Abstract
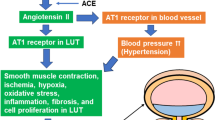
Lower urinary tract symptoms (LUTS)—constituting a spectrum disorder that encompasses weak stream, nocturia, and sensations of incomplete emptying and intermittent or hesitant urination—are indicative of lower urinary tract dysfunction (LUTD). LUTD is a progressive disease that can lead to bladder dysfunction if left untreated or treated ineffectively. Sequelae include urinary retention, recurrent UTI, bladder calculi, and, eventually, renal impairment. LUTD involving the prostate is associated with both ageing and inflammation. Tissue inflammation resulting from ageing, infection, or other inflammatory disease processes (for example, type 2 diabetes mellitus) is epidemiologically associated with the subsequent development of tissue fibrosis in multiple organ systems, including the prostate. Recent studies show that tissue fibrosis in the lower urinary tract is associated with LUTD, and suggest that fibrosis might be a previously unrecognized pathobiology that contributes to LUTD. Thus, antifibrotic therapeutic agents should be considered as a new approach to efficaciously treating men with LUTD, especially those who don't experience durable responses to 5α-reductase inhibitors or α-adrenergic receptor antagonists.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Laborde, E. E. & McVary, K. T. Medical management of lower urinary tract symptoms. Nat. Rev. Urol. 11, S19–S25 (2009).
Wei, J. T., Calhoun, E. & Jacobsen, S. J. Urologic diseases in America project: benign prostatic hyperplasia. J. Urol. 173, 1256–1261 (2005).
Kupelian, V. et al. Prevalence of lower urinary tract symptoms and effect on quality of life in a racially and ethnically diverse random sample: the Boston Area Community Health (BACH) Survey. Arch. Intern. Med. 166, 2381–2387 (2006).
Parsons, J. K., Bergstrom, J., Silberstein, J. & Barrett-Connor, E. Prevalence and characteristics of lower urinary tract symptoms in men aged > or = 80 years. Urology 72, 318–321 (2008).
Irwin, D. E., Kopp, Z. S., Agatep, B., Milsom, I. & Abrams, P. Worldwide prevalence estimates of lower urinary tract symptoms, overactive bladder, urinary incontinence and bladder outlet obstruction. BJU Int. 108, 1132–1138 (2011).
Malaeb, B. S., Yu, X., McBean, A. M. & Elliott, S. P. National trends in surgical therapy for benign prostatic hyperplasia in the United States (2000–2008). Urology 79, 1111–1116 (2012).
Yu, X., Elliott, S. P., Wilt, T. J. & McBean, A. M. Practice patterns in benign prostatic hyperplasia surgical therapy: the dramatic increase in minimally invasive technologies. J. Urol. 180, 241–245 (2008).
Strope, S. A., Yang, L., Nepple, K. G., Andriole, G. L. & Owens, P. L. Population based comparative effectiveness of transurethral resection of the prostate and laser therapy for benign prostatic hyperplasia. J. Urol. 187, 1341–1345 (2012).
Krambeck, A. E. et al. Effectiveness of medical and surgical therapies for lower urinary tract symptoms in the community setting. BJU Int. 110, 1332–1337 (2012).
McConnell, J. D. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N. Engl. J. Med. 349, 2387–2398 (2003).
Bautista, O. M. et al. Study design of the Medical Therapy of Prostatic Symptoms (MTOPS) trial. Control. Clin. Trials 24, 224–243 (2003).
Nickel, J. C., Downey, J., Young, I. & Boag, S. Asymptomatic inflammation and/or infection in benign prostatic hyperplasia. BJU Int. 84, 976–981 (1999).
Robert, G. et al. Inflammation in benign prostatic hyperplasia: A 282 patients' immunohistochemical analysis. Prostate 69, 1774–1780 (2009).
Delongchamps, N. B. et al. Evaluation of prostatitis in autopsied prostates--is chronic inflammation more associated with benign prostatic hyperplasia or cancer? J. Urol. 179, 1736–1740 (2008).
Sutcliffe, S., Giovannucci, E., De Marzo, A. M., Willett, W. C. & Platz, E. A. Sexually transmitted infections, prostatitis, ejaculation frequency, and the odds of lower urinary tract symptoms. Am. J. Epidemiol. 162, 898–906 (2005).
St Sauver, J. L. & Jacobsen, S. J. Inflammatory mechanisms associated with prostatic inflammation and lower urinary tract symptoms. Curr. Prostate Rep. 6, 67–73 (2008).
St Sauver, J. L. et al. Longitudinal association between prostatitis and development of benign prostatic hyperplasia. Urology 71, 475–479 (2008).
Krieger, J. N. et al. Epidemiology of prostatitis. Int. J. Antimicrob. Agents 31 (Suppl. 1), S85–S90 (2008).
Colodner, R., Eliasberg, T., Chazan, B. & Raz, R. Clinical significance of bacteriuria with low colony counts of Enterococcus species. Eur. J. Clin. Microbiol. Infect. Dis. 25, 238–241 (2006).
Begley, L., Monteleon, C., Shah, R. B., Macdonald, J. W. & Macoska, J. A. CXCL12 overexpression and secretion by aging fibroblasts enhance human prostate epithelial proliferation in vitro. Aging Cell 4, 291–298 (2005).
Begley, L. A., Kasina, S., MacDonald, J. & Macoska, J. A. The inflammatory microenvironment of the aging prostate facilitates cellular proliferation and hypertrophy. Cytokine 43, 194–199 (2008).
Begley, L. A. et al. CXCL5 promotes prostate cancer progression. Neoplasia 10, 244–254 (2008).
Begley, L. A., MacDonald, J. W., Day, M. L. & Macoska, J. A. CXCL12 activates a robust transcriptional response in human prostate epithelial cells. J. Biol. Chem. 282, 26767–26774 (2007).
Dimri, G. P. et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl Acad. Sci. USA 92, 9363–9367 (1995).
Nishimura, F. et al. Comparison of in vitro proliferative capacity of human periodontal ligament cells in juvenile and aged donors. Oral Dis. 3, 162–166 (1997).
Hjelmeland, L. M., Cristofolo, V. J., Funk, W., Rakoczy, E. & Katz, M. L. Senescence of the retinal pigment epithelium. Mol. Vis. 5, 33 (1999).
Kajstura, J. et al. Telomere shortening is an in vivo marker of myocyte replication and aging. Am. J. Pathol. 156, 813–819 (2000).
Chkhotua, A. et al. Replicative senescence in organ transplantation-mechanisms and significance. Transpl. Immunol. 9, 165–171 (2002).
Bavik, C. The gene expression program of prostate fibroblast senescence modulates neoplastic epithelial cell proliferation through paracrine mechanisms. Cancer Res. 66, 794–802 (2006).
Coppe, J. P. et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumour suppressor. PLoS Biol. 6, 2853–2868 (2008).
Penna, G. et al. Human benign prostatic hyperplasia stromal cells as inducers and targets of chronic immuno-mediated inflammation. J. Immunol. 182, 4056–4064 (2009).
Fujita, K. et al. Monocyte chemotactic protein-1 (MCP-1/CCL2) is associated with prostatic growth dysregulation and benign prostatic hyperplasia. Prostate 70, 473–481 (2010).
Schauer, I. G., Ressler, S. J., Tuxhorn, J. A., Dang, T. D. & Rowley, D. R. Elevated epithelial expression of interleukin-8 correlates with myofibroblast reactive stroma in benign prostatic hyperplasia. Urology 72, 205–213 (2008).
Tuxhorn, J. A. et al. Reactive stroma in human prostate cancer: induction of myofibroblast phenotype and extracellular matrix remodeling. Clin. Cancer Res. 8, 2912–2923 (2002).
Schauer, I. G., Ressler, S. J. & Rowley, D. R. Keratinocyte-derived chemokine induces prostate epithelial hyperplasia and reactive stroma in a novel transgenic mouse model. Prostate 69, 373–384 (2009).
Gharaee-Kermani, M. et al. CXC-type chemokines promote myofibroblast phenoconversion and prostatic fibrosis. PLoS ONE 7, e49278 (2012).
Sarma, A. V., Parsons, J. K., McVary, K. & Wei, J. T. Diabetes and benign prostatic hyperplasia/lower urinary tract symptoms—what do we know? J. Urol. 182, S32–S37 (2009).
Michel, M. C., Mehlburger, L., Schumacher, H., Bressel, H. U. & Goepel, M. Effect of diabetes on lower urinary tract symptoms in patients with benign prostatic hyperplasia. J. Urol. 163, 1725–1729 (2000).
Pradhan, A. D., Manson, J. E., Rifai, N., Buring, J. E. & Ridker, P. M. C-reactive protein, interleukin 6, and risk of developing type 2 diabetes mellitus. JAMA 286, 327–334 (2001).
Morelli, A. et al. Testosterone and farnesoid X receptor agonist INT-747 counteract high fat diet-induced bladder alterations in a rabbit model of metabolic syndrome. J. Steroid Biochem. Mol. Biol. 132, 80–92 (2012).
Azadzoi, K. M., Tarcan, T., Siroky, M. B. & Krane, R. J. Atherosclerosis-induced chronic ischemia causes bladder fibrosis and non-compliance in the rabbit. J. Urol. 161, 1626–1635 (1999).
Gharaee-Kermani, M. et al. Obesity-induced diabetes and lower urinary tract fibrosis promote urinary voiding dysfunction in a mouse model. Prostate 73, 1123–1133 (2013).
Donath, M. Y. et al. Islet inflammation in type 2 diabetes: from metabolic stress to therapy. Diabetes Care 31, S161–S164 (2008).
Detlefsen, S., Sipos, B., Feyerabend, B. & Klöppel, G. Pancreatic fibrosis associated with age and ductal papillary hyperplasia. Virchows Archiv. 447, 800–805 (2005).
Gharaee-Kermani, M., Hu, B., Phan, S. H. & Gyetko, M. R. Recent advances in molecular targets and treatment of idiopathic pulmonary fibrosis: focus on TGFβ signalling and the myofibroblast. Curr. Med. Chem. 16, 1400–1417 (2009).
Scotton, C. J. & Chambers, R. C. Molecular targets in pulmonary fibrosis: the myofibroblast in focus. Chest 132, 1311–1321 (2007).
Novo, E., Valfrè di Bonzo, L., Cannito, S., Colombatto, S. & Parola, M. Hepatic myofibroblasts: A heterogeneous population of multifunctional cells in liver fibrogenesis. Int. J. Biochem. Cell Biol. 41, 2089–2093 (2009).
Frith, J., Day, C. P., Henderson, E., Burt, A. D. & Newton, J. L. Non-alcoholic fatty liver disease in older people. Gerontology 55, 607–613 (2009).
Rieder, F. & Fiocchi, C. Intestinal fibrosis in IBD—a dynamic, multifactorial process. Nat. Rev. Gastroenterol. Hepatol. 6, 228–235 (2009).
Johnston, R. D. & Logan, R. F. A. What is the peak age for onset of IBD? Inflamm. Bowel Dis. 14 (Suppl. 2), S4–S5 (2008).
Goldacre, M. J. Demography of aging and the epidemiology of gastrointestinal disorders in the elderly. Best Pract. Res. Clin. Gastroenterol. 23, 793–804 (2009).
Pohlers, D. et al. TGF-β and fibrosis in different organs — molecular pathway imprints. Biochim. Biophys. Acta 1792, 746–756 (2009).
Hinz, B. Formation and Function of the Myofibroblast during Tissue Repair. J. Invest. Dermatol. 127, 526–537 (2007).
Wynn, T. A. Cellular and molecular mechanisms of fibrosis. J. Pathol. 214, 199–210 (2008).
Hinz, B. et al. The myofibroblast. Am. J. Pathol. 170, 1807–1816 (2007).
Ma, J. et al. Prostatic fibrosis is associated with lower urinary tract symptoms. J. Urol. 188, 1375–1381 (2012).
Rocco, B. et al. Current status of salvage robot-assisted laparoscopic prostatectomy for radiorecurrent prostate cancer. Curr. Urol. Rep. 13, 195–201 (2012).
Crawford, E. D. & Kavanagh, B. D. The role of alpha-blockers in the management of lower urinary tract symptoms in prostate cancer patients treated with radiation therapy. Am. J. Clin. Oncol. 29, 517–523 (2006).
Rosenzweig-Bublil, N. & Abramovici, A. Stromal fibrosis reaction in rat prostates induced by alpha 1 adrenergic stimulation. J. Androl. 27, 276–284 (2006).
Delella, F. K., Lacorte, L. M., Almeida, F. L., Pai, M. D. & Felisbino, S. L. Fibrosis-related gene expression in the prostate is modulated by doxazosin treatment. Life Sci. 91, 1281–1287 (2012).
Guinan, P. et al. The effect of androgen deprivation on malignant and benign prostate tissue. Med. Oncol. 14, 145–152 (1997).
Roznovanu, S. L., Radulescu, D., Novac, C. & Stolnicu, S. The morphologic changes induced by hormone and radiation therapy on prostate carcinoma. Rev. Med. Chir. Soc. Med. Nat. Iasi. 109, 337–342 (2005).
Welsh, M. et al. Smooth muscle cell-specific knockout of androgen receptor: a new model for prostatic disease. Endocrinology 152, 3541–3551 (2011).
Wynn, T. A. & Ramalingam, T. R. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat. Med. 18, 1028–1040 (2012).
Author information
Authors and Affiliations
Contributions
Both authors contributed towards researching, writing, editing, discussing, and reviewing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Rodriguez-Nieves, J., Macoska, J. Prostatic fibrosis, lower urinary tract symptoms, and BPH. Nat Rev Urol 10, 546–550 (2013). https://doi.org/10.1038/nrurol.2013.149
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2013.149
This article is cited by
-
Mechanism of RhoA regulating benign prostatic hyperplasia: RhoA-ROCK-β-catenin signaling axis and static & dynamic dual roles
Molecular Medicine (2023)
-
Alterations in the balance of sex hormones may affect rat prostatic inflammation and fibrosis, and osteopontin might be involved in this process
International Urology and Nephrology (2023)
-
Estrogen and G protein-coupled estrogen receptor accelerate the progression of benign prostatic hyperplasia by inducing prostatic fibrosis
Cell Death & Disease (2022)
-
Smoothened inhibition leads to decreased cell proliferation and suppressed tissue fibrosis in the development of benign prostatic hyperplasia
Cell Death Discovery (2021)
-
Genome-wide association study identifies a role for the progesterone receptor in benign prostatic hyperplasia risk
Prostate Cancer and Prostatic Diseases (2021)