Abstract
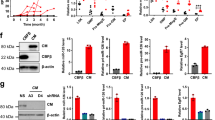
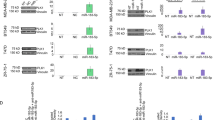
Multiple myeloma (MM) is the second most prevalent hematologic malignancy. Aberrant microRNAs (miRNAs) expression has been shown to be involved in the pathogenesis of MM. In this study, we further demonstrated that miR-137 was significantly downregulated in MM and negatively correlated with clinical prognosis. Moreover, we described the epigenetic regulation of miR-137 and its association with progression-free survival in MM patients. Furthermore, overexpression of miR-137 in MM cell line (miR-137 OE) increased its sensitivity to bortezomib and eprirubicin in vitro. Also, some high-risk genetic abnormalities in MM, including deletion of chromosome 1p22.2, 14q or 17p13, and gain of chromosome 1p22.2 were detected in NCI-H929 empty vector (NCI-H929 EV) treated cells but not in the NCI-H929 miR-137 overexpression (NCI-H929 miR-137 OE) cells. Luciferase reporter assays demonstrated that miR-137 targeted AURKA. Ectopic expression of miR-137 strongly reduced the expression of AURKA and p-ATM/Chk2 in MM cells, and increased the expression of p53, and p21. Importantly, miR-137 overexpression together with bortezomib treatment significantly inhibited tumor growth in MM xenograft model. Taken together, this study demonstrates that miR-137 is epigenetically silenced in MM, and overexpression of miR-137 could reduce drug resistance and overcome chromosomal instability of the MM cells via affecting the apoptosis and DNA damage pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Raab MS, Podar K, Breitkreutz I, Richardson PG, Anderson KC . Multiple myeloma. Lancet 2009; 374: 324–339.
Kyle RA, Rajkumar SV . Multiple myeloma. Blood 2008; 111: 2962–2972.
Cottini F, Anderson K . Novel therapeutic targets in multiple myeloma. Clin Adv Hematol Oncol 2015; 13: 236–248.
Yang H, Kong W, He L, Zhao JJ, O'Donnell JD, Wang J et al. MicroRNA expression profiling in human ovarian cancer: miR-214 induces cell survival and cisplatin resistance by targeting PTEN. Cancer Res 2008; 68: 425–433.
Kovalchuk O, Filkowski J, Meservy J, Ilnytskyy Y, Tryndyak VP, Chekhun VF et al. Involvement of microRNA-451 in resistance of the MCF-7 breast cancer cells to chemotherapeutic drug doxorubicin. Mol Cancer Ther 2008; 7: 2152–2159.
Miller TE, Ghoshal K, Ramaswamy B, Miller TE, Ghoshal K, Ramaswamy B et al. MicroRNA-221/222 confers tamoxifen resistance in breast cancer by targeting p27Kip1. J Biol Chem 2008; 283: 29897–29903.
Sun CY, She XM, Qin Y, Chu ZB, Chen L, Ai LS et al. miR-15a and miR-16 affect the angiogenesis of multiple myeloma by targeting VEGF. Carcinogenesis 2012; 34: 426–435.
Roccaro AM, Sacco A, Thompson B, Leleu X, Azab AK, Azab F et al. MicroRNAs 15a and 16 regulate tumor proliferation in multiple myeloma. Blood 2009; 113: 6669–6680.
Chen L, Li C, Zhang R, Gao X, Qu X, Zhao M et al. miR-17-92 cluster microRNAs confers tumorigenicity in multiple myeloma. Cancer Lett 2011; 309: 62–70.
Gao X, Zhang R, Qu X, Zhao M, Zhang S, Wu H et al. MiR-15a, miR-16-1 and miR-17-92 cluster expression are linked to poor prognosis in multiple myeloma. Leuk Res 2012; 36: 1505–1509.
Chang H, Qi X, Jiang A, Xu W, Young T, Reece D . 1p21 deletions are strongly associated with 1q21 gains and are an independent adverse prognostic factor for the outcome of high-dose chemotherapy in patients with multiple myeloma. Bone Marrow Transplant 2010; 45: 117–121.
Chang H, Ning Y, Qi X, Yeung J, Xu W . Chromosome 1p21 deletion is a novel prognostic marker in patients with multiple myeloma. Br J Haematol 2007; 139: 51–54.
Yang Y, Li F, Saha MN, Abdi J, Qiu L, Chang H . miR-137 and miR-197 Induce Apoptosis and Suppress Tumorigenicity by Targeting MCL-1 in Multiple Myeloma. Clin Cancer Res 2015; 21: 2399–2411.
Wong KY, Yim RL, So CC, Jin DY, Liang R, Chim CS . Epigenetic inactivation of the MIR34B/C in multiple myeloma. Blood 2011; 118: 5901–5904.
Balaguer F, Link A, Lozano JJ, Cuatrecasas M, Nagasaka T, Boland CR et al. Epigenetic silencing of miR-137 is an early event in colorectal carcinogenesis. Cancer Res 2010; 70: 6609–6618.
Chen X, Wang J, Shen H, Lu J, Li C, Hu DN et al. Epigenetics, microRNAs, and carcinogenesis: functional role of microRNA-137 in uveal melanoma. Invest Ophthalmol Vis Sci 2011; 52: 1193–1199.
Li LC, Dahiya R . MethPrimer: designing primers for methylation PCRs. Bioinformatics 2002; 18: 1427–1431.
Zhu X, Li Y, Shen H, Li H, Long L, Hui L et al. miR-137 inhibits the proliferation of lung cancer cells by targeting Cdc42 and Cdk6. FEBS Lett 2013; 587: 73–81.
Loffler D, Brocke-Heidrich K, Pfeifer G, Stocsits C, Hackermuller J, Kretzschmar AK et al. Interleukin-6 dependent survival of multiple myeloma cells involves the Stat3-mediated induction of microRNA-21 through a highly conserved enhancer. Blood 2007; 110: 1330–1333.
Lionetti M, Biasiolo M, Agnelli L, Todoerti K, Mosca L, Fabris S et al. Identification of microRNA expression patterns and definition of a microRNA/mRNA regulatory network in distinct molecular groups of multiple myeloma. Blood 2009; 114: e20–e26.
Pichiorri F, Suh SS, Ladetto M, Kuehl M, Palumbo T, Drandi D et al. MicroRNAs regulate critical genes associated with multiple myeloma pathogenesis. Proc Natl Acad Sci USA 2008; 105: 12885–12890.
Bemis LT, Chen R, Amato CM, Classen EH, Robinson SE, Coffey DG et al. MicroRNA-137 targets microphthalmia-associated transcription factor in melanoma cell lines. Cancer Res 2008; 68: 1362–1368.
Steponaitiene R, Kupcinskas J, Langner C, Balaguer F, Venclauskas L, Pauzas H et al. Epigenetic silencing of miR-137 is a frequent event in gastric carcinogenesis.LID - 10.1002/mc.22287 [doi]. Mol Carcinog 2015; 55: 376–386.
Takwi AA, Wang YM, Wu J, Michaelis M, Cinatl J, Chen T . miR-137 regulates the constitutive androstane receptor and modulates doxorubicin sensitivity in parental and doxorubicin-resistant neuroblastoma cells. Oncogene 2014; 33: 3717–3729.
Morgan GJ, Walker BA, Davies FE . The genetic architecture of multiple myeloma. Nat Rev Cancer 2012; 12: 335–348.
Hanamura I, Stewart JP, Huang Y, Zhan F, Santra M, Sawyer JR et al. Frequent gain of chromosome band 1q21 in plasma-cell dyscrasias detected by fluorescence in situ hybridization: incidence increases from MGUS to relapsed myeloma and is related to prognosis and disease progression following tandem stem-cell transplantation. Blood 2006; 108: 1724–1732.
An G, Xu Y, Shi L, Shizhen Z, Deng S, Xie Z et al. Chromosome 1q21 gains confer inferior outcomes in multiple myeloma treated with bortezomib but copy number variation and percentage of plasma cells involved have no additional prognostic value. Haematologica 2014; 99: 353–359.
Klein U, Jauch A, Hielscher T, Hillengass J, Raab MS, Seckinger A et al. Chromosomal aberrations +1q21 and del(17p13) predict survival in patients with recurrent multiple myeloma treated with lenalidomide and dexamethasone. Cancer 2011; 117: 2136–2144.
Reece D, Song KW, Fu T, Roland B, Chang H, Horsman DE et al. Influence of cytogenetics in patients with relapsed or refractory multiple myeloma treated with lenalidomide plus dexamethasone: adverse effect of deletion 17p13. Blood 2009; 114: 522–525.
Carter SL, Eklund AC, Kohane IS, Harris LN, Szallasi Z . A signature of chromosomal instability inferred from gene expression profiles predicts clinical outcome in multiple human cancers. Nat Genet 2006; 38: 1043–1048.
Chng WJ, Ahmann GJ, Henderson K, Santana-Davila R, Greipp PR, Gertz MA et al. Clinical implication of centrosome amplification in plasma cell neoplasm. Blood 2006; 107: 3669–3675.
Fu J, Bian M, Jiang Q, Zhang C . Roles of Aurora kinases in mitosis and tumorigenesis. Mol Cancer Res 2007; 5: 1–10.
Hirota T, Kunitoku N, Sasayama T, Marumoto T, Zhang D, Nitta M et al. Aurora-A and an interacting activator, the LIM protein Ajuba, are required for mitotic commitment in human cells. Cell 2003; 114: 585–598.
Furukawa T, Kanai N, Shiwaku HO, Soga N, Uehara A, Horii A . AURKA is one of the downstream targets of MAPK1/ERK2 in pancreatic cancer. Oncogene 2006; 25: 4831–4839.
Cammareri P, Scopelliti A, Todaro M, Eterno V, Francescangeli F, Moyer MP et al. Aurora-a is essential for the tumorigenic capacity and chemoresistance of colorectal cancer stem cells. Cancer Res 2010; 70: 4655–4665.
Mosquera JM, Beltran H, Park K, MacDonald TY, Robinson BD, Tagawa ST et al. Concurrent AURKA and MYCN gene amplifications are harbingers of lethal treatment-related neuroendocrine prostate cancer. Neoplasia 2013; 15: 1–10.
Katsha A, Soutto M, Sehdev V, Peng D, Washington MK, Piazuelo MB et al. Aurora kinase A promotes inflammation and tumorigenesis in mice and human gastric neoplasia. Gastroenterology 2013; 145: 1312–1322 e1–8.
Zou Z, Yuan Z, Zhang Q, Long Z, Chen J, Tang Z et al. Aurora kinase A inhibition-induced autophagy triggers drug resistance in breast cancer cells. Autophagy 2012; 8: 1798–1810.
Katayama H, Sasai K, Kawai H, Yuan ZM, Bondaruk J, Suzuki F et al. Phosphorylation by aurora kinase A induces Mdm2-mediated destabilization and inhibition of p53. Nat Genet 2004; 36: 55–62.
Dar AA, Zaika A, Piazuelo MB, Correa P, Koyama T, Belkhiri A et al. Frequent overexpression of Aurora Kinase A in upper gastrointestinal adenocarcinomas correlates with potent antiapoptotic functions. Cancer 2008; 112: 1688–1698.
Yang H, He L, Kruk P, Nicosia SV, Cheng JQ . Aurora-A induces cell survival and chemoresistance by activation of Akt through a p53-dependent manner in ovarian cancer cells. Int J Cancer 2006; 119: 2304–2312.
Dutta-Simmons J, Zhang Y, Gorgun G, Gatt M, Mani M, Hideshima T et al. Aurora kinase A is a target of Wnt/beta-catenin involved in multiple myeloma disease progression. Blood 2009; 114: 2699–2708.
Gorgun G, Calabrese E, Hideshima T, Ecsedy J, Perrone G, Mani M et al. A novel Aurora-A kinase inhibitor MLN8237 induces cytotoxicity and cell-cycle arrest in multiple myeloma. Blood 2010; 115: 5202–5213.
Zhou W, Yang Y, Xia J, Wang H, Salama ME, Xiong W et al. NEK2 induces drug resistance mainly through activation of efflux drug pumps and is associated with poor prognosis in myeloma and other cancers. Cancer Cell 2013; 23: 48–62.
Shi Y, Reiman T, Li W, Maxwell CA, Sen S, Pilarski L et al. Targeting aurora kinases as therapy in multiple myeloma. Blood 2007; 109: 3915–3921.
Hose D, Reme T, Meissner T, Moreaux J, Seckinger A, Lewis J et al. Inhibition of aurora kinases for tailored risk-adapted treatment of multiple myeloma. Blood 2009; 113: 4331–4340.
Michaelis M, Selt F, Rothweiler F, Wiese M, Cinatl J Jr . ABCG2 impairs the activity of the aurora kinase inhibitor tozasertib but not of alisertib. BMC Res Notes 2015; 8: 484.
Long ZJ, Wang LX, Zheng FM, Chen JJ, Luo Y, Tu XX et al. A novel compound against oncogenic Aurora kinase A overcomes imatinib resistance in chronic myeloid leukemia cells. Int J Oncol 2015; 46: 2488–2496.
Kelly KR, Shea TC, Goy A, Berdeja JG, Reeder CB, McDonagh KT et al. Phase I study of MLN8237—investigational Aurora A kinase inhibitor—in relapsed/refractory multiple myeloma, non-Hodgkin lymphoma and chronic lymphocytic leukemia. Invest New Drugs 2014; 32: 489–499.
Caputo E, Miceli R, Motti ML, Tate R, Fratangelo F, Botti G et al. AurkA inhibitors enhance the effects of B-RAF and MEK inhibitors in melanoma treatment. J Transl Med 2014; 12: 216.
Sun H, Wang Y, Wang Z, Meng J, Qi Z, Yang G . Aurora-A controls cancer cell radio- and chemoresistance via ATM/Chk2-mediated DNA repair networks. Biochim Biophys Acta 2014; 1843: 934–944.
Yang G, Chang B, Yang F, Guo X, Cai KQ, Xiao XS et al. Aurora kinase A promotes ovarian tumorigenesis through dysregulation of the cell cycle and suppression of BRCA2. Clin Cancer Res 2010; 16: 3171–3181.
Acknowledgements
This work was supported by the grants from the National Natural Science Foundation of China (81172255, 81400174, 81400175, 81400169, 81502019, and 81630007), Science and Technology Infrastructure Program of Tianjin(12ZCDZSY17600) and National Basic Research Program of China (973 program: 2012CB966504).
Author contributions
YQ, SZ, XQ and FL performed experiments; SD, GA, YX, MH and YY analyzed the results; WZ, HC and LQ designed the research; YQ and WZ wrote the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Rights and permissions
About this article
Cite this article
Qin, Y., Zhang, S., Deng, S. et al. Epigenetic silencing of miR-137 induces drug resistance and chromosomal instability by targeting AURKA in multiple myeloma. Leukemia 31, 1123–1135 (2017). https://doi.org/10.1038/leu.2016.325
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2016.325
This article is cited by
-
miRNA-seq identification and clinical validation of CD138+ and circulating miR-25 in treatment response of multiple myeloma
Journal of Translational Medicine (2023)
-
Blocking glycine utilization inhibits multiple myeloma progression by disrupting glutathione balance
Nature Communications (2022)
-
miRNA-seq and clinical evaluation in multiple myeloma: miR-181a overexpression predicts short-term disease progression and poor post-treatment outcome
British Journal of Cancer (2022)
-
A NOTCH1/LSD1/BMP2 co-regulatory network mediated by miR-137 negatively regulates osteogenesis of human adipose-derived stem cells
Stem Cell Research & Therapy (2021)
-
Effects of Aurora kinase A on mouse decidualization via Stat3-plk1-cdk1 pathway
Reproductive Biology and Endocrinology (2021)