Abstract
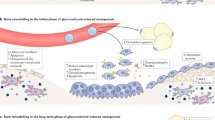
Glucocorticoid-induced osteoporosis is a common condition that results in significant morbidity and mortality. The skeletal effects of glucocorticoids include both direct and indirect actions on bone that result in an early, transient increase in bone resorption accompanied by a decrease in bone formation, which is maintained for the duration of glucocorticoid therapy. Rapid bone loss and increased fracture risk occur soon after the initiation of glucocorticoid therapy and are dose dependent. The increase in fracture risk is partly independent of bone mineral density, probably as a result of changes in bone material properties and an increased risk of falling. Bisphosphonates are the front-line choice for prevention of fracture in glucocorticoid-treated patients, with teriparatide as the second-line option; calcium and vitamin D supplements should be co-prescribed in the majority of individuals. Future guidelines for the management of glucocorticoid-induced osteoporosis should recognize the limitations of FRAX® in assessing fracture risk in glucocorticoid-treated patients, and should include recently approved interventions, such as zoledronate and teriparatide.
Key Points
-
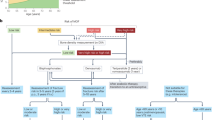
Glucocorticoid therapy is a common cause of osteoporosis, but remains under-recognized and under-treated
-
Direct effects of glucocorticoids on bone include an early, transient increase in bone resorption and long-term suppression of bone formation at the tissue and cellular levels
-
Rapid bone loss and increased fracture risk occur early in the course of glucocorticoid therapy, emphasizing the importance of primary prevention in those at high risk of fracture
-
Use of the FRAX® fracture risk assessment tool will often underestimate fracture probability in glucocorticoid-treated individuals
-
Bisphosphonates are the first-line treatment option for the prevention of fracture in patients receiving glucocorticoids
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Compston, J. E. Emerging consensus on prevention and treatment of glucocorticoid-induced osteoporosis. Curr. Rheumatol. Rep. 9, 78–84 (2007).
Curtis, J. R. et al. Longitudinal patterns in the prevention of osteoporosis in glucocorticoid-treated patients. Arthritis Rheum. 52, 2485–2494 (2005).
Feldstein, A. C., Elmer, P. J., Nichols, G. A. & Herson, M. Practice patterns in patients at risk for glucocorticoid-induced osteoporosis. Osteoporos. Int. 16, 2168–2174 (2005).
Duyvendak, M., Naunton, M., Atthobari, J., van den Berg, P. B. & Brouwers, J. R. Corticosteroid-induced osteoporosis prevention: longitudinal practice patterns in The Netherlands 2001–2005. Osteoporos. Int. 18, 1429–1433 (2007).
Newman, E. D. et al. Glucocorticoid-Induced Osteoporosis Program (GIOP): a novel, comprehensive, and highly successful care program with improved outcomes at 1 year. Osteoporos. Int. 17, 1428–1434 (2006).
Blalock, S. J., Norton, L. L., Patel, R. A. & Dooley, M. A. Patient knowledge, beliefs, and behavior concerning the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Rheum. 53, 732–739 (2005).
van Staa, T. P. et al. Use of oral corticosteroids in the United Kingdom. QJM 93, 105–111 (2000).
van Staa, T. P., Leufkens, H. G., Abenhaim, L., Zhang, B. & Cooper, C. Use of oral corticosteroids and risk of fractures. J. Bone Miner. Res. 15, 933–1000 (2000).
De Vries, F. et al. Fracture risk with intermittent high-dose glucocorticoid therapy. Arthritis Rheum. 56, 208–214 (2007).
van Staa, T. P., Leufkens, H. G., Abenhaim, L., Zhang, B. & Cooper, C. Oral corticosteroids and fracture risk: relationship to daily and cumulative doses. Rheumatology (Oxford) 39, 1383–1389 (2000).
van Staa, T. P., Leufkens, H. G. & Cooper, C. The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos. Int. 13, 777–787 (2002).
Ip, M., Lam, K., Yam, L., Kung, A. & Ng, M. Decreased bone mineral density in premenopausal asthma patients receiving long-term inhaled steroids. Chest 105, 1722–1727 (1994).
Toogood, J. H. et al. Bone mineral density and the risk of fracture in patients receiving long-term inhaled steroid therapy for asthma. J. Allergy Clin. Immunol. 96, 157–166 (1995).
Wong, C. A. et al. Inhaled corticosteroid use and bone-mineral density in patients with asthma. Lancet 355, 1399–1403 (2000).
van Staa, T. P., Leufkens, H. G. & Cooper, C. Use of inhaled corticosteroids and risk of fractures. J. Bone Miner. Res. 16, 581–588 (2001).
van Staa, T. P., Bishop, N., Leufkens, H. G. & Cooper, C. Are inhaled corticosteroids associated with an increased risk of fracture in children? Osteoporos. Int. 15, 785–791 (2004).
Dalle Carbonare, L. et al. Comparison of trabecular bone microarchitecture and remodeling in glucocorticoid-induced and postmenopausal osteoporosis. J. Bone Miner. Res. 16, 97–103 (2001).
Chappard, D. et al. Altered trabecular architecture induced by corticosteroids: a bone histomorphometric study. J. Bone Miner. Res. 11, 676–685 (1996).
Bressot, C. et al. Histomorphometric profile, pathophysiology and reversibility of corticosteroid induced osteoporosis. Metab. Bone Dis. Relat. Res. 1, 303–311 (1979).
Dempster, D. W. Bone histomorphometry in glucocorticoid-induced osteoporosis. J. Bone Miner. Res. 4, 137–147 (1989).
Dempster, D. W., Arlot, M. A. & Meunier, P. J. Mean wall thickness and formation periods of trabecular bone packets in corticosteroid-induced osteoporosis. Calcif. Tissue Int. 35, 410–417 (1983).
Compston, J. et al. Recommendations for the registration of agents for prevention and treatment of glucocorticoid-induced osteoporosis: an update from the Group for the Respect of Ethics and Excellence in Science. Osteoporos. Int. 19, 1247–1250 (2008).
Rubin, J. et al. Dexamethasone promotes expression of membrane-bound macrophage colony-stimulating factor in murine osteoblast-like cells. Endocrinology 139, 1006–1012 (1998).
Hofbauer, L. C. et al. Stimulation of osteoprotegerin ligand and inhibition of osteoprotegerin production by glucocorticoids in human osteoblastic lineage cells: potential paracrine mechanisms of glucocorticoid-induced osteoporosis. Endocrinology 140, 4382–4389 (1999).
Canalis, E. Mechanisms of glucocorticoid action in bone. Curr. Osteoporos. Rep. 3, 98–102 (2005).
Ohnaka, K., Tanabe, M., Kawate, H., Nawata, H. & Takayanagi, R. Glucocorticoid suppresses the canonical Wnt signal in cultured human osteoblasts. Biochem. Biophys. Res. Commun. 329, 177–181 (2005).
Smith, E. & Frenkel, B. Glucocorticoids inhibit the transcriptional activity of LEF/TCF in differentiating osteoblasts in a glycogen synthase kinase-3β-dependent and -independent manner. J. Biol. Chem. 280, 2388–2394 (2005).
Wu, Z., Bucher, N. L. & Farmer, S. R. Induction of peroxisome proliferator-activated receptor γ during the conversion of 3T3 fibroblasts into adipocytes is mediated by C/EBPβ, C/EBPδ, and glucocorticoids. Mol. Cell Biol. 16, 4128–4136 (1996).
Weinstein, R. S., Jilka, R. L., Parfitt, A. M. & Manolagas, S. C. Inhibition of osteoblastogenesis and promotion of apoptosis of osteoblasts and osteocytes by glucocorticoids. Potential mechanisms of their deleterious effects on bone. J. Clin. Invest. 102, 274–282 (1998).
Weinstein, R. S. Glucocorticoids, osteocytes and skeletal fragility: the role of bone vascularity. Bone doi:10.1016/j.bone.2009.06.030.
Lane, N. E. et al. Glucocorticoid-treated mice have localized changes in trabecular bone material properties and osteocyte lacunar size that are not observed in placebo-treated or estrogen-deficient mice. J. Bone Miner. Res. 21, 466–476 (2006).
Kanis, J. A. Diagnosis of osteoporosis and assessment of fracture risk. Lancet 359, 1929–1936 (2002).
University of Sheffield. FRAX®x2014;WHO Fracture Risk Assessment Tool [online]
Kanis, J. A. et al. A meta-analysis of prior corticosteroid use and fracture risk. J. Bone Miner. Res. 19, 893–899 (2004).
Luengo, M. et al. Treatment of steroid-induced osteopenia with calcitonin in corticosteroid-dependent asthma. A one-year follow-up study. Am. Rev. Respir. Dis. 142, 104–107 (1990).
van Staa, T. P. et al. Bone density threshold and other predictors of vertebral fracture in patients receiving oral glucocorticoid therapy. Arthritis Rheum. 48, 3224–3229 (2003).
Homik, J. et al. Bisphosphonates for steroid induced osteoporosis. Cochrane Database of Systematic Reviews 1999, Issue 1. Art. No.: CD001347.doi:10.1002/14651858.CD001347.
Adachi, J. D. et al. Intermittent etidronate therapy to prevent corticosteroid-induced osteoporosis. N. Engl. J. Med. 337, 382–387 (1997).
Saag, K. G. et al. Alendronate for the prevention and treatment of glucocorticoid-induced osteoporosis. Glucocorticoid-Induced Osteoporosis Intervention Study Group. N. Engl. J. Med. 339, 292–299 (1998).
Adachi, J. D. et al. Two-year effects of alendronate on bone mineral density and vertebral fracture in patients receiving glucocorticoids: a randomized, double-blind, placebo-controlled extension trial. Arthritis Rheum. 44, 202–211 (2001).
De Nijs, R. N. et al. Alendronate or alfacalcidol in glucocorticoid-induced osteoporosis. N. Engl. J. Med. 355, 675–684 (2006).
Stoch, S. A. et al. Once-weekly oral alendronate 70 mg in patients with glucocorticoid-induced bone loss: a 12-month randomized, placebo-controlled clinical trial. J. Rheumatol. 36, 1705–1714 (2009).
Cohen, S. et al. Risedronate therapy prevents corticosteroid-induced bone loss: a twelve-month, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Arthritis Rheum. 42, 2309–2318 (1999).
Reid, D. M. et al. Efficacy and safety of daily risedronate in the treatment of corticosteroid-induced osteoporosis in men and women: a randomized trial. European Corticosteroid-Induced Osteoporosis Treatment Study. J. Bone Miner. Res. 15, 1006–1013 (2000).
Wallach, S. et al. Effects of risedronate treatment on bone density and vertebral fracture in patients on corticosteroid therapy. Calcif. Tissue Int. 67, 277–285 (2000).
Reid, D. M., Adami, S., Devogelaer, J. P. & Chines, A. A. Risedronate increases bone density and reduces vertebral fracture risk within one year in men on corticosteroid therapy. Calcif. Tissue Int. 69, 242–247 (2001).
Ringe, J. D., Dorst, A., Faber, H., Ibach, K. & Sorenson, F. Intermittent intravenous ibandronate injections reduce vertebral fracture risk in corticosteroid-induced osteoporosis: results from a long-term comparative study. Osteoporos. Int. 14, 801–807 (2003).
Reid, D. M. et al. Zoledronic acid and risedronate in the prevention and treatment of glucocorticoid-induced osteoporosis (HORIZON): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet 373, 1253–1263 (2009).
Weinstein, R. S. et al. Promotion of osteoclast survival and antagonism of bisphosphonate-induced osteoclast apoptosis by glucocorticoids. J. Clin. Invest. 109, 1041–1048 (2002).
Khan, A. A. et al. Bisphosphonate associated osteonecrosis of the jaw. J. Rheumatol. 36, 478–490 (2009).
Ing-Lorenzini, K. et al. Low-energy femoral fractures associated with the long-term use of bisphosphonates: a case series from a Swiss university hospital. Drug Saf. 32, 775–785 (2009).
Compston, J. E. Skeletal actions of intermittent parathyroid hormone: effects on bone remodeling and structure. Bone 40, 1447–1452 (2007).
Lane, N. E. et al. Parathyroid hormone treatment can reverse corticosteroid-induced osteoporosis. Results of a randomized controlled clinical trial. J. Clin. Invest. 102, 1627–1633 (1998).
Lane, N. E. et al. Bone mass continues to increase at the hip after parathyroid hormone treatment is discontinued in glucocorticoid-induced osteoporosis: results of a randomized controlled clinical trial. J. Bone Miner. Res. 15, 944–951 (2000).
Saag, K. G. et al. Teriparatide or alendronate in glucocorticoid-induced osteoporosis. N. Engl. J. Med. 357, 2028–2039 (2007).
Langdahl, B. L. et al. Teriparatide versus alendronate for treating glucocorticoid-induced osteoporosis: an analysis by gender and menopausal status. Osteoporos. Int. doi:10.1007/s00198-009-0917-y.
Neer, R. M. et al. Effect of parathyroid hormone (1-34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N. Engl. J. Med. 344, 1434–1441 (2001).
Oxlund, H. et al. The anabolic effect of PTH on bone is attenuated by simultaneous glucocorticoid treatment. Bone 39, 244–252 (2006).
Nishida, S. et al. Increased bone formation by intermittent parathyroid hormone administration is due to the stimulation of proliferation and differentiation of osteoprogenitor cells in bone marrow. Bone 15, 717–723 (1994).
Jilka, R. L. et al. Increased bone formation by prevention of osteoblast apoptosis by parathyroid hormone. J. Clin. Invest. 104, 439–446 (1999).
Saag, K. G. et al. Effects of teriparatide versus alendronate for treating glucocorticoid-induced osteoporosis: thirty-six-month results of a randomized, double-blind, controlled trial. Arthritis Rheum. 60, 3346–3355 (2009).
Richy, F., Ethgen, O., Bruyere, O. & Reginster, J. Y. Efficacy of alphacalcidol and calcitriol in primary and corticosteroid-induced osteoporosis: a meta-analysis of their effects on bone mineral density and fracture rate. Osteoporos. Int. 15, 301–310 (2004).
Richy, F. et al. Vitamin D analogs versus native vitamin D in preventing bone loss and osteoporosis-related fractures: a comparative meta-analysis. Calcif. Tissue Int. 76, 176–186 (2005).
De Nijs, R. N. et al. Alendronate or alfacalcidol in glucocorticoid-induced osteoporosis. N. Engl. J. Med. 355, 675–684 (2006).
Devogelaer, J. P. et al. Evidence-based guidelines for the prevention and treatment of glucocorticoid-induced osteoporosis: a consensus document of the Belgian Bone Club. Osteoporos. Int. 17, 8–19 (2006).
Gourlay, M., Franceschini, N. & Sheyn, Y. Prevention and treatment strategies for glucocorticoid-induced osteoporosis. Clin. Rheumatol. 26, 144–153 (2007).
Guidelines Working Group for the Bone and Tooth Society, National Osteoporosis Society and Royal College of Physicians. Glucocorticoid-induced osteoporosis: guidelines for prevention and treatment (Royal College of Physicians, London, 2002).
American College of Rheumatology ad hoc Committee on Glucocorticoid-induced Osteoporosis. Recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis: 2001 update. Arthritis Rheum. 44, 1496–1503 (2001).
Brown, J. P. et al. 2002 clinical practice guidelines for the diagnosis and management of osteoporosis in Canada. CMAJ 167 (10 Suppl.), S1–S34 (2002).
Nawata, H. et al. Guidelines on the management and treatment of glucocorticoid-induced osteoporosis of the Japanese Society of Bone and Mineral Research (2004). J. Bone Miner. Metab. 23, 105–109 (2005).
Geusens, P. et al. Review and evaluation of the Dutch guidelines for osteoporosis. J. Eval. Clin. Pract. 12, 539–548 (2006).
National Osteoporosis Foundation. Clinician's guide to prevention and treatment of osteoporosis [online] (2008).
Compston, J. et al. Guidelines for the diagnosis and management of osteoporosis in postmenopausal women and men from the age of 50 years in the UK. Maturitas 62, 105–108 (2009).
Acknowledgements
The author acknowledges the support of the NHS National Institute of Health Research (UK). Désirée Lie, University of California, Orange, CA, is the author of and is solely responsible for the content of the learning objectives, questions and answers of the MedscapeCME-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author, the Journal Editor J. Buckland and the CME questions author D. Lie declare no competing interests.
Rights and permissions
About this article
Cite this article
Compston, J. Management of glucocorticoid-induced osteoporosis. Nat Rev Rheumatol 6, 82–88 (2010). https://doi.org/10.1038/nrrheum.2009.259
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2009.259
This article is cited by
-
Bone turnover decreases and bone structure improves during treatment with weekly high-dose methylprednisolone for 12 weeks in Graves’ orbitopathy
Endocrine (2023)
-
Prednisolone induces osteocytes apoptosis by promoting Notum expression and inhibiting PI3K/AKT/GSK3β/β-catenin pathway
Journal of Molecular Histology (2021)
-
Effects of denosumab on bone mineral density and bone turnover markers in rheumatoid arthritis patients switching from bisphosphonates
Journal of Experimental Orthopaedics (2019)
-
Long-Term Therapy with Corticosteroids in Nasal Polyposis: A Bone Metabolism Assessment
Indian Journal of Otolaryngology and Head & Neck Surgery (2019)
-
Denosumab for glucocorticoid-induced osteoporosis
Nature Reviews Endocrinology (2018)