Abstract
In men, the lower urinary tract comprises the urinary bladder, urethra, and prostate, and its primary functions include urine storage and voiding. Hypertension is a condition that causes multi-organ damage and an age-dependent condition. Hypertension and the renin-angiotensin system activation are associated with the development of lower urinary tract dysfunction. Hypertensive animal models show bladder dysfunction, urethral dysfunction, and prostatic hyperplasia. In the renin-angiotensin system, angiotensin II and the angiotensin II type 1 receptor, which are expressed in the lower urinary tract, have been implicated in the pathogenesis of lower urinary tract dysfunction. Moreover, among the several antihypertensives, renin-angiotensin system inhibitors have proven effective in human and animal models of lower urinary tract dysfunction. This review aimed to elucidate the hitherto known mechanisms underlying the development of lower urinary tract dysfunction in relation to hypertension and the angiotensin II/angiotensin II type 1 receptor axis and the effect of renin-angiotensin system inhibitors on lower urinary tract dysfunction.

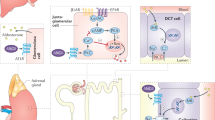
Possible mechanisms through which hypertension or activation of Ang II/AT1 receptor axis causes LUTD such as bladder dysfunction, urethral dysfunction, and prostatic hyperplasia. LUT: lower urinary tract, LUTD: lower urinary tract dysfunction, AT1: angiotensin II type 1, ACE: angiotensin-converting enzyme.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
de Groat WC, Griffiths D, Yoshimura N. Neural control of the lower urinary tract. Compr Physiol. 2015;5:327–96.
Umemura S, Arima H, Arima S, Asayama K, Dohi Y, Hirooka Y, et al. The Japanese society of hypertension guidelines for the management of hypertension (JSH 2019). Hypertens Res. 2019;42:1235–481.
(NCD-RisC) NRFC. Worldwide trends in hypertension prevalence and progress in treatment and control from 1990 to 2019: a pooled analysis of 1201 population-representative studies with 104 million participants. Lancet. 2021;398:957–80.
Sugaya K, Kadekawa K, Ikehara A, Nakayama T, Gakiya M, Nashiro F, et al. Influence of hypertension on lower urinary tract symptoms in benign prostatic hyperplasia. Int J Urol. 2003;10:569–74.
Ponholzer A, Temml C, Wehrberger C, Marszalek M, Madersbacher S. The association between vascular risk factors and lower urinary tract symptoms in both sexes. Eur Urol. 2006;50:581–6.
Ito H, Yoshiyasu T, Yamaguchi O, Yokoyama O. Male lower urinary tract symptoms: hypertension as a risk factor for storage symptoms, but not voiding symptoms. Low Urin Trac Symptoms. 2012;4:68–72.
Ohishi M, Kubozono T, Higuchi K, Akasaki Y. Hypertension, cardiovascular disease, and nocturia: a systematic review of the pathophysiological mechanisms. Hypertens Res. 2021;44:733–9.
Paz Ocaranza M, Riquelme JA, García L, Jalil JE, Chiong M, Santos RAS, et al. Counter-regulatory renin-angiotensin system in cardiovascular disease. Nat Rev Cardiol. 2020;17:116–29.
Ghatage T, Goyal SG, Dhar A, Bhat A. Novel therapeutics for the treatment of hypertension and its associated complications: peptide- and nonpeptide-based strategies. Hypertens Res. 2021;44:740–55.
Comiter C. Local renin-angiotensin systems in the genitourinary tract. Naunyn Schmiedebergs Arch Pharm. 2012;385:13–26.
Shimizu S. Therapeutic targets in the brain for overactive bladder: a focus on angiotensin II type 1 receptor. J Pharm Sci. 2023;153:69–72.
Takahashi K, Tanaka T, Yoshizawa Y, Fujisaki-Sueda-Sakai M, Son BK, Iijima K. Lower urinary tract symptoms and functional ability in older adults: a community-based cross-sectional study. BMJ Open. 2022;12:e054530.
Irwin DE, Kopp ZS, Agatep B, Milsom I, Abrams P. Worldwide prevalence estimates of lower urinary tract symptoms, overactive bladder, urinary incontinence and bladder outlet obstruction. BJU Int. 2011;108:1132–8.
Abrams P, Cardozo L, Fall M, Griffiths D, Rosier P, Ulmsten U, et al. The standardisation of terminology of lower urinary tract function: report from the Standardisation Sub-committee of the International Continence Society. Neurourol Urodyn. 2002;21:167–78.
Peyronnet B, Mironska E, Chapple C, Cardozo L, Oelke M, Dmochowski R, et al. A comprehensive review of overactive bladder pathophysiology: on the way to tailored treatment. Eur Urol. 2019;75:988–1000.
Madersbacher S, Sampson N, Culig Z. Pathophysiology of benign prostatic hyperplasia and benign prostatic enlargement: a mini-review. Gerontology. 2019;65:458–64.
Arnold MJ, Gaillardetz A, Ohiokpehai J. Benign prostatic hyperplasia: rapid evidence review. Am Fam Physician. 2023;107:613–22.
Collaborators GBPH. The global, regional, and national burden of benign prostatic hyperplasia in 204 countries and territories from 2000 to 2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet Healthy Longev. 2022;3:e754–e76.
Wu YH, Chueh KS, Chuang SM, Long CY, Lu JH, Juan YS. Bladder hyperactivity induced by oxidative stress and bladder ischemia: a review of treatment strategies with antioxidants. Int J Mol Sci. 2021;22:6014.
Tarcan T, Choi HP, Azadzoi KM. Molecular regulation of concomitant lower urinary tract symptoms and erectile dysfunction in pelvic ischemia. Int J Mol Sci. 2022;23:15988.
Shimizu S, Tsounapi P, Shimizu T, Honda M, Inoue K, Dimitriadis F, et al. Lower urinary tract symptoms, benign prostatic hyperplasia/benign prostatic enlargement and erectile dysfunction: are these conditions related to vascular dysfunction? Int J Urol. 2014;21:856–64.
Saito M, Tsounapi P, Oikawa R, Shimizu S, Honda M, Sejima T, et al. Prostatic ischemia induces ventral prostatic hyperplasia in the SHR; possible mechanism of development of BPH. Sci Rep. 2014;4:3822.
Akbar A, Liu K, Michos ED, Bancks MP, Brubaker L, Markossian T, et al. Association of overactive bladder with hypertension and blood pressure control: the multi-ethnic study of atherosclerosis (MESA). Am J Hypertens. 2022;35:22–30.
Yokoyama O, Nishizawa O, Homma Y, Takeda M, Gotoh M, Kakizaki H, et al. Nocturnal polyuria and hypertension in patients with lifestyle related diseases and overactive bladder. J Urol. 2017;197:423–31.
Sugaya K, Nishijima S, Oda M, Owan T, Miyazato M, Ogawa Y. Biochemical and body composition analysis of nocturia in the elderly. Neurourol Urodyn. 2008;27:205–11.
Michel MC, Heemann U, Schumacher H, Mehlburger L, Goepel M. Association of hypertension with symptoms of benign prostatic hyperplasia. J Urol. 2004;172:1390–3.
Hwang EC, Kim SO, Nam DH, Yu HS, Hwang I, Jung SI, et al. Men with hypertension are more likely to have severe lower urinary tract symptoms and large prostate volume. Low Urin Trac Symptoms. 2015;7:32–6.
White WB, Moon T. Treatment of benign prostatic hyperplasia in hypertensive men. J Clin Hypertens. 2005;7:212–7.
Edwards KM, Wilson KL, Sadja J, Ziegler MG, Mills PJ. Effects on blood pressure and autonomic nervous system function of a 12-week exercise or exercise plus DASH-diet intervention in individuals with elevated blood pressure. Acta Physiol. 2011;203:343–50.
Hubeaux K, Deffieux X, Ismael SS, Raibaut P, Amarenco G. Autonomic nervous system activity during bladder filling assessed by heart rate variability analysis in women with idiopathic overactive bladder syndrome or stress urinary incontinence. J Urol. 2007;178:2483–7.
McVary KT, Rademaker A, Lloyd GL, Gann P. Autonomic nervous system overactivity in men with lower urinary tract symptoms secondary to benign prostatic hyperplasia. J Urol. 2005;174:1327–433.
McVary KT, Razzaq A, Lee C, Venegas MF, Rademaker A, McKenna KE. Growth of the rat prostate gland is facilitated by the autonomic nervous system. Biol Reprod. 1994;51:99–107.
Fahed G, Aoun L, Bou Zerdan M, Allam S, Bou Zerdan M, Bouferraa Y, et al. Metabolic syndrome: updates on pathophysiology and management in 2021. Int J Mol Sci. 2022;23:786.
Pinggera GM, Mitterberger M, Steiner E, Pallwein L, Frauscher F, Aigner F, et al. Association of lower urinary tract symptoms and chronic ischaemia of the lower urinary tract in elderly women and men: assessment using colour Doppler ultrasonography. BJU Int. 2008;102:470–4.
Berger AP, Bartsch G, Deibl M, Alber H, Pachinger O, Fritsche G, et al. Atherosclerosis as a risk factor for benign prostatic hyperplasia. BJU Int. 2006;98:1038–42.
Spitsbergen JM, Clemow DB, McCarty R, Steers WD, Tuttle JB. Neurally mediated hyperactive voiding in spontaneously hypertensive rats. Brain Res. 1998;790:151–9.
Steers WD, Clemow DB, Persson K, Sherer TB, Andersson KE, Tuttle JB. The spontaneously hypertensive rat: insight into the pathogenesis of irritative symptoms in benign prostatic hyperplasia and young anxious males. Exp Physiol. 1999;84:137–47.
Yono M, Yamamoto Y, Yoshida M, Ueda S, Latifpour J. Effects of doxazosin on blood flow and mRNA expression of nitric oxide synthase in the spontaneously hypertensive rat genitourinary tract. Life Sci. 2007;81:218–22.
Saito M, Ohmasa F, Tsounapi P, Inoue S, Dimitriadis F, Kinoshita Y, et al. Nicorandil ameliorates hypertension-related bladder dysfunction in the rat. Neurourol Urodyn. 2012;31:695–701.
Shen S, Xia CM, Qiao LY. The urinary bladder of spontaneously hypertensive rat demonstrates bladder hypertrophy, inflammation, and fibrosis but not hyperplasia. Life Sci. 2015;121:22–7.
Shen JD, Chen SJ, Chen HY, Chiu KY, Chen YH, Chen WC. Review of animal models to study urinary bladder function. Biology. 2021;10:1316.
Shimizu S, Saito M, Oiwa H, Ohmasa F, Tsounapi P, Oikawa R, et al. Olmesartan ameliorates urinary dysfunction in the spontaneously hypertensive rat via recovering bladder blood flow and decreasing oxidative stress. Neurourol Urodyn. 2014;33:350–7.
Kim KH, Jin LH, Choo GY, Lee HJ, Choi BH, Kwak J, et al. Nonselective blocking of the sympathetic nervous system decreases detrusor overactivity in spontaneously hypertensive rats. Int J Mol Sci. 2012;13:5048–59.
Golomb E, Rosenzweig N, Eilam R, Abramovici A. Spontaneous hyperplasia of the ventral lobe of the prostate in aging genetically hypertensive rats. J Androl. 2000;21:58–64.
Zhang X, Na Y, Guo Y. Biologic feature of prostatic hyperplasia developed in spontaneously hypertensive rats. Urology. 2004;63:983–8.
Matityahou A, Rosenzweig N, Golomb E. Rapid proliferation of prostatic epithelial cells in spontaneously hypertensive rats: a model of spontaneous hypertension and prostate hyperplasia. J Androl. 2003;24:263–9.
Yamashita M, Zhang X, Shiraishi T, Uetsuki H, Kakehi Y. Determination of percent area density of epithelial and stromal components in development of prostatic hyperplasia in spontaneously hypertensive rats. Urology. 2003;61:484–9.
Shimizu S, Shimizu T, Tsounapi P, Higashi Y, Martin DT, Nakamura K, et al. Effect of silodosin, an alpha1A-adrenoceptor antagonist, on ventral prostatic hyperplasia in the spontaneously hypertensive rat. PLoS One. 2015;10:e0133798.
Morelli A, Sarchielli E, Comeglio P, Filippi S, Mancina R, Gacci M, et al. Phosphodiesterase type 5 expression in human and rat lower urinary tract tissues and the effect of tadalafil on prostate gland oxygenation in spontaneously hypertensive rats. J Sex Med. 2011;8:2746–60.
Shimizu S, Nagao Y, Kataoka T, Kamada S, Shimizu T, Higashi Y, et al. Protective effects of tadalafil on prostatic hyperplasia in spontaneously hypertensive rats. Eur J Pharm. 2020;882:173313.
Shimizu S, Nagao Y, Shimizu T, Higashi Y, Karashima T, Saito M. Therapeutic effects of losartan on prostatic hyperplasia in spontaneously hypertensive rats. Life Sci. 2021;266:118924.
Nagao Y, Shimizu S, Kurabayashi A, Shimizu T, Tsuda M, Higashi Y, et al. Effects of silodosin and tadalafil on bladder dysfunction in spontaneously hypertensive rats: possible role of bladder blood flow. Int J Urol. 2020;27:258–65.
Kurokawa T, Zha X, Ito H, Aoki Y, Akino H, Kobayashi M, et al. Underlying mechanisms of urine storage dysfunction in rats with salt-loading hypertension. Life Sci. 2015;141:8–12.
Velasquez Flores M, Mossa AH, Cammisotto P, Campeau L. Bladder overdistension with polyuria in a hypertensive rat model. Neurourol Urodyn. 2018;37:1904–12.
Ramos-Filho AC, Mónica FZ, Franco-Penteado CF, Rojas-Moscoso JA, Báu FR, Schenka AA, et al. Characterization of the urinary bladder dysfunction in renovascular hypertensive rats. Neurourol Urodyn. 2011;30:1392–402.
Ramos-Filho AC, Moscoso JA, Calmasini F, de Almeida Faria J, Anhê GF, Mónica FZ, et al. Blockade of renin-angiotensin system prevents micturition dysfunction in renovascular hypertensive rats. Eur J Pharm. 2014;738:285–92.
Mónica FZ, Bricola AA, Báu FR, Freitas LL, Teixeira SA, Muscará MN, et al. Long-term nitric oxide deficiency causes muscarinic supersensitivity and reduces beta(3)-adrenoceptor-mediated relaxation, causing rat detrusor overactivity. Br J Pharm. 2008;153:1659–68.
Noguchi K, Sugaya K, Nishijima S, Sakanashi M, Kadekawa K, Ashitomi K, et al. Evaluation of a rat model of functional urinary bladder outlet obstruction produced by chronic inhibition of nitric oxide synthase. Life Sci. 2019;234:116772.
Lee WC, Chien CT, Yu HJ, Lee SW. Bladder dysfunction in rats with metabolic syndrome induced by long-term fructose feeding. J Urol. 2008;179:2470–6.
Chung SD, Chien CT, Yu HJ. Alterations in peripheral purinergic and muscarinic signaling of rat bladder after long-term fructose-induced metabolic syndrome. Eur J Nutr. 2013;52:347–59.
Nakazawa R, Tanaka M, Takahashi T, Kobayashi S, Iwamoto T. Effects of castration and testosterone administration on angiotensin II receptor mRNA expression and apoptosis-related proteins in rat urinary bladder. Endocr J. 2007;54:211–9.
Ramos-Filho AC, Faria JA, Calmasini FB, Teixeira SA, Mónica FZ, Muscará MN, et al. The renin-angiotensin system plays a major role in voiding dysfunction of ovariectomized rats. Life Sci. 2013;93:820–9.
Weaver-Osterholtz D, Reams G, De Vergel CF, Bauer JH. The bladder angiotensin system in female rats: response to infusions of angiotensin I and the angiotensin converting enzyme inhibitor enalaprilat. J Urol. 2001;165:1735–8.
Yamada S, Takeuchi C, Oyunzul L, Ito Y. Bladder angiotensin-II receptors: characterization and alteration in bladder outlet obstruction. Eur Urol. 2009;55:482–9.
Cho ST, Park EY, Kim JC. Effect of angiotensin II receptor antagonist telmisartan on detrusor overactivity in rats with bladder outlet obstruction. Urology. 2012;80:1163.e1–7.
Tobu S, Noguchi M, Hatada T, Mori K, Matsuo M, Sakai H. Changes in angiotensin II type 1 receptor expression in the rat bladder by bladder outlet obstruction. Urol Int. 2012;89:241–5.
Lim I, Mitsui R, Kameda M, Sellers DJ, Chess-Williams R, Hashitani H. Comparative effects of angiotensin II on the contractility of muscularis mucosae and detrusor in the pig urinary bladder. Neurourol Urodyn. 2021;40:102–11.
Shirotake S, Miyajima A, Kosaka T, Tanaka N, Maeda T, Kikuchi E, et al. Angiotensin II type 1 receptor expression and microvessel density in human bladder cancer. Urology. 2011;77:1009.e19–25.
Erspamer V, Ronzoni G, Falconieri Erspamer G. Effects of active peptides on the isolated muscle of the human urinary bladder. Investig Urol. 1981;18:302–4.
Andersson KE, Hedlund H, Stahl M. Contractions induced by angiotensin I, angiotensin II and bradykinin in isolated smooth muscle from the human detrusor. Acta Physiol Scand. 1992;145:253–9.
Lam DS, Dias LS, Moore KH, Burcher E. Angiotensin II in child urinary bladder: functional and autoradiographic studies. BJU Int. 2000;86:494–501.
Waldeck K, Lindberg BF, Persson K, Andersson KE. Characterization of angiotensin II formation in human isolated bladder by selective inhibitors of ACE and human chymase: a functional and biochemical study. Br J Pharm. 1997;121:1081–6.
Frara N, Giaddui D, Braverman AS, Jawawdeh K, Wu C, Ruggieri MR Sr., et al. Mechanisms involved in nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (Nox)-derived reactive oxygen species (ROS) modulation of muscle function in human and dog bladders. PLoS One. 2023;18:e0287212.
Tanabe N, Ueno A, Tsujimoto G. Angiotensin II receptors in the rat urinary bladder smooth muscle: type 1 subtype receptors mediate contractile responses. J Urol. 1993;150:1056–9.
Persson K, Pandita RK, Waldeck K, Andersson KE. Angiotensin II and bladder obstruction in the rat: influence on hypertrophic growth and contractility. Am J Physiol. 1996;271:R1186–92.
Dolber PC, Jin H, Nassar R, Coffman TM, Gurley SB, Fraser MO. The effects of Ins2(Akita) diabetes and chronic angiotensin II infusion on cystometric properties in mice. Neurourol Urodyn. 2015;34:72–8.
Cheng EY, Grammatopoulos T, Lee C, Sensibar J, Decker R, Kaplan WE, et al. Angiotensin II and basic fibroblast growth factor induce neonatal bladder stromal cell mitogenesis. J Urol. 1996;156:593–7.
Cheng EY, Decker RS, Lee C. Role of angiotensin II in bladder smooth muscle growth and function. Adv Exp Med Biol. 1999;462:183–91.
Siregar S, Parardya A, Sibarani J, Romdan T, Adi K, Hernowo BS, et al. AT(1) expression in human urethral stricture tissue. Res Rep Urol. 2017;9:181–6.
Nassis L, Frauman AG, Ohishi M, Zhuo J, Casley DJ, Johnston CI, et al. Localization of angiotensin-converting enzyme in the human prostate: pathological expression in benign prostatic hyperplasia. J Pathol. 2001;195:571–9.
Dinh DT, Frauman AG, Somers GR, Ohishi M, Zhou J, Casley DJ, et al. Evidence for activation of the renin-angiotensin system in the human prostate: increased angiotensin II and reduced AT(1) receptor expression in benign prostatic hyperplasia. J Pathol. 2002;196:213–9.
Dinh DT, Frauman AG, Sourial M, Casley DJ, Johnston CI, Fabiani ME. Identification, distribution, and expression of angiotensin II receptors in the normal human prostate and benign prostatic hyperplasia. Endocrinology. 2001;142:1349–56.
O’Mahony OA, Barker S, Puddefoot JR, Vinson GP. Synthesis and secretion of angiotensin II by the prostate gland in vitro. Endocrinology. 2005;146:392–8.
Fabiani ME, Hawkes DJ, Frauman AG, Tikellis C, Johnston CI, Wilkinson-Berka JL. Regulation of angiotensin II receptors in the prostate of the transgenic (mRen-2)27 rat: effect of angiotensin-converting enzyme inhibition. Int J Biochem Cell Biol. 2003;35:973–83.
Lin L, Li P, Liu X, Xie X, Liu L, Singh AK, et al. Systematic review and meta-analysis of candidate gene association studies of benign prostate hyperplasia. Syst Rev. 2022;11:60.
Lamy GB, Cafarchio EM, do Vale B, Antonio BB, Venancio DP, de Souza JS, et al. Unveiling the Angiotensin-(1-7) actions on the urinary bladder in female rats. Front Physiol. 2022;13:920636.
Domińska K, Kowalska K, Habrowska-Górczyńska DE, Urbanek KA, Ochędalski T, Piastowska-Ciesielska AW. The opposite effects of angiotensin 1-9 and angiotensin 3-7 in prostate epithelial cells. Biochem Biophys Res Commun. 2019;519:868–73.
Domińska K, Okła P, Kowalska K, Habrowska-Górczyńska DE, Urbanek KA, Ochędalski T, et al. Influence and mechanism of Angiotensin 1-7 on biological properties of normal prostate epithelial cells. Biochem Biophys Res Commun. 2018;502:152–9.
Dinh DT, Frauman AG, Casley DJ, Johnston CI, Fabiani ME. Angiotensin AT(4) receptors in the normal human prostate and benign prostatic hyperplasia. Mol Cell Endocrinol. 2001;184:187–92.
Andersson KE, Nomiya M, Yamaguchi O. Chronic pelvic ischemia: contribution to the pathogenesis of lower urinary tract symptoms (LUTS): a new target for pharmacological treatment? Low Urin Trac Symptoms. 2015;7:1–8.
Nomiya M, Andersson KE, Yamaguchi O. Chronic bladder ischemia and oxidative stress: new pharmacotherapeutic targets for lower urinary tract symptoms. Int J Urol. 2015;22:40–6.
Andersson KE. Oxidative stress and its relation to lower urinary tract symptoms. Int Neurourol J. 2022;26:261–7.
Masuda H, Kihara K, Saito K, Matsuoka Y, Yoshida S, Chancellor MB, et al. Reactive oxygen species mediate detrusor overactivity via sensitization of afferent pathway in the bladder of anaesthetized rats. BJU Int. 2008;101:775–80.
Phull H, Salkini M, Escobar C, Purves T, Comiter CV. The role of angiotensin II in stress urinary incontinence: a rat model. Neurourol Urodyn. 2007;26:81–8.
Ye G, Jin X. Action of autonomic drugs on the in vivo bladder base and proximal urethra in dog. Chin Med J. 1997;110:173–6.
Lin J, Freeman MR. Transactivation of ErbB1 and ErbB2 receptors by angiotensin II in normal human prostate stromal cells. Prostate. 2003;54:1–7.
Fabiani ME, Sourial M, Thomas WG, Johnston CI, Johnston CI, Frauman AG. Angiotensin II enhances noradrenaline release from sympathetic nerves of the rat prostate via a novel angiotensin receptor: implications for the pathophysiology of benign prostatic hyperplasia. J Endocrinol. 2001;171:97–108.
Strittmatter F, Walther S, Gratzke C, Göttinger J, Beckmann C, Roosen A, et al. Inhibition of adrenergic human prostate smooth muscle contraction by the inhibitors of c-Jun N-terminal kinase, SP600125 and BI-78D3. Br J Pharm. 2012;166:1926–35.
Jarari N, Rao N, Peela JR, Ellafi KA, Shakila S, Said AR, et al. A review on prescribing patterns of antihypertensive drugs. Clin Hypertens. 2015;22:7.
Ito H, Taga M, Tsuchiyama K, Akino H, Yokoyama O. IPSS is lower in hypertensive patients treated with angiotensin-II receptor blocker: posthoc analyses of a lower urinary tract symptoms population. Neurourol Urodyn. 2013;32:70–4.
Elliott CS, Comiter CV. The effect of angiotensin inhibition on urinary incontinence: data from the National Health and Nutrition Examination Survey (2001-2008). Neurourol Urodyn. 2014;33:1178–81.
Shirazi M, Khezri A, Samani SM, Monabbati A, Kojoori J, Hassanpour A. Effect of intraurethral captopril gel on the recurrence of urethral stricture after direct vision internal urethrotomy: phase II clinical trial. Int J Urol. 2007;14:203–8.
Hall SA, Chiu GR, Kaufman DW, Wittert GA, Link CL, McKinlay JB. Commonly used antihypertensives and lower urinary tract symptoms: results from the Boston Area Community Health (BACH) Survey. BJU Int. 2012;109:1676–84.
Salman M, Khan AH, Syed Sulaiman SA, Khan JH, Hussain K, Shehzadi N. Effect of calcium channel blockers on lower urinary tract symptoms: a systematic review. Biomed Res Int. 2017;2017:4269875.
Bulpitt CJ, Connor M, Schulte M, Fletcher AE. Bisoprolol and nifedipine retard in elderly hypertensive patients: effect on quality of life. J Hum Hypertens. 2000;14:205–12.
Washino S, Ugata Y, Saito K, Miyagawa T. Calcium channel blockers are associated with nocturia in men aged 40 years or older. J Clin Med. 2021;10:1603.
Aikawa K, Sakai T, Ishibashi K, Shiomi H, Sagawa K, Kumagai S, et al. Involvement of angiotensin II type 1 receptor on pathological remodeling and dysfunction in obstructed bladder. Int J Urol. 2012;19:457–64.
Comiter C, Phull HS. Angiotensin II type 1 (AT-1) receptor inhibition partially prevents the urodynamic and detrusor changes associated with bladder outlet obstruction: a mouse model. BJU Int. 2012;109:1841–6.
Juszczak K, Maciukiewicz P. The angiotensin II receptors type 1 blockage affects the urinary bladder activity in hyperosmolar-induced detrusor overactivity in rats: preliminary results. Adv Clin Exp Med. 2017;26:1047–51.
Mohamed BM, Ismail RS, Saleh IG, Abo-Salem OM, El-Sayed EM. Olmesartan ameliorates cyclophosphamide-induced hemorrhagic cystitis in rats via Nrf2/HO-1 signaling pathway. Tissue Cell. 2022;78:101877.
Siregar S, Farenia R, Sugandi S, Roesli RM. Effect of angiotensin II receptor blocker on TGF-β1, MMP-1, and collagen type I and type III concentration in New Zealand rabbit urethral stricture model. Res Rep. Urol. 2018;10:127–33.
Furukawa S, Yamamoto O, Tamura T, Masuda Y, Shudo C, Hayashi K, et al. Influence of efonidipine hydrochloride, calcium antagonist on the epithelium of prostates in spontaneously hypertensive rats. J Toxicol Sci. 1994;19:213–7.
Shen F, Dong LN, Zhang XY, Zhao XK, Zeng XF, Qu XB. [Inhibitory effect of losartan on prostatic hyperplasia in spontaneous hypertension rats and its pathophysiological mechanism]. Zhonghua Nan Ke Xue. 2012;18:600–5.
Yu W, Zhao YY, Zhang ZW, Guo YL, Jin J. Angiotension II receptor 1 blocker modifies the expression of apoptosis-related proteins and transforming growth factor-beta1 in prostate tissue of spontaneously hypertensive rats. BJU Int. 2007;100:1161–5.
Mostafa F, Mantawy EM, Azab SS, El-Demerdash E. The angiotensin converting enzyme inhibitor captopril attenuates testosterone-induced benign prostatic hyperplasia in rats; a mechanistic approach. Eur J Pharm. 2019;865:172729.
Ishola IO, Anunobi CC, Tijani KH, Afolayan O, Udokwu VU. Potential of telmisartan in the treatment of benign prostatic hyperplasia. Fundam Clin Pharm. 2017;31:643–51.
Acknowledgements
This study was supported by JSPS KAKENHI (grant numbers 19K09673 and 22K09450). The author thanks Editage (www.editage.com) for the English language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shimizu, S. Insights into the associative role of hypertension and angiotensin II receptor in lower urinary tract dysfunction. Hypertens Res 47, 987–997 (2024). https://doi.org/10.1038/s41440-024-01597-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41440-024-01597-8