Abstract
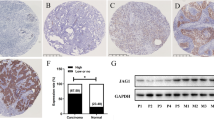
Notch signaling is often and aberrantly activated by hypoxia during tumor progression; however, the exact pathological role of hypoxia-induced Notch signaling in tumor metastasis is as yet poorly understood. In this study, we aimed to define the mechanism of Notch-ligand activation by hypoxia in both primary tumor and bone stromal cells in the metastatic niche and to clarify their roles in tumor progression. We have analyzed the expression profiles of various Notch ligands in 779 breast cancer patients in GEO database and found that the expression of Jagged2 among all five ligands is most significantly correlated with the overall- and metastasis-free survival of breast cancer patients. The results of our immunohistochemical (IHC) analysis for Jagged2 in 61 clinical samples also revealed that both Jagged2 and Notch signaling were strongly upregulated at the hypoxic invasive front. Activation of Jagged2 by hypoxia in tumor cells induced EMT and also promoted cell survival in vitro. Notably, a γ-secretase inhibitor significantly blocked Notch-mediated invasion and survival under hypoxia by promoting expression of E-cadherin and inhibiting Akt phosphorylation. Importantly, Jagged2 was also found to be upregulated in bone marrow stroma under hypoxia and promoted the growth of cancer stem-like cells by activating their Notch signaling. Therefore, hypoxia-induced Jagged2 activation in both tumor invasive front and normal bone stroma has a critical role in tumor progression and metastasis, and Jagged2 is considered to be a valuable prognostic marker and may serve as a novel therapeutic target for metastatic breast cancer.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Artavanis-Tsakonas S, Rand MD, Lake RJ . (1999). Notch signaling: cell fate control and signal integration in development. Science 284: 770–776.
Bandyopadhyay S, Wang Y, Zhan R, Pai SK, Watabe M, Iiizumi M et al. (2006). The tumor metastasis suppressor gene Drg-1 down-regulates the expression of activating transcription factor 3 in prostate cancer. Cancer Res 66: 11983–11990.
Bedogni B, Warneke JA, Nickoloff BJ, Giaccia AJ, Powell MB . (2008). Notch1 is an effector of Akt and hypoxia in melanoma development. J Clin Invest 118: 3660–3670.
Bertout JA, Patel SA, Fryer BH, Durham AC, Covello KL, Olive KP et al. (2009). Heterozygosity for hypoxia inducible factor 1alpha decreases the incidence of thymic lymphomas in a p53 mutant mouse model. Cancer Res 69: 3213–3220.
Cejudo-Martin P, Johnson RS . (2005). A new notch in the HIF belt: how hypoxia impacts differentiation. Dev Cell 9: 575–576.
Chen Y, De Marco MA, Graziani I, Gazdar AF, Strack PR, Miele L et al. (2007). Oxygen concentration determines the biological effects of NOTCH-1 signaling in adenocarcinoma of the lung. Cancer Res 67: 7954–7959.
Choi K, Ahn YH, Gibbons DL, Tran HT, Creighton CJ, Girard L et al. (2009). Distinct biological roles for the notch ligands Jagged-1 and Jagged-2. J Biol Chem 284: 17766–17774.
D'Souza B, Meloty-Kapella L, Weinmaster G . (2010). Canonical and non-canonical Notch ligands. Curr Top Dev Biol 92: 73–129.
Das B, Tsuchida R, Malkin D, Koren G, Baruchel S, Yeger H . (2008). Hypoxia enhances tumor stemness by increasing the invasive and tumorigenic side population fraction. Stem Cells 26: 1818–1830.
Dickson BC, Mulligan AM, Zhang H, Lockwood G, O'Malley FP, Egan SE et al. (2007). High-level JAG1 mRNA and protein predict poor outcome in breast cancer. Mod Pathol 20: 685–693.
Eliasz S, Liang S, Chen Y, De Marco MA, Machek O, Skucha S et al. (2010). Notch-1 stimulates survival of lung adenocarcinoma cells during hypoxia by activating the IGF-1R pathway. Oncogene 29: 2488–2498.
Ellisen LW, Bird J, West DC, Soreng AL, Reynolds TC, Smith SD et al. (1991). TAN-1, the human homolog of the Drosophila notch gene, is broken by chromosomal translocations in T lymphoblastic neoplasms. Cell 66: 649–661.
Felli MP, Maroder M, Mitsiadis TA, Campese AF, Bellavia D, Vacca A et al. (1999). Expression pattern of notch1, 2 and 3 and Jagged1 and 2 in lymphoid and stromal thymus components: distinct ligand-receptor interactions in intrathymic T cell development. Int Immunol 11: 1017–1025.
Furuta E, Pai SK, Zhan R, Bandyopadhyay S, Watabe M, Mo YY et al. (2008). Fatty acid synthase gene is up-regulated by hypoxia via activation of Akt and sterol regulatory element binding protein-1. Cancer Res 68: 1003–1011.
Gustafsson MV, Zheng X, Pereira T, Gradin K, Jin S, Lundkvist J et al. (2005). Hypoxia requires notch signaling to maintain the undifferentiated cell state. Dev Cell 9: 617–628.
Haase VH . (2009). Oxygen regulates epithelial-to-mesenchymal transition: insights into molecular mechanisms and relevance to disease. Kidney Int 76: 492–499.
Horree N, van Diest PJ, Sie-Go DM, Heintz AP . (2007). The invasive front in endometrial carcinoma: higher proliferation and associated derailment of cell cycle regulators. Hum Pathol 38: 1232–1238.
Kiaris H, Politi K, Grimm LM, Szabolcs M, Fisher P, Efstratiadis A et al. (2004). Modulation of notch signaling elicits signature tumors and inhibits hras1-induced oncogenesis in the mouse mammary epithelium. Am J Pathol 165: 695–705.
Klinakis A, Szabolcs M, Politi K, Kiaris H, Artavanis-Tsakonas S, Efstratiadis A . (2006). Myc is a Notch1 transcriptional target and a requisite for Notch1-induced mammary tumorigenesis in mice. Proc Natl Acad Sci USA 103: 9262–9267.
Koch U, Radtke F . (2007). Notch and cancer: a double-edged sword. Cell Mol Life Sci 64: 2746–2762.
Kominsky SL, Davidson NE . (2006). A ‘bone’ fide predictor of metastasis? Predicting breast cancer metastasis to bone. J Clin Oncol 24: 2227–2229.
Lambin P, Theys J, Landuyt W, Rijken P, van der Kogel A, van der Schueren E et al. (1998). Colonisation of Clostridium in the body is restricted to hypoxic and necrotic areas of tumours. Anaerobe 4: 183–188.
Lee SH, Jeong EG, Yoo NJ, Lee SH . (2007). Mutational analysis of NOTCH1, 2, 3 and 4 genes in common solid cancers and acute leukemias. Apmis 115: 1357–1363.
Leong KG, Karsan A . (2006). Recent insights into the role of Notch signaling in tumorigenesis. Blood 107: 2223–2233.
Main H, Lee KL, Yang H, Haapa-Paananen S, Edgren H, Jin S et al. (2010). Interactions between Notch- and hypoxia-induced transcriptomes in embryonic stem cells. Exp Cell Res 316: 1610–1624.
Maynard MA, Ohh M . (2007). The role of hypoxia-inducible factors in cancer. Cell Mol Life Sci 64: 2170–2180.
Mullendore ME, Koorstra JB, Li YM, Offerhaus GJ, Fan X, Henderson CM et al. (2009). Ligand-dependent Notch signaling is involved in tumor initiation and tumor maintenance in pancreatic cancer. Clin Cancer Res 15: 2291–2301.
Nam DH, Jeon HM, Kim S, Kim MH, Lee YJ, Lee MS et al. (2008). Activation of notch signaling in a xenograft model of brain metastasis. Clin Cancer Res 14: 4059–4066.
Parmar K, Mauch P, Vergilio JA, Sackstein R, Down JD . (2007). Distribution of hematopoietic stem cells in the bone marrow according to regional hypoxia. Proc Natl Acad Sci USA 104: 5431–5436.
Pouyssegur J, Dayan F, Mazure NM . (2006). Hypoxia signalling in cancer and approaches to enforce tumour regression. Nature 441: 437–443.
Reedijk M, Odorcic S, Chang L, Zhang H, Miller N, McCready DR et al. (2005). High-level coexpression of JAG1 and NOTCH1 is observed in human breast cancer and is associated with poor overall survival. Cancer Res 65: 8530–8537.
Roodman GD . (2004). Mechanisms of bone metastasis. N Engl J Med 350: 1655–1664.
Sahlgren C, Gustafsson MV, Jin S, Poellinger L, Lendahl U . (2008). Notch signaling mediates hypoxia-induced tumor cell migration and invasion. Proc Natl Acad Sci USA 105: 6392–6397.
Schindl M, Schoppmann SF, Samonigg H, Hausmaninger H, Kwasny W, Gnant M et al. (2002). Overexpression of hypoxia-inducible factor 1alpha is associated with an unfavorable prognosis in lymph node-positive breast cancer. Clin Cancer Res 8: 1831–1837.
Stylianou S, Clarke RB, Brennan K . (2006). Aberrant activation of notch signaling in human breast cancer. Cancer Res 66: 1517–1525.
Tomes L, Emberley E, Niu Y, Troup S, Pastorek J, Strange K et al. (2003). Necrosis and hypoxia in invasive breast carcinoma. Breast Cancer Res Treat 81: 61–69.
Wang Z, Li Y, Ahmad A, Azmi AS, Banerjee S, Kong D et al. (2010). Targeting Notch signaling pathway to overcome drug resistance for cancer therapy. Biochim Biophys Acta 1806: 258–267.
Yin T, Li L . (2006). The stem cell niches in bone. J Clin Invest 116: 1195–1201.
Yu X, Zou J, Ye Z, Hammond H, Chen G, Tokunaga A et al. (2008). Notch signaling activation in human embryonic stem cells is required for embryonic, but not trophoblastic, lineage commitment. Cell Stem Cell 2: 461–471.
Zhao N, Guo Y, Zhang M, Lin L, Zheng Z . (2010). Akt-mTOR signaling is involved in Notch-1-mediated glioma cell survival and proliferation. Oncol Rep 23: 1443–1447.
Zhong H, De Marzo AM, Laughner E, Lim M, Hilton DA, Zagzag D et al. (1999). Overexpression of hypoxia-inducible factor 1alpha in common human cancers and their metastases. Cancer Res 59: 5830–5835.
Acknowledgements
This work was supported by NIH (R01CA124650, R01CA129000 to KW), the US Department of Defense (BC085424, BC085590 to KW) and Susan Komen Foundation (KG080477 to KW).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Xing, F., Okuda, H., Watabe, M. et al. Hypoxia-induced Jagged2 promotes breast cancer metastasis and self-renewal of cancer stem-like cells. Oncogene 30, 4075–4086 (2011). https://doi.org/10.1038/onc.2011.122
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.122
Keywords
This article is cited by
-
The Role of Breast Cancer Cells in Bone Metastasis: Suitable Seeds for Nourishing Soil
Current Osteoporosis Reports (2024)
-
Regulation and signaling pathways in cancer stem cells: implications for targeted therapy for cancer
Molecular Cancer (2023)
-
Ferroptosis and EMT: key targets for combating cancer progression and therapy resistance
Cellular and Molecular Life Sciences (2023)
-
Notch signaling pathway: a comprehensive prognostic and gene expression profile analysis in breast cancer
BMC Cancer (2022)
-
Hypoxia signaling in human health and diseases: implications and prospects for therapeutics
Signal Transduction and Targeted Therapy (2022)