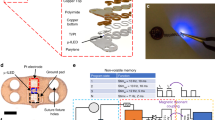
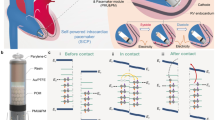
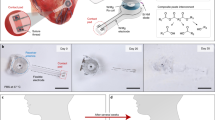
Abstract
Genetic engineering and implantable bioelectronics have transformed investigations of cardiovascular physiology and disease. However, the two approaches have been difficult to combine in the same species: genetic engineering is applied primarily in rodents, and implantable devices generally require larger animal models. We recently developed several miniature cardiac bioelectronic devices suitable for mice and rats to enable the advantages of molecular tools and implantable devices to be combined. Successful implementation of these device-enabled studies requires microsurgery approaches that reliably interface bioelectronics to the beating heart with minimal disruption to native physiology. Here we describe how to perform an open thoracic surgical technique for epicardial implantation of wireless cardiac pacemakers in adult rats that has lower mortality than transvenous implantation approaches. In addition, we provide the methodology for a full biocompatibility assessment of the physiological response to the implanted device. The surgical implantation procedure takes ~40 min for operators experienced in microsurgery to complete, and six to eight surgeries can be completed in 1 d. Implanted pacemakers provide programmed electrical stimulation for over 1 month. This protocol has broad applications to harness implantable bioelectronics to enable fully conscious in vivo studies of cardiovascular physiology in transgenic rodent disease models.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Code availability
Our custom MATLAB software for quantification of our Masson’s trichrome histology images can be downloaded for free at Github: https://github.com/optocardiography/massonstrichromequantification.
References
Mor, M. et al. Speckle-tracking echocardiography elucidates the effect of pacing site on left ventricular synchronization in the normal and infarcted rat myocardium. PLoS ONE 9, e99191 (2014).
Recchia, F. A. & Lionetti, V. Animal models of dilated cardiomyopathy for translational research. Vet. Res. Commun. 31, 35–41 (2007).
Camacho, P., Fan, H., Liu, Z. & He, J.-Q. Small mammalian animal models of heart disease. Am. J. Cardiovasc. Dis. 6, 70–80 (2016).
Klocke, R., Tian, W., Kuhlmann, M. T. & Nikol, S. Surgical animal models of heart failure related to coronary heart disease. Cardiovasc. Res. 74, 29–38 (2007).
Vaseghi, M. & Shivkumar, K. The role of the autonomic nervous system in sudden cardiac death. Prog. Cardiovasc. Dis. 50, 404–419 (2008).
Schrickel, J. W. et al. Induction of atrial fibrillation in mice by rapid transesophageal atrial pacing. Basic Res. Cardiol. 97, 452–460 (2002).
Nyns, E. C. A. et al. An automated hybrid bioelectronic system for autogenous restoration of sinus rhythm in atrial fibrillation. Sci. Transl. Med. 11, 1–12 (2019).
Berul, C. I., Aronovitz, M. J., Wang, P. J. & Mendelsohn, M. E. In vivo cardiac electrophysiology studies in the mouse. Circulation 94, 2641–2648 (1996).
Lujan, H. L. & DiCarlo, S. E. Cardiac electrophysiology and the susceptibility to sustained ventricular tachycardia in intact, conscious mice. Am. J. Physiol. Circ. Physiol. 306, H1213–H1221 (2014).
Ståhlberg, M. et al. Chronic atrial and ventricular pacing in the mouse: application to model cardiac dyssynchrony and resynchronization in heart failure. Circ. Hear. Fail. 12, 1–11 (2019).
Klapper-Goldstein, H. et al. An implantable system for long-term assessment of atrial fibrillation substrate in unanesthetized rats exposed to underlying pathological conditions. Sci. Rep. 10, 553 (2020).
Gutruf, P. et al. Wireless, battery-free, fully implantable multimodal and multisite pacemakers for applications in small animal models. Nat. Commun. 10, 5742 (2019).
Choi, Y. S. et al. Fully implantable and bioresorbable cardiac pacemakers without leads or batteries. Nat. Biotechnol. https://doi.org/10.1038/s41587-021-00948-x (2021).
Wengrowski, A. M., Kuzmiak-Glancy, S., Jaimes, R. & Kay, M. W. NADH changes during hypoxia, ischemia, and increased work differ between isolated heart preparations. Am. J. Physiol. Circ. Physiol. 306, H529–H537 (2014).
Moreno, A., Kowalik, G., Mendelowitz, D. & Kay, M. W. Optogenetic control of cardiac autonomic neurons in transgenic mice. Methods Mol. Biol. 2191, 309–321 (2021).
Yang, Q. et al. Photocurable, bioresorbable adhesives as functional interfaces between flexible bioelectronic devices and soft biological tissues. Nat. Mater. https://doi.org/10.1038/s41563-021-01051-x (2021).
Choi, Y. S. et al. A transient, closed-loop network of wireless, body-integrated devices for autonomous electrotherapy. Science 376, 1006–1012 (2022).
deAlmeida, A. C., van Oort, R. J. & Wehrens, X. H. T. T. Transverse aortic constriction in mice. J. Vis. Exp. https://doi.org/10.3791/1729 (2010).
Michael, L. H. et al. Myocardial ischemia and reperfusion: a murine model. Am. J. Physiol. Circ. Physiol. 269, H2147–H2154 (1995).
George, S. A. et al. p38δ genetic ablation protects female mice from anthracycline cardiotoxicity. Am. J. Physiol. Circ. Physiol. 319, H775–H786 (2020).
Lee, H., Yoon, D. E. & Kim, K. Genome editing methods in animal models. Anim. Cells Syst. 24, 8–16 (2020).
Obaid, S. N. et al. Multifunctional flexible biointerfaces for simultaneous colocalized optophysiology and electrophysiology. Adv. Funct. Mater. 30, 1910027 (2020).
Chen, Z. et al. Flexible and transparent metal nanowire microelectrode arrays and interconnects for electrophysiology, optogenetics, and optical mapping. Adv. Mater. Technol. 6, 2100225 (2021).
Chen, Z. et al. Flexible and transparent metal oxide/metal grid hybrid interfaces for electrophysiology and optogenetics. Adv. Mater. Technol. 5, 2000322 (2020).
Laughner, J. I. et al. A fully implantable pacemaker for the mouse: from battery to wireless power. PLoS ONE 8, e76291 (2013).
Bilchick, K. C. et al. Differential regional gene expression from cardiac dyssynchrony induced by chronic right ventricular free wall pacing in the mouse. Physiol. Genomics 26, 109–115 (2006).
Sridharan, D. et al. A one-stop protocol to assess myocardial fibrosis in frozen and paraffin sections. Methods Protoc. 5, 13 (2022).
Cardiac Bioelectric Therapy: Mechanisms and Practical Implications (Springer, 2021).
Fishbein, M. C. et al. Cardiac pathology of transvenous pacemakers in dogs. Am. Heart J. 93, 73–81 (1977).
Bowman, T. A. & Hughes, H. C. Swine as an in vivo model for electrophysiologic evaluation of cardiac pacing parameters. Pacing Clin. Electrophysiol. 7, 187–194 (1984).
Wijffels, M. C. E. F., Kirchhof, C. J. H. J., Dorland, R. & Allessie, M. A. Atrial fibrillation begets atrial fibrillation. Circ. J. 92, 1954–1968 (1995).
Vuolteenaho, O., Ala-Kopsala, M. & Ruskoaho, H. BNP as a biomarker in heart disease. Adv. Clin. Chem. 40, 1–36 (2005).
Watson, L. E., Sheth, M., Denyer, R. F. & Dostal, D. E. Baseline echocardiographic values for adult male rats. J. Am. Soc. Echocardiogr. 17, 161–167 (2004).
Acknowledgements
We thank the staff of the Office of Animal Research at The George Washington University for their dedication in caring for our animals. We acknowledge funding by the Leducq Foundation (project RHYTHM) and NIH grant R01 HL141470 (I.R.E. and J.A.R.). R.T.Y. acknowledges support from the American Heart Association Predoctoral Fellowship (19PRE34380781). Y.S.C. acknowledges support from the NIH grant K99 HL155844. A.N.M. acknowledges support from the Henry Luce Foundation for the Luce Undergraduate Research Fellowship. E.A.W. acknowledges support from grant number 2020-225578 from the Chan Zuckerberg Initiative DAF, an advised fund of Silicon Valley Community Foundation.
Author information
Authors and Affiliations
Contributions
R.T.Y., S.W.C., K.B.L. and I.R.E. led the development of the concepts, designed the experiments and interpreted results. S.W.C., K.B.L., M.A.N., T.J.H., J.B.L. and A.M.-B. performed the surgeries. Y.S.C., J.K., J.A., P.G. and J.A.R. developed fabrication methods and devices. Q.Y. created the bioadhesive and adhesion strategy. R.T.Y. and G.K. completed preoperative and postoperative care of animals. E.A.W., A.B. and C.R.H. performed magnetic resonance imaging scans and image segmentation. R.T.Y., A.N.M., H.S.K. and B.A.R. completed histology and echocardiography. R.T.Y. and H.S.K. quantified behavioral monitoring of rats. R.T.Y., H.S.K., B.A.R., A.K. and T.E. devised and performed ELISA biomarker analysis. R.T.Y., S.W.C. and K.B.L. weighed the animals. R.T.Y. completed all ECG recordings and dissections. R.T.Y., S.W.C., K.B.L., M.A.N., T.J.H., G.D.T. and I.R.E. wrote the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
J.A.R. is a cofounder of NeuroLux Inc.
Peer review
Peer review information
Nature Protocols thanks Daniël Pijnappels and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Gutruf, P. et al. Nat. Commun. 10, 5742 (2019): https://doi.org/10.1038/s41467-019-13637-w
Choi, Y. S. et al. Nat. Biotechnol. 39, 1228–1238 (2021): https://doi.org/10.1038/s41587-021-00948-x
Yang, Q. et al. Nat. Mater. 20, 1559–1570 (2021): https://doi.org/10.1038/s41563-021-01051-x
Extended data
Extended Data Fig. 1 Assessment of surgery time.
a, The surgery workflow timeline is as follows: induction, intubation, incision, closure and extubation. b, For an experienced surgeon performing this procedure, the total time for surgery from induction to extubation is ~43 min, including initiation of anesthesia, intubation, incision, affixation of pacemaker, closure and extubation. Average times for an experienced surgeon to complete each section of the workflow timeline are provided. For operators just beginning to learn this technique, intubation may take several additional attempts. Additional time is required pre- and postoperatively to fulfill responsibilities such as preparation of equipment, retrieval and return of animals to the housing facility, administration of follow-up analgesic doses and recovery observation.
Extended Data Fig. 2 Open thoracic implantation technique for biventricular pacemaker implantation.
a, Electrodes of biventricular pacemaker positioned onto ventricle of Langendorff-perfused mouse heart. Scale bar, 5 mm. b, Electrodes of the biventricular pacemaker sutured onto left and right ventricles during implantation surgery. Scale bar, 5 mm. c, Anterior and d, cross-sectional CT visualization of the biventricular pacemaker placed within a rat’s anatomy.
Extended Data Fig. 3 Implantation of miniature battery-free wireless pacemakers using hydrogel adhesive.
a, This technique allows for pacemakers to be attached with adhesives. Following opening of the chest, the pacemaker is affixed to the epicardial surface of the ventricles with a soft injectable bioadhesive. UV light is used to illuminate the bioadhesive to cure and secure the electrode pad in place. Scale bar, 5 mm. b, ECG traces of animals implanted with pacemakers were recorded daily postoperation. Pacemakers were able to capture and drive the heart rhythm for up to 8 d postsurgery.
Supplementary information
Supplementary Information
Supplementary Figs. 1–6.
Supplementary Data 1
Raw data of surgery mortality rate relating to operator experience
Supplementary Data 2
Raw data on functional lifetime of pacemakers in all rat subjects
Supplementary Data 3
Raw data of systemic assessment of animal behavior, appearance, and reactivity to handling
Supplementary Data 4
Raw data of ELISA assessment of BNP 45 for rats with pacemakers attached to the atrium
Supplementary Data 5
Raw data of ELISA assessment of CK-MB for rats with pacemakers attached to the ventricles
Supplementary Data 6
Raw data of echocardiography parameters (fractional shortening, cardiac output, diastolic volume, diastolic diameter, systolic volume, and systolic diameter)
Source data
Source Data Fig. 6
Raw data.
Source Data Extended Data Fig. 1
Raw data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yin, R.T., Chen, S.W., Benjamin Lee, K. et al. Open thoracic surgical implantation of cardiac pacemakers in rats. Nat Protoc 18, 374–395 (2023). https://doi.org/10.1038/s41596-022-00770-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-022-00770-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.