Abstract
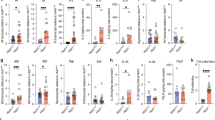
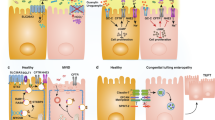
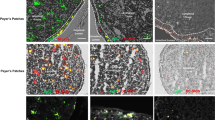
To elucidate the specific role of somatic hypermutation (SHM) in mucosal immunity, we generated mice carrying a knock-in point mutation in Aicda, which encodes activation-induced cytidine deaminase (AID), an enzyme essential to SHM and class-switch recombination (CSR). These mutant AIDG23S mice had much less SHM but had normal amounts of immunoglobulin in both serum and intestinal secretions. AIDG23S mice developed hyperplasia of germinal center B cells in gut-associated lymphoid tissues, accompanied by expansion of microflora in the small intestine. Moreover, AIDG23S mice had more translocation of Yersinia enterocolitica into mesenteric lymph nodes and were more susceptible than wild-type mice to oral challenge with cholera toxin. Together our results indicate that SHM is critical in maintaining intestinal homeostasis and efficient mucosal defense.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Artis, D. Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nat. Rev. Immunol. 8, 411–420 (2008).
Round, J.L. & Mazmanian, S.K. The gut microbiota shapes intestinal immune responses during health and disease. Nat. Rev. Immunol. 9, 313–323 (2009).
Sansonetti, P.J. War and peace at mucosal surfaces. Nat. Rev. Immunol. 4, 953–964 (2004).
Hayday, A.C. & Spencer, J. Barrier immunity. Semin. Immunol. 21, 99–100 (2009).
Honda, K. & Takeda, K. Regulatory mechanisms of immune responses to intestinal bacteria. Mucosal Immunol. 2, 187–196 (2009).
Ishii, K.J., Koyama, S., Nakagawa, A., Coban, C. & Akira, S. Host innate immune receptors and beyond: making sense of microbial infections. Cell Host Microbe 3, 352–363 (2008).
Fagarasan, S., Kawamoto, S., Kanagawa, O. & Suzuki, K. Adaptive immune regulation in the gut: T cell-dependent and T cell-independent IgA synthesis. Annu. Rev. Immunol. 28, 243–273 (2010).
Cerutti, A. & Rescigno, M. The biology of intestinal immunoglobulin A responses. Immunity 28, 740–750 (2008).
Macpherson, A.J., McCoy, K.D., Johansen, F.E. & Brandtzaeg, P. The immune geography of IgA induction and function. Mucosal Immunol. 1, 11–22 (2008).
Woof, J.M. & Mestecky, J. Mucosal immunoglobulins. Immunol. Rev. 206, 64–82 (2005).
Cerutti, A. The regulation of IgA class switching. Nat. Rev. Immunol. 8, 421–434 (2008).
Fagarasan, S. Evolution, development, mechanism and function of IgA in the gut. Curr. Opin. Immunol. 20, 170–177 (2008).
Latiff, A.H. & Kerr, M.A. The clinical significance of immunoglobulin A deficiency. Ann. Clin. Biochem. 44, 131–139 (2007).
Harriman, G.R. et al. Targeted deletion of the IgA constant region in mice leads to IgA deficiency with alterations in expression of other Ig isotypes. J. Immunol. 162, 2521–2529 (1999).
Arulanandam, B.P. et al. IgA immunodeficiency leads to inadequate Th cell priming and increased susceptibility to influenza virus infection. J. Immunol. 166, 226–231 (2001).
Lycke, N., Erlandsson, L., Ekman, L., Schon, K. & Leanderson, T. Lack of J chain inhibits the transport of gut IgA and abrogates the development of intestinal antitoxic protection. J. Immunol. 163, 913–919 (1999).
Uren, T.K. et al. Role of the polymeric Ig receptor in mucosal B cell homeostasis. J. Immunol. 170, 2531–2539 (2003).
Uren, T.K. et al. Vaccine-induced protection against gastrointestinal bacterial infections in the absence of secretory antibodies. Eur. J. Immunol. 35, 180–188 (2005).
Chen, J. et al. Immunoglobulin gene rearrangement in B cell deficient mice generated by targeted deletion of the JH locus. Int. Immunol. 5, 647–656 (1993).
Moore, M.L., McKissic, E.L., Brown, C.C., Wilkinson, J.E. & Spindler, K.R. Fatal disseminated mouse adenovirus type 1 infection in mice lacking B cells or Bruton's tyrosine kinase. J. Virol. 78, 5584–5590 (2004).
Harris, N.L. et al. Mechanisms of neonatal mucosal antibody protection. J. Immunol. 177, 6256–6262 (2006).
Macpherson, A.J. & Uhr, T. Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science 303, 1662–1665 (2004).
Kitamura, D., Roes, J., Kuhn, R. & Rajewsky, K.A. B cell-deficient mouse by targeted disruption of the membrane exon of the immunoglobulin μ chain gene. Nature 350, 423–426 (1991).
Lund, F.E. et al. B cells are required for generation of protective effector and memory CD4 cells in response to Pneumocystis lung infection. J. Immunol. 176, 6147–6154 (2006).
Magez, S. et al. The role of B-cells and IgM antibodies in parasitemia, anemia, and VSG switching in Trypanosoma brucei-infected mice. PLoS Pathog. 4, e1000122 (2008).
Fagarasan, S. et al. Critical roles of activation-induced cytidine deaminase in the homeostasis of gut flora. Science 298, 1424–1427 (2002).
Suzuki, K. et al. Aberrant expansion of segmented filamentous bacteria in IgA-deficient gut. Proc. Natl. Acad. Sci. USA 101, 1981–1986 (2004).
Muramatsu, M. et al. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 102, 553–563 (2000).
Revy, P. et al. Activation-induced cytidine deaminase (AID) deficiency causes the autosomal recessive form of the hyper-IgM syndrome (HIGM2). Cell 102, 565–575 (2000).
Shinkura, R. et al. Separate domains of AID are required for somatic hypermutation and class-switch recombination. Nat. Immunol. 5, 707–712 (2004).
Doi, T. et al. The C-terminal region of activation-induced cytidine deaminase is responsible for a recombination function other than DNA cleavage in class switch recombination. Proc. Natl. Acad. Sci. USA 106, 2758–2763 (2009).
Shivarov, V., Shinkura, R. & Honjo, T. Dissociation of in vitro DNA deamination activity and physiological functions of AID mutants. Proc. Natl. Acad. Sci. USA 105, 15866–15871 (2008).
Komeno, Y. et al. AID-induced T-lymphoma or B-leukemia/lymphoma in a mouse BMT model. Leukemia 24, 1018–1024 (2010).
Dorsett, Y. et al. MicroRNA-155 suppresses activation-induced cytidine deaminase-mediated Myc-Igh translocation. Immunity 28, 630–638 (2008).
Takizawa, M. et al. AID expression levels determine the extent of cMyc oncogenic translocations and the incidence of B cell tumor development. J. Exp. Med. 205, 1949–1957 (2008).
Jolly, C.J., Klix, N. & Neuberger, M.S. Rapid methods for the analysis of immunoglobulin gene hypermutation: application to transgenic and gene targeted mice. Nucleic Acids Res. 25, 1913–1919 (1997).
Sait, L.C. et al. Secretory antibodies reduce systemic antibody responses against the gastrointestinal commensal flora. Int. Immunol. 19, 257–265 (2007).
Wijburg, O.L. et al. Innate secretory antibodies protect against natural Salmonella typhimurium infection. J. Exp. Med. 203, 21–26 (2006).
Sanchez, J. & Holmgren, J. Cholera toxin structure, gene regulation and pathophysiological and immunological aspects. Cell. Mol. Life Sci. 65, 1347–1360 (2008).
Salzman, N.H. et al. Enteric defensins are essential regulators of intestinal microbial ecology. Nat. Immunol. 11, 76–83 (2010).
Ivanov, I.I. et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 (2009).
Ubeda, C. et al. Vancomycin-resistant Enterococcus domination of intestinal microbiota is enabled by antibiotic treatment in mice and precedes bloodstream invasion in humans. J. Clin. Invest. 120, 4332–4341 (2010).
Kadaoui, K.A. & Corthesy, B. Secretory IgA mediates bacterial translocation to dendritic cells in mouse Peyer's patches with restriction to mucosal compartment. J. Immunol. 179, 7751–7757 (2007).
Macpherson, A.J. IgA adaptation to the presence of commensal bacteria in the intestine. Curr. Top. Microbiol. Immunol. 308, 117–136 (2006).
Takaku, K. et al. Intestinal tumorigenesis in compound mutant mice of both Dpc4 (Smad4) and Apc genes. Cell 92, 645–656 (1998).
Wakeland, E., Morel, L., Achey, K., Yui, M. & Longmate, J. Speed congenics: a classic technique in the fast lane (relatively speaking). Immunol. Today 18, 472–477 (1997).
Kamata, T. et al. Increased frequency of surface IgA-positive plasma cells in the intestinal lamina propria and decreased IgA excretion in hyper IgA (HIGA) mice, a murine model of IgA nephropathy with hyperserum IgA. J. Immunol. 165, 1387–1394 (2000).
Wang, Q., Garrity, G.M., Tiedje, J.M. & Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73, 5261–5267 (2007).
deVos, T. & Dick, T.A. A rapid method to determine the isotype and specificity of coproantibodies in mice infected with Trichinella or fed cholera toxin. J. Immunol. Methods 141, 285–288 (1991).
Kelly, B.S., Levy, J.G. & Sikora, L. The use of the enzyme-linked immunosorbent assay (ELISA) for the detection and quantification of specific antibody from cell cultures. Immunology 37, 45–52 (1979).
Hase, K. et al. Uptake through glycoprotein 2 of FimH+ bacteria by M cells initiates mucosal immune response. Nature 462, 226–230 (2009).
Peng, X. et al. Morphine inhibits mucosal antibody responses and TGF-β mRNA in gut-associated lymphoid tissue following oral cholera toxin in mice. J. Immunol. 167, 3677–3681 (2001).
Acknowledgements
We thank H. Nagaoka, M. Muramatsu, K. Kinoshita and H. Ohno for critical comments, and Y. Shiraki for help in preparing the manuscript. Supported by the Ministry of Education, Culture, Sports, Science and Technology of Japan (Grant-in-Aid for Specially Promoted Research 17002015, and Grant-in Aid for Scientific Research on Priority Areas 16043228) and by the Mitsubishi Pharma Research Foundation.
Author information
Authors and Affiliations
Contributions
M.W., R.S. and T.H. designed the study; M.W., R.S., Y.D. and M.M. did the research; and M.W., R.S., S.F. and T.H. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Tables 1–2 (PDF 495 kb)
Rights and permissions
About this article
Cite this article
Wei, M., Shinkura, R., Doi, Y. et al. Mice carrying a knock-in mutation of Aicda resulting in a defect in somatic hypermutation have impaired gut homeostasis and compromised mucosal defense. Nat Immunol 12, 264–270 (2011). https://doi.org/10.1038/ni.1991
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1991
This article is cited by
-
The P2X7 receptor in mucosal adaptive immunity
Purinergic Signalling (2024)
-
Tango of B cells with T cells in the making of secretory antibodies to gut bacteria
Nature Reviews Gastroenterology & Hepatology (2023)
-
TCDD exposure alters fecal IgA concentrations in male and female mice
BMC Pharmacology and Toxicology (2022)
-
Accurate identification and quantification of commensal microbiota bound by host immunoglobulins
Microbiome (2021)
-
Bruton’s tyrosine kinase regulates gut immune homeostasis through attenuating Th1 response
Cell Death & Disease (2021)