Key Points
-
Pain in rheumatoid arthritis (RA) results from interplay between joint pathology and processing of pain signals by peripheral nerves, spinal and supraspinal pain pathways
-
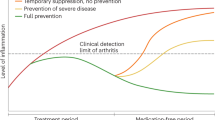
Despite good control of inflammation, pain often remains problematic in RA
-
Animal models and modern imaging techniques have illuminated the complex mechanisms that underlie RA pain
-
Fatigue, stiffness and psychological distress are integral components of the RA pain experience
-
Multimodal management of RA pain includes pharmacological, psychological and exercise-based interventions
Abstract
People with rheumatoid arthritis (RA) identify pain as their most important symptom, one that often persists despite optimal control of inflammatory disease. RA pain arises from multiple mechanisms, involving inflammation, peripheral and central pain processing and, with disease progression, structural change within the joint. Consequently, RA pain has a wide range of characteristics—constant or intermittent, localized or widespread—and is often associated with psychological distress and fatigue. Dominant pain mechanisms in an individual are identified by critical evaluation of clinical symptoms and signs, and by laboratory and imaging tests. Understanding these mechanisms is essential for effective management, although evidence from preclinical models should be interpreted with caution. A range of pharmacological analgesic and immunomodulatory agents, psychological interventions and surgery may help manage RA pain. Pain contributes importantly to the clinical assessment of inflammatory disease activity, and noninflammatory components of RA pain should be considered when gauging eligibility for or response to biologic agents. Further randomized controlled trials are required to determine the optimal usage of analgesics in RA, and novel agents with greater efficacy and lower propensity for adverse events are urgently needed. Meanwhile, targeted use of existing treatments could reduce pain in people with RA.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Merskey, H. & Bogduk, N. (eds) Classification of Chronic Pain (IASP Publications, Seattle, 1994).
Heiberg, T. & Kvien, T. K. Preferences for improved health examined in 1,024 patients with rheumatoid arthritis: pain has highest priority. Arthritis Rheum. 47, 391–397 (2002).
da Rocha Castelar Pinheiro, G., Khandker, R. K., Sato, R., Rose, A. & Piercy, J. Impact of rheumatoid arthritis on quality of life, work productivity and resource utilisation: an observational, cross-sectional study in Brazil. Clin. Exp. Rheumatol. 31, 334–340 (2013).
Lee, Y. C. et al. Pain persists in DAS28 rheumatoid arthritis remission but not in ACR/EULAR remission: a longitudinal observational study. Arthritis Res. Ther. 13, R83 (2011).
Burckhardt, C. S. The use of the McGill Pain Questionnaire in assessing arthritis pain. Pain 19, 305–314 (1984).
Charter, R. A., Nehemkis, A. M., Keenan, M. A., Person, D. & Prete, P. E. The nature of arthritis pain. Br. J. Rheumatol. 24, 53–60 (1985).
Roche, P. A., Klestov, A. C. & Heim, H. M. Description of stable pain in rheumatoid arthritis: a 6 year study. J. Rheumatol. 30, 1733–1738 (2003).
Harden, R. N. Chronic neuropathic pain. Mechanisms, diagnosis, and treatment. Neurologist 11, 111–122 (2005).
Perrot, S., Dieude, P., Perocheau, D. & Allanore, Y. Comparison of pain, pain burden, coping strategies, and attitudes between patients with systemic sclerosis and patients with rheumatoid arthritis: a cross-sectional study. Pain Med. 14, 1776–1785 (2013).
Cook, D. B. et al. Functional imaging of pain in patients with primary fibromyalgia. J. Rheumatol. 31, 364–378 (2004).
Hochman, J. R., Davis, A. M., Elkayam, J., Gagliese, L. & Hawker, G. A. Neuropathic pain symptoms on the modified painDETECT correlate with signs of central sensitization in knee osteoarthritis. Osteoarthritis Cartilage 21, 1236–1242 (2013).
Mengshoel, A. M. & Forre, O. Pain and fatigue in patients with rheumatic disorders. Clin. Rheumatol. 12, 515–521 (1993).
Zautra, A. J., Hamilton, N. A., Potter, P. & Smith, B. Field research on the relationship between stress and disease activity in rheumatoid arthritis. Ann. NY Acad. Sci. 876, 397–412 (1999).
Lems, W. F. & Dijkmans, B. A. C. in Rheumatoid Arthritis: New Frontiers in Pathogenesis and Treatment (eds Firestein, G. S., Panayi, G. S. & Wollheim, F. A.) 213–225 (OUP, Oxford, 2000).
Wolfe, F., Hawley, D. J. & Wilson, K. The prevalence and meaning of fatigue in rheumatic disease. J. Rheumatol. 23, 1407–1417 (1996).
Crosby, L. J. Factors which contribute to fatigue associated with rheumatoid arthritis. J. Adv. Nurs. 16, 974–981 (1991).
Pollard, L. C., Choy, E. H., Gonzalez, J., Khoshaba, B. & Scott, D. L. Fatigue in rheumatoid arthritis reflects pain, not disease activity. Rheumatology (Oxford) 45, 885–889 (2006).
Belza, B. L. Comparison of self-reported fatigue in rheumatoid arthritis and controls. J. Rheumatol. 22, 639–643 (1995).
Wolfe, F. Fibromyalgia: the clinical syndrome. Rheum. Dis. Clin. North Am. 15, 1–18 (1989).
Feuerstein, M., Carter, R. L. & Papciak, A. S. A prospective analysis of stress and fatigue in recurrent low back pain. Pain 31, 333–344 (1987).
Nicassio, P. M. et al. The contribution of pain and depression to self-reported sleep disturbance in patients with rheumatoid arthritis. Pain 153, 107–112 (2012).
Lee, Y. C. et al. The role of sleep problems in central pain processing in rheumatoid arthritis. Arthritis Rheum. 65, 59–68 (2013).
Scarvell, J. & Elkins, M. R. Aerobic exercise is beneficial for people with rheumatoid arthritis. Br. J. Sports Med. 45, 1008–1009 (2011).
Munsterman, T., Takken, T. & Wittink, H. Low aerobic capacity and physical activity not associated with fatigue in patients with rheumatoid arthritis: a cross-sectional study. J. Rehabil. Med. 45, 164–169 (2013).
Edwards, R. R., Cahalan, C., Mensing, G., Smith, M. & Haythornthwaite, J. A. Pain, catastrophizing, and depression in the rheumatic diseases. Nat. Rev. Rheumatol. 7, 216–224 (2011).
Jones, A. K., Huneke, N. T., Lloyd, D. M., Brown, C. A. & Watson, A. Role of functional brain imaging in understanding rheumatic pain. Curr. Rheumatol. Rep. 14, 557–567 (2012).
Wolfe, F., Hauser, W., Hassett, A. L., Katz, R. S. & Walitt, B. T. The development of fibromyalgia-I: examination of rates and predictors in patients with rheumatoid arthritis (RA). Pain 152, 291–299 (2011).
Lee, Y. C. et al. Incidence and predictors of secondary fibromyalgia in an early arthritis cohort. Ann. Rheum. Dis. 72, 949–954 (2013).
Schaible, H.-G. & Brauer, R. in Pain Models: Translational Relevance and Applications (eds Handwerker, H. O. & Arendt-Nielsen, L.) 149–162 (IASP Press, 2013).
Vincent, T. L., Williams, R. O., Maciewicz, R., Silman, A. & Garside, P. Mapping pathogenesis of arthritis through small animal models. Rheumatology (Oxford) 51, 1931–1941 (2012).
McQueen, D. S., Iggo, A., Birrell, G. J. & Grubb, B. D. Effects of paracetamol and aspirin on neural activity of joint mechanonociceptors in adjuvant arthritis. Br. J. Pharmacol. 104, 178–182 (1991).
Shinoda, M., Ozaki, N., Asai, H., Nagamine, K. & Sugiura, Y. Changes in P2X3 receptor expression in the trigeminal ganglion following monoarthritis of the temporomandibular joint in rats. Pain 116, 42–51 (2005).
Shubayev, V. I., Kato, K. & Myers, R. E. in Translational Pain Research: from Mouse to Man (eds Kruger, L. & Light, A. R.) (CRC Press, 2010).
Brennan, F. M. & McInnes, I. B. Evidence that cytokines play a role in rheumatoid arthritis. J. Clin. Invest. 118, 3537–3545 (2008).
Scuri, M., Samsell, L. & Piedimonte, G. The role of neurotrophins in inflammation and allergy. Inflamm. Allergy Drug Targets 9, 173–180 (2010).
Antonelli, A. et al. Chemokine (C-X-C motif) ligand (CXCL)10 in autoimmune diseases. Autoimmun. Rev. 13, 272–280 (2014).
Fava, R. A. et al. Vascular permeability factor/endothelial growth factor (VPF/VEGF): accumulation and expression in human synovial fluids and rheumatoid synovial tissue. J. Exp. Med. 180, 341–346 (1994).
Toth, K. et al. Synovial fluid β-endorphin level in avascular necrosis, rheumatoid arthritis, and osteoarthritis of the femoral head and knee. A controlled pilot study. Clin. Rheumatol. 30, 537–540 (2011).
Denko, C. W. & Malemud, C. J. Role of the growth hormone/insulin-like growth factor-1 paracrine axis in rheumatic diseases. Semin. Arthritis Rheum. 35, 24–34 (2005).
Wright, H. L., Bucknall, R. C., Moots, R. J. & Edwards, S. W. Analysis of SF and plasma cytokines provides insights into the mechanisms of inflammatory arthritis and may predict response to therapy. Rheumatology (Oxford) 51, 451–459 (2012).
Xu, Z. Z. et al. Resolvins RvE1 and RvD1 attenuate inflammatory pain via central and peripheral actions. Nat. Med. 16, 592–597 (2010).
Giera, M. et al. Lipid and lipid mediator profiling of human synovial fluid in rheumatoid arthritis patients by means of LC-MS/MS. Biochim. Biophys. Acta 1821, 1415–1424 (2012).
Meeus, M. et al. Central sensitization in patients with rheumatoid arthritis: a systematic literature review. Semin. Arthritis. Rheum. 41, 556–567 (2012).
Malfait, A.-M. & Schnitzer, T. J. Towards a mechanism-based approach to pain management in osteoarthritis. Nat. Rev. Rheumatol. 9, 654–664 (2013).
Edwards, R. R. et al. Enhanced reactivity to pain in patients with rheumatoid arthritis. Arthritis Res. Ther. 11, R61 (2009).
Pollard, L. C., Ibrahim, F., Choy, E. H. & Scott, D. L. Pain thresholds in rheumatoid arthritis: the effect of tender point counts and disease duration. J. Rheumatol. 39, 28–31 (2012).
Wendler, J. et al. Patients with rheumatoid arthritis adapt differently to repetitive painful stimuli compared to healthy controls. J. Clin. Neurosci. 8, 272–277 (2001).
Arendt-Nielsen, L. & Petersen-Felix, S. Wind-up and neuroplasticity: is there a correlation to clinical pain? Eur. J. Anaesthesiol. Suppl. 10, 1–7 (1995).
Morris, V. H., Cruwys, S. C. & Kidd, B. L. Characterisation of capsaicin-induced mechanical hyperalgesia as a marker for altered nociceptive processing in patients with rheumatoid arthritis. Pain 71, 179–186 (1997).
Shenker, N. G., Haigh, R. C., Mapp, P. I., Harris, N. & Blake, D. R. Contralateral hyperalgesia and allodynia following intradermal capsaicin injection in man. Rheumatology (Oxford) 47, 1417–1421 (2008).
Hummel, T., Schiessl, C., Wendler, J. & Kobal, G. Peripheral and central nervous changes in patients with rheumatoid arthritis in response to repetitive painful stimulation. Int. J. Psychophysiol. 37, 177–183 (2000).
Hess, A. et al. Blockade of TNF-α rapidly inhibits pain responses in the central nervous system. Proc. Natl Acad. Sci. USA 108, 3731–3736 (2011).
Schweinhardt, P. et al. Investigation into the neural correlates of emotional augmentation of clinical pain. Neuroimage 40, 759–766 (2008).
Robinson, M. J. et al. Depression and pain. Front. Biosci. (Landmark Ed.) 14, 5031–5051 (2009).
Jones, A. K. et al. Endogenous opiate response to pain in rheumatoid arthritis and cortical and subcortical response to pain in normal volunteers using positron emission tomography. Int. J. Clin. Pharmacol. Res. 11, 261–266 (1991).
Wartolowska, K. et al. Structural changes of the brain in rheumatoid arthritis. Arthritis Rheum. 64, 371–379 (2012).
Leffler, A. S., Kosek, E., Lerndal, T., Nordmark, B. & Hansson, P. Somatosensory perception and function of diffuse noxious inhibitory controls (DNIC) in patients suffering from rheumatoid arthritis. Eur. J. Pain 6, 161–176 (2002).
Jones, A. K. & Derbyshire, S. W. Reduced cortical responses to noxious heat in patients with rheumatoid arthritis. Ann. Rheum. Dis. 56, 601–607 (1997).
Meeus, M. et al. Does acetaminophen activate endogenous pain inhibition in chronic fatigue syndrome/fibromyalgia and rheumatoid arthritis? A double-blind randomized controlled cross-over trial. Pain Physician 16, E61–E70 (2013).
Lee, Y. C. et al. The role of sleep problems in central pain processing in rheumatoid arthritis. Arthritis Rheum. 65, 59–68 (2013).
Edwards, R. R., Bingham, C. O. 3rd, Bathon, J. & Haythornthwaite, J. A. Catastrophizing and pain in arthritis, fibromyalgia, and other rheumatic diseases. Arthritis Rheum. 55, 325–332 (2006).
Edwards, R. R., Cahalan, C., Mensing, G., Smith, M. & Haythornthwaite, J. A. Pain, catastrophizing, and depression in the rheumatic diseases. Nat. Rev. Rheumatol. 7, 216–224 (2011).
Goldenberg, D. L. The interface of pain and mood disturbances in the rheumatic diseases. Semin. Arthritis Rheum. 40, 15–31 (2010).
Brown, S. C., Glass, J. M. & Park, D. C. The relationship of pain and depression to cognitive function in rheumatoid arthritis patients. Pain 96, 279–284 (2002).
Odegard, S., Finset, A., Mowinckel, P., Kvien, T. K. & Uhlig, T. Pain and psychological health status over a 10-year period in patients with recent onset rheumatoid arthritis. Ann. Rheum. Dis. 66, 1195–1201 (2007).
Wolfe, F. & Michaud, K. Predicting depression in rheumatoid arthritis: the signal importance of pain extent and fatigue, and comorbidity. Arthritis Rheum. 61, 667–673 (2009).
Fifield, J., Tennen, H., Reisine, S. & McQuillan, J. Depression and the long-term risk of pain, fatigue, and disability in patients with rheumatoid arthritis. Arthritis Rheum. 41, 1851–1857 (1998).
Zautra, A. J. et al. Depression history, stress, and pain in rheumatoid arthritis patients. J. Behav. Med. 30, 187–197 (2007).
Conner, T. S. et al. Coping with rheumatoid arthritis pain in daily life: within-person analyses reveal hidden vulnerability for the formerly depressed. Pain 126, 198–209 (2006).
Rupp, I., Boshuizen, H. C., Dinant, H. J., Jacobi, C. E. & van den Bos, G. A. M. Disability and health-related quality of life among patients with rheumatoid arthritis: association with radiographic joint damage, disease activity, pain, and depressive symptoms. Scand. J. Rheumatol. 35, 175–181 (2006).
Smedstad, L. M., Vaglum, P., Kvien, T. K. & Moum, T. The relationship between self-reported pain and sociodemographic variables, anxiety, and depressive symptoms in rheumatoid arthritis. J. Rheumatol. 22, 514–520 (1995).
Keefe, F. J. et al. Pain coping strategies and coping efficacy in rheumatoid arthritis: a daily process analysis. Pain 69, 35–42 (1997).
Urrows, S., Affleck, G., Tennen, H. & Higgins, P. Unique clinical and psychological correlates of fibromyalgia tender points and joint tenderness in rheumatoid arthritis. Arthritis Rheum. 37, 1513–1520 (1994).
Lee, Y. C. et al. The relationship between disease activity, sleep, psychiatric distress and pain sensitivity in rheumatoid arthritis: a cross-sectional study. Arthritis Res. Ther. 11, R160 (2009).
Dharmshaktu, P., Tayal, V. & Kalra, B. S. Efficacy of antidepressants as analgesics: a review. J. Clin. Pharmacol. 52, 6–17 (2012).
Zautra, A. J. et al. Comparison of cognitive behavioral and mindfulness meditation interventions on adaptation to rheumatoid arthritis for patients with and without history of recurrent depression. J. Consult. Clin. Psychol. 76, 408–421 (2008).
Edwards, R. R. et al. Moderators of the negative effects of catastrophizing in arthritis. Pain Med. 11, 591–599 (2010).
Fitzcharles, M.-A., DaCosta, D., Ware, M. A. & Shir, Y. Patient barriers to pain management may contribute to poor pain control in rheumatoid arthritis. J. Pain 10, 300–305 (2009).
Holtzman, S. & Delongis, A. One day at a time: the impact of daily satisfaction with spouse responses on pain, negative affect and catastrophizing among individuals with rheumatoid arthritis. Pain 131, 202–213 (2007).
Covic, T., Adamson, B., Spencer, D. & Howe, G. A biopsychosocial model of pain and depression in rheumatoid arthritis: a 12-month longitudinal study. Rheumatology (Oxford) 42, 1287–1294 (2003).
Covic, T., Adamson, B. & Hough, M. The impact of passive coping on rheumatoid arthritis pain. Rheumatology (Oxford) 39, 1027–1030 (2000).
Knittle, K. P. et al. Effect of self-efficacy and physical activity goal achievement on arthritis pain and quality of life in patients with rheumatoid arthritis. Arthritis Care Res. (Hoboken) 63, 1613–1619 (2011).
Reese, J. B., Somers, T. J., Keefe, F. J., Mosley-Williams, A. & Lumley, M. A. Pain and functioning of rheumatoid arthritis patients based on marital status: is a distressed marriage preferable to no marriage? J. Pain 11, 958–964 (2012).
Waltz, M., Kriegel, W. & van't Pad Bosch, P. The social environment and health in rheumatoid arthritis: marital quality predicts individual variability in pain severity. Arthritis Care Res. (Hoboken) 11, 356–374 (1998).
Mapp, P. I. & Walsh, D. A. Mechanisms and targets of angiogenesis and nerve growth in osteoarthritis. Nat. Rev. Rheumatol. 8, 390–398 (2012).
Black, P. H. Stress and the inflammatory response: a review of neurogenic inflammation. Brain Behav. Immun. 16, 622–653 (2002).
Edwards, R. R. et al. Association of catastrophizing with interleukin-6 responses to acute pain. Pain 140, 135–144 (2008).
Geenen, R., van Middendorp, H. & Bijlsma, J. W. The impact of stressors on health status and hypothalamic–pituitary–adrenal axis and autonomic nervous system responsiveness in rheumatoid arthritis. Ann. NY Acad. Sci. 1069, 77–97 (2006).
Dekkers, J. C. et al. Biopsychosocial mediators and moderators of stress-health relationships in patients with recently diagnosed rheumatoid arthritis. Arthritis Rheum. 45, 307–316 (2001).
Steptoe, A., Hamer, M. & Chida, Y. The effects of acute psychological stress on circulating inflammatory factors in humans: a review and meta-analysis. Brain Behav. Immun. 21, 901–912 (2007).
Pace, T. W. & Heim, C. M. A short review on the psychoneuroimmunology of posttraumatic stress disorder: from risk factors to medical comorbidities. Brain Behav. Immun. 25, 6–13 (2011).
Gimeno, D. et al. Adult socioeconomic position, C-reactive protein and interleukin-6 in the Whitehall II prospective study. Eur. J. Epidemiol. 22, 675–683 (2007).
Qureshi, S. U., Pyne, J. M., Magruder, K. M., Schulz, P. E. & Kunik, M. E. The link between post-traumatic stress disorder and physical comorbidities: a systematic review. Psychiatr. Q. 80, 87–97 (2009).
Peters, M. J. et al. Genome-wide association study meta-analysis of chronic widespread pain: evidence for involvement of the 5p15.2 region. Ann. Rheum. Dis. 72, 427–436 (2013).
McWilliams, D. F. et al. Predictors of change in bodily pain in early rheumatoid arthritis: an inception cohort study. Arthritis Care Res. (Hoboken) 64, 1505–1513 (2012).
Sharpe, L., Sensky, T. & Allard, S. The course of depression in recent onset rheumatoid arthritis: the predictive role of disability, illness perceptions, pain and coping. J. Psychosom. Res. 51, 713–719 (2001).
Odegard, S., Finset, A., Mowinckel, P., Kvien, T. K. & Uhlig, T. Pain and psychological health status over a 10-year period in patients with recent onset rheumatoid arthritis. Ann. Rheum. Dis. 66, 1195–1201 (2007).
Pollard, L. C., Kingsley, G. H., Choy, E. H. & Scott, D. L. Fibromyalgic rheumatoid arthritis and disease assessment. Rheumatology (Oxford) 49, 924–928 (2010).
Walsh, D. A. & McWilliams, D. F. Pain in rheumatoid arthritis. Curr. Pain Headache Rep. 16, 509–517 (2012).
Kroenke, K., Krebs, E. E. & Bair, M. J. Pharmacotherapy of chronic pain: a synthesis of recommendations from systematic reviews. Gen. Hosp. Psychiatry 31, 206–219 (2009).
Doherty, M. et al. A randomised controlled trial of ibuprofen, paracetamol or a combination tablet of ibuprofen/paracetamol in community-derived people with knee pain. Ann. Rheum. Dis. 70, 1534–1541 (2011).
Zhang, W., Robertson, J., Jones, A. C., Dieppe, P. A. & Doherty, M. The placebo effect and its determinants in osteoarthritis: meta-analysis of randomised controlled trials. Ann. Rheum. Dis. 67, 1716–1723 (2008).
Frank, R. G. et al. Antidepressant analgesia in rheumatoid arthritis. J. Rheumatol. 15, 1632–1638 (1988).
Lemmel, E. M. et al. Efficacy and safety of meloxicam in patients with rheumatoid arthritis. J. Rheumatol. 24, 282–290 (1997).
Blyth, T., Hunter, J. A. & Stirling, A. Pain relief in the rheumatoid knee after steroid injection. A single-blind comparison of hydrocortisone succinate, and triamcinolone acetonide or hexacetonide. Br. J. Rheumatol. 33, 461–463 (1994).
Kirwan, J. R. et al. A randomised placebo controlled 12 week trial of budesonide and prednisolone in rheumatoid arthritis. Ann. Rheum. Dis. 63, 688–695 (2004).
Choy, E. H., Kingsley, G. H., Corkill, M. M. & Panayi, G. S. Intramuscular methylprednisolone is superior to pulse oral methylprednisolone during the induction phase of chrysotherapy. Br. J. Rheumatol. 32, 734–739 (1993).
Kirwan, J. R. The effect of glucocorticoids on joint destruction in rheumatoid arthritis. The Arthritis and Rheumatism Council Low-Dose Glucocorticoid Study Group. N. Engl. J. Med. 333, 142–146 (1995).
Hickling, P., Jacoby, R. K. & Kirwan, J. R. Joint destruction after glucocorticoids are withdrawn in early rheumatoid arthritis. Arthritis and Rheumatism Council Low Dose Glucocorticoid Study Group. Br. J. Rheumatol. 37, 930–936 (1998).
Williams, H. J. et al. Comparison of low-dose oral pulse methotrexate and placebo in the treatment of rheumatoid arthritis. A controlled clinical trial. Arthritis Rheum. 28, 721–730 (1985).
Pinals, R. S., Kaplan, S. B., Lawson, J. G. & Hepburn, B. Sulfasalazine in rheumatoid arthritis. A double-blind, placebo-controlled trial. Arthritis Rheum. 29, 1427–1434 (1986).
Smolen, J. S. et al. Efficacy and safety of leflunomide compared with placebo and sulphasalazine in active rheumatoid arthritis: a double-blind, randomised, multicentre trial. European Leflunomide Study Group. Lancet 353, 259–266 (1999).
National Collaborating Centre for Chronic conditions (UK). Rheumatoid arthritis: national clinical guidelines for management and treatment in adults (NICE Clinical Guidelines, No. 79) [online], (2009).
van der Kooij, S. M. et al. Patient-reported outcomes in a randomized trial comparing four different treatment strategies in recent-onset rheumatoid arthritis. Arthritis Rheum. 61, 4–12 (2009).
Weinblatt, M. E. et al. A trial of etanercept, a recombinant tumor necrosis factor receptor:Fc fusion protein, in patients with rheumatoid arthritis receiving methotrexate. N. Engl. J. Med. 340, 253–259 (1999).
Keystone, E. C. et al. Radiographic, clinical, and functional outcomes of treatment with adalimumab (a human anti-tumor necrosis factor monoclonal antibody) in patients with active rheumatoid arthritis receiving concomitant methotrexate therapy: a randomized, placebo-controlled, 52-week trial. Arthritis Rheum. 50, 1400–1411 (2004).
Andratsch, M. et al. A key role for gp130 expressed on peripheral sensory nerves in pathological pain. J. Neurosci. 29, 13473–13483 (2009).
Russell, F. A., Fernandes, E. S., Courade, J. P., Keeble, J. E. & Brain, S. D. Tumour necrosis factor α mediates transient receptor potential vanilloid 1-dependent bilateral thermal hyperalgesia with distinct peripheral roles of interleukin-1β, protein kinase C and cyclooxygenase-2 signalling. Pain 142, 264–274 (2009).
Nishimoto, N. et al. Study of active controlled monotherapy used for rheumatoid arthritis, an IL-6 inhibitor (SAMURAI): evidence of clinical and radiographic benefit from an X ray reader-blinded randomised controlled trial of tocilizumab. Ann. Rheum. Dis. 66, 1162–1167 (2007).
Rech, J. et al. Association of brain functional magnetic resonance activity with response to tumor necrosis factor inhibition in rheumatoid arthritis. Arthritis Rheum. 65, 325–333 (2013).
Drossaers-Bakker, K. W. et al. Long-term outcome in rheumatoid arthritis: a simple algorithm of baseline parameters can predict radiographic damage, disability, and disease course at 12-year followup. Arthritis Rheum. 47, 383–390 (2002).
Sokka, T., Kankainen, A. & Hannonen, P. Scores for functional disability in patients with rheumatoid arthritis are correlated at higher levels with pain scores than with radiographic scores. Arthritis Rheum. 43, 386–389 (2000).
Sarzi-Puttini, P. et al. Correlation of the score for subjective pain with physical disability, clinical and radiographic scores in recent onset rheumatoid arthritis. BMC Musculoskelet. Disord. 3, 18 (2002).
Judge, A. et al. Predictors of outcomes of total knee replacement surgery. Rheumatology (Oxford) 51, 1804–1813 (2012).
Dore, D. et al. Natural history and clinical significance of MRI-detected bone marrow lesions at the knee: a prospective study in community dwelling older adults. Arthritis Res. Ther. 12, R223 (2010).
Allaart, C. F. & Huizinga, T. W. Treatment strategies in recent onset rheumatoid arthritis. Curr. Opin. Rheumatol. 23, 241–244 (2011).
Schaible, H. G. et al. The role of proinflammatory cytokines in the generation and maintenance of joint pain. Ann. NY Acad. Sci. 1193, 60–69 (2010).
Schipper, L. G., Fransen, J., den Broeder, A. A. & Van Riel, P. L. Time to achieve remission determines time to be in remission. Arthritis Res. Ther. 12, R97 (2010).
Pradhan, E. K. et al. Effect of mindfulness-based stress reduction in rheumatoid arthritis patients. Arthritis Rheum. 57, 1134–1142 (2007).
Knittle, K., Maes, S. & de Gucht, V. Psychological interventions for rheumatoid arthritis: examining the role of self-regulation with a systematic review and meta-analysis of randomized controlled trials. Arthritis Care Res. (Hoboken) 62, 1460–1472 (2010).
Koltyn, K. F. & Umeda, M. Exercise, hypoalgesia and blood pressure. Sports Med. 36, 207–214 (2006).
Tendzegolskis, Z., Viru, A. & Orlova, E. Exercise-induced changes of endorphin contents in hypothalamus, hypophysis, adrenals and blood plasma. Int. J. Sports Med. 12, 495–497 (1991).
Guieu, R., Blin, O., Pouget, J. & Serratrice, G. Nociceptive threshold and physical activity. Can. J. Neurol. Sci. 19, 69–71 (1992).
Baillet, A. et al. Efficacy of cardiorespiratory aerobic exercise in rheumatoid arthritis: meta-analysis of randomized controlled trials. Arthritis Care Res. (Hoboken) 62, 984–992 (2010).
Uhlig, T., Fongen, C., Steen, E., Christie, A. & Odegard, S. Exploring Tai Chi in rheumatoid arthritis: a quantitative and qualitative study. BMC Musculoskelet. Disord. 11, 43 (2010).
Kosek, E., Ekholm, J. & Hansson, P. Modulation of pressure pain thresholds during and following isometric contraction in patients with fibromyalgia and in healthy controls. Pain 64, 415–423 (1996).
Radboud University Nijmegen Medical Center. DAS-score.nl: disease activity score in rheumatoid arthritis [online], (2014).
Janta, I. et al. Patient self-assessment and physician's assessment of rheumatoid arthritis activity: which is more realistic in remission status? A comparison with ultrasonography. Rheumatology (Oxford) http://dx.doi.org/10.1093/rheumatology/ket297 (2013).
Wakefield, R. J. et al. Should oligoarthritis be reclassified? Ultrasound reveals a high prevalence of subclinical disease. Ann. Rheum. Dis. 63, 382–385 (2004).
Shinoda, M. et al. Nerve terminals extend into the temporomandibular joint of adjuvant arthritic rats. Eur. J. Pain 7, 493–505 (2003).
Yamazaki, Y., Ren, K., Shimada, M. & Iwata, K. Modulation of paratrigeminal nociceptive neurons following temporomandibular joint inflammation in rats. Exp. Neurol. 214, 209–218 (2008).
Schaible, H. G., Ebersberger, A. & Von Banchet, G. S. Mechanisms of pain in arthritis. Ann. NY Acad. Sci. 966, 343–354 (2002).
Imbe, H. et al. Orofacial deep and cutaneous tissue inflammation and trigeminal neuronal activation. Implications for persistent temporomandibular pain. Cells Tissues Organs 169, 238–247 (2001).
Cruz, C. D., Neto, F. L., Castro-Lopes, J., McMahon, S. B. & Cruz, F. Inhibition of ERK phosphorylation decreases nociceptive behaviour in monoarthritic rats. Pain 116, 411–419 (2005).
Sharif Naeini, R., Cahill, C. M., Ribeiro-da-Silva, A., Menard, H. A. & Henry, J. L. Remodelling of spinal nociceptive mechanisms in an animal model of monoarthritis. Eur. J. Neurosci. 22, 2005–2015 (2005).
Abbadie, C., Besson, J. M. & Calvino, B. c-Fos expression in the spinal cord and pain-related symptoms induced by chronic arthritis in the rat are prevented by pretreatment with Freund adjuvant. J. Neurosci. 14, 5865–5871 (1994).
Chen, Y., Willcockson, H. H. & Valtschanoff, J. G. Influence of the vanilloid receptor TRPV1 on the activation of spinal cord glia in mouse models of pain. Exp. Neurol. 220, 383–390 (2009).
Hains, L. E. et al. Pain intensity and duration can be enhanced by prior challenge: initial evidence suggestive of a role of microglial priming. J. Pain 11, 1004–1014 (2010).
Bao, L. et al. Adjuvant-induced arthritis: IL-1 β, IL-6 and TNF-α are up-regulated in the spinal cord. Neuroreport 12, 3905–3908 (2001).
Honor, P. et al. Spinal substance P receptor expression and internalization in acute, short-term, and long-term inflammatory pain states. J. Neurosci. 19, 7670–7678 (1999).
Pezet, S., Onteniente, B., Grannec, G. & Calvino, B. Chronic pain is associated with increased TrkA immunoreactivity in spinoreticular neurons. J. Neurosci. 19, 5482–5492 (1999).
Sun, S. et al. New evidence for the involvement of spinal fractalkine receptor in pain facilitation and spinal glial activation in rat model of monoarthritis. Pain 129, 64–75 (2007).
Guilbaud, G., Benoist, J. M., Gautron, M. & Kayser, V. Aspirin clearly depresses responses of ventrobasal thalamus neurons to joint stimuli in arthritic rats. Pain 13, 153–163 (1982).
Neto, F. L. et al. Supraspinal metabolic activity changes in the rat during adjuvant monoarthritis. Neuroscience 94, 607–621 (1999).
Omura, K., Imai, S., Maeda, T. & Hukuda, S. Prolonged and increasing expression of Fos related antigens in the hippocampus of adjuvant arthritic rats. J. Rheumatol. 25, 936–944 (1998).
Williams, F. G. & Beitz, A. J. Chronic pain increases brainstem proneurotensin/neuromedin-N mRNA expression: a hybridization-histochemical and immunohistochemical study using three different rat models for chronic nociception. Brain Res. 611, 87–102 (1993).
Xie, H., Ma, F., Zhang, Y.-Q., Gao, X. & Wu, G.-C. Expression of 5-HT2A receptor mRNA in some nuclei of brain stem enhanced in monoarthritic rats. Brain Res. 954, 94–99 (2002).
Danziger, N., Weil-Fugazza, J., Le Bars, D. & Bouhassira, D. Alteration of descending modulation of nociception during the course of monoarthritis in the rat. J. Neurosci. 19, 2394–2400 (1999).
Constandil, L. et al. Effect of interleukin-1β on spinal cord nociceptive transmission of normal and monoarthritic rats after disruption of glial function. Arthritis Res. Ther. 11, R105 (2009).
Clark, A. K., Grist, J., Al-Kashi, A., Perretti, M. & Malcangio, M. Spinal cathepsin S and fractalkine contribute to chronic pain in the collagen-induced arthritis model. Arthritis Rheum. 64, 2038–2047 (2012).
Christianson, C. A. et al. Characterization of the acute and persistent pain state present in K/BxN serum transfer arthritis. Pain 151, 394–403 (2010).
von Banchet, G. S., Petrow, P. K., Brauer, R. & Schaible, H. G. Monoarticular antigen-induced arthritis leads to pronounced bilateral upregulation of the expression of neurokinin 1 and bradykinin 2 receptors in dorsal root ganglion neurons of rats. Arthritis Res. 2, 424–427 (2000).
Segond von Banchet, G., Petrow, P. K., Brauer, R. & Schaible, H. G. Monoarticular antigen-induced arthritis leads to pronounced bilateral upregulation of the expression of neurokinin 1 and bradykinin 2 receptors in dorsal root ganglion neurons of rats. Arthritis Res. 2, 424–427 (2000).
Boettger, M. K., Weber, K., Gajda, M., Brauer, R. & Schaible, H. G. Spinally applied ketamine or morphine attenuate peripheral inflammation and hyperalgesia in acute and chronic phases of experimental arthritis. Brain Behav. Immun. 24, 474–485 (2010).
Boettger, M. K. et al. Spinal tumor necrosis factor α neutralization reduces peripheral inflammation and hyperalgesia and suppresses autonomic responses in experimental arthritis: a role for spinal tumor necrosis factor α during induction and maintenance of peripheral inflammation. Arthritis Rheum. 62, 1308–1318 (2010).
Natura, G. et al. Neuronal prostaglandin E2 receptor subtype EP3 mediates antinociception during inflammation. Proc. Natl Acad. Sci. USA 110, 13648–13653 (2013).
Hazlewood, G., van der Heijde, D. M. & Bombardier, C. Paracetamol for the management of pain in inflammatory arthritis: a systematic literature review. J. Rheumatol. Suppl. 90, 11–16 (2012).
Wienecke, T. & Gotzsche, P. C. Paracetamol versus nonsteroidal anti-inflammatory drugs for rheumatoid arthritis. Cochrane Database of Systematic Reviews 2004, Issue 1. Art. No.: CD003789. http://dx.doi.org/10.1002/14651858.CD003789.pub2.
McCormack, P. L. Celecoxib: a review of its use for symptomatic relief in the treatment of osteoarthritis, rheumatoid arthritis and ankylosing spondylitis. Drugs 71, 2457–2489 (2011).
Chen, Y. F. et al. Cyclooxygenase-2 selective non-steroidal anti-inflammatory drugs (etodolac, meloxicam, celecoxib, rofecoxib, etoricoxib, valdecoxib and lumiracoxib) for osteoarthritis and rheumatoid arthritis: a systematic review and economic evaluation. Health Technol. Assess. 12, 1–278 (2008).
Whittle, S. L., Richards, B. L., van der Heijde, D. M. & Buchbinder, R. The efficacy and safety of opioids in inflammatory arthritis: a Cochrane systematic review. J. Rheumatol. Suppl. 90, 40–46 (2012).
Richards, B. L., Whittle, S. L., van der Heijde, D. M. & Buchbinder, R. The efficacy and safety of antidepressants in inflammatory arthritis: a Cochrane systematic review. J. Rheumatol. Suppl. 90, 21–27 (2012).
Richards, B. L., Whittle, S. L. & Buchbinder, R. Neuromodulators for pain management in rheumatoid arthritis. Cochrane Database of Systematic Reviews 2012, Issue 1. Art. No.: CD008921. http://dx.doi.org/10.1002/14651858.CD008921.pub2.
International Association for the Study of Pain. IASP Taxonomy: pain terms [online], (2012).
Author information
Authors and Affiliations
Contributions
Both authors researched the data for the article, provided substantial contributions to discussions of content, wrote the article and undertook review and/or editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
D.A.W. is the holder of an Inflammation Competitive Research Programme (I-CRP) grant from Pfizer UK, and has previously acted as a consultant for Pfizer. D.F.M. is supported by the I-CRP grant.
Rights and permissions
About this article
Cite this article
Walsh, D., McWilliams, D. Mechanisms, impact and management of pain in rheumatoid arthritis. Nat Rev Rheumatol 10, 581–592 (2014). https://doi.org/10.1038/nrrheum.2014.64
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2014.64
This article is cited by
-
Anti-inflammatory and anti-arthritic potential of Coagulansin-A: in vitro and in vivo studies
Inflammopharmacology (2024)
-
Galectin-3 activates spinal microglia to induce inflammatory nociception in wild type but not in mice modelling Alzheimer’s disease
Nature Communications (2023)
-
Heat of the night: sleep disturbance activates inflammatory mechanisms and induces pain in rheumatoid arthritis
Nature Reviews Rheumatology (2023)
-
Roles of Phosphorylation of N-Methyl-d-Aspartate Receptor in Chronic Pain
Cellular and Molecular Neurobiology (2023)
-
Janus kinase inhibitors effectively improve pain across different disease activity states in rheumatoid arthritis
Internal and Emergency Medicine (2023)