika S, Ohnishi Y, et al. Smad3 signaling is required for epithelial-mesenchymal transition of lens epithelium after injury. Am J Pathol 2004;164:651–663.
Saika S . TGFβ pathobiology in the eye. Lab Invest 2006;86:106–115.
Hay ED . Extracellular matrix alters epithelial differentiation. Curr Opin Cell Biol 1989;14:455–457.
Zuk A, Hay ED . Expression of β1 integrins changes during transformation of avian lens epithelium to mesenchyme in collagen gels. Dev Dyn 1994;201:378–393.
Saika S, Miyamoto T, Tanaka S, et al. Response of lens epithelial cells to injury: role of lumican in epithelial-mesenchymal transition. Invest Ophthalmol Vis Sci 2003;44:2094–2102.
Zeisberg M, Bonner G, Maeshima Y, et al. Collagen composition and assembly regulates epithelial-mesenchymal transdifferentiation. Am J Pathol 2001;159:1313–1321.
Ashkar S, Weber GF, Panoutsakopoulou V, et al. Eta-1 (osteopontin): an early component of type I (cell-mediated) immunity. Science 2000;287:860–864.
O'Regan AW, Nau GJ, Chupp GL, et al. Osteopontin (Eta-1) in cell-mediated immunity: teaching an old dog new tricks. Immunol Today 2000;21:475–478.
Chabas D, Baranzini SE, Mitchell D, et al. The influence of the proinflammatory cytokine, osteopontin, on autoimmune demyelinating disease. Science 2001;294:1731–1735.
Denhardt DT, Noda M, O'Regan AW, et al. Osteopontin as a means to cope with environmental insults: regulation of inflammation, tissue remodeling, and cell survival. J Clin Invest 2001;107:1055–1061.
Gravallese EM . Osteopontin: a bridge between bone and the immune system. J Clin Invest 2003;112:147–149 (Review). Erratum in: J Clin Invest. 2003;112:627.
O'Regan A . The role of osteopontin in lung disease. Cytokine Growth Factor Rev 2003;14:479–488.
Standal T, Borset M, Sundan A . Role of osteopontin in adhesion, migration, cell survival and bone remodeling. Exp Oncol 2004;26:179–184.
Rangaswami H, Bulbule A, Kundu GC . Osteopontin: role in cell signaling and cancer progression. Trends Cell Biol 2006;16:79–87.
Saika S, Miyamoto T, Ishida I, et al. Osteopontin: a component of matrix in capsular opacification and subcapsular cataract. Invest Ophthalmol Vis Sci 2003;44:1622–1628.
Jampel HD, Roche N, Stark WJ, et al. Transforming growth factor-beta in human aqueous humor. Curr Eye Res 1990;9:963–969.
Cousins SW, McCabe MM, Danielpour D, et al. Identification of transforming growth factor-beta as an immunosuppressive factor in aqueous humor. Invest Ophthalmol Vis Sci 199;32:2201–2211.
Saika S, Saika S, Liu CY, et al. TGFβ2 in corneal morphogenesis during mouse embryonic development. Dev Biol 2001;240:419–432.
Saika S, Ikeda K, Yamanaka O, et al. Transient adenoviral gene transfer of Smad7 prevents injury-induced epithelial-mesenchymal transition of lens epithelium in mice. Lab Invest 2004;84:1259–1270.
Berman JS, Serlin D, Li X, et al. Altered bleomycin-induced lung fibrosis in osteopontin-deficient mice. Am J Physiol Lung Cell Mol Physiol 2004;286:L1311–L1318.
Gadeau AP, Campan M, Millet D, et al. Osteopontin overexpression is associated with arterial smooth muscle cell proliferation in vitro. Arterioscler Thromb 1993;13:120–125.
Gardner HA, Berse B, Senger DR . Specific reduction in osteopontin synthesis by antisense RNA inhibits the tumorigenicity of transformed Rat1 fibroblasts. Oncogene 1994;9:2321–2326.
Ashizawa N, Graf K, Do YS, et al. Osteopontin is produced by rat cardiac fibroblasts and mediates A(II)-induced DNA synthesis and collagen gel contraction. J Clin Invest 1996;98:2218–2227.
Liaw L, Birk D, Ballas C, et al. Altered wound healing in mice lacking a functional osteopontin gene (spp1). J Clin Invest 1998;101:1468–1478.
Ophascharoensuk V, Giachelli CM, Gordon K, et al. Obstructive uropathy in the mouse: role of osteopontin in interstitial fibrosis and apoptosis. Kidney Int 1999;56:571–580.
Trueblood NA, Xie Z, Communal C, et al. Exaggerated left ventricular dilation and reduced collagen deposition after myocardial infarction in mice lacking osteopontin. Circ Res 2001;88:1080–1087.
Lee SH, Seo GS, Park YN, et al. Effects and regulation of osteopontin in rat hepatic stellate cells. Biochem Pharmacol 2004;68:2367–2378.
Lorena D, Darby IA, Gadeau AP, et al. Osteopontin expression in normal and fibrotic liver. altered liver healing in osteopontin-deficient mice. J Hepatol 2006;44:383–390.
Saika S, Kono-Saika S, Tanaka T, et al. Smad3 is required for dedifferentiation of retinal pigment epithelium following retinal detachment in mice. Lab Invest 2004;84:1245–1258.
Tran KT, Griffith L, Wells A . Extracellular matrix signaling through growth factor receptors during wound healing. Wound Repair Regen 2004;12:262–268.
Duca L, Floquet N, Alix AJ, et al. Elastin as a matrikine. Crit Rev Oncol Hematol 2004;49:235–244.
Tran KT, Lamb P, Deng JS . Matrikines and matricryptins: Implications for cutaneous cancers and skin repair. J Dermatol Sci 2005;40:11–20.
Iozzo RV . Heparan sulfate proteoglycans: intricate molecules with intriguing functions. J Clin Invest 2001;108:165–167.
Saika S, Shiraishi A, Liu CY, et al. Role of lumican in the corneal epithelium during wound healing. J Biol Chem 2000;275:2607–2612.
Carlson EC, Liu CY, Chikama T, et al. Keratocan, a cornea-specific keratan sulfate proteoglycan, is regulated by lumican. J Biol Chem 2005;280:25541–25547.
Acknowledgements
This study was supported by a Grant from the Ministry of Education, Science, Sports and Culture of Japan (C11591871, to SS), NIH Grant EY 13755, Research to Prevent Blindness, and Ohio Lions Eye Research Foundation (to WW-YK).
Author information
Authors and Affiliations
Corresponding author
Additional information
Financial interests
None for all authors.
Rights and permissions
About this article
Cite this article
Saika, S., Shirai, K., Yamanaka, O. et al. Loss of osteopontin perturbs the epithelial-mesenchymal transition in an injured mouse lens epithelium. Lab Invest 87, 130–138 (2007). https://doi.org/10.1038/labinvest.3700508
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.3700508