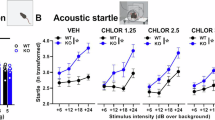
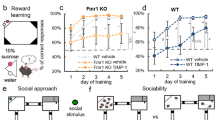
Abstract
Fragile X syndrome (FXS), the most common monogenic cause of inherited intellectual disability and autism1, is caused by the silencing of the FMR1 gene, leading to the loss of fragile X mental retardation protein (FMRP)2, a synaptically expressed RNA-binding protein regulating translation3. The Fmr1 knockout model recapitulates the main traits of the disease4. Uncontrolled activity of metabotropic glutamate receptor 5 (mGluR5)5,6 and mammalian target of rapamycin (mTOR) signaling7,8,9 seem crucial in the pathology of this disease. The endocannabinoid system (ECS) is a key modulator of synaptic plasticity, cognitive performance, anxiety, nociception and seizure susceptibility10, all of which are affected in FXS. The cannabinoid receptors CB1 (CB1R) and CB2 (CB2R) are activated by phospholipid-derived endocannabinoids, and CB1R-driven long-term regulation of synaptic strength, as a consequence of mGluR5 activation11, is altered in several brain areas of Fmr1 knockout mice12,13,14. We found that CB1R blockade in male Fmr1 knockout (Fmr1−/y) mice through pharmacological and genetic approaches normalized cognitive impairment, nociceptive desensitization, susceptibility to audiogenic seizures, overactivated mTOR signaling and altered spine morphology, whereas pharmacological blockade of CB2R normalized anxiolytic-like behavior. Some of these traits were also reversed by pharmacological inhibition of mTOR or mGluR5. Thus, blockade of ECS is a potential therapeutic approach to normalize specific alterations in FXS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
de Vries, B.B., Halley, D.J., Oostra, B.A. & Niermeijer, M.F. The fragile X syndrome. J. Med. Genet. 35, 579–589 (1998).
Verkerk, A.J. et al. Identification of a gene (FMR-1) containing a CGG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell 65, 905–914 (1991).
Darnell, J.C. et al. FMRP stalls ribosomal translocation on mRNAs linked to synaptic function and autism. Cell 146, 247–261 (2011).
Bakker, C.E. et al. Fmr1 knockout mice: a model to study fragile X mental retardation. The Dutch-Belgian Fragile X Consortium. Cell 78, 23–33 (1994).
Bear, M.F., Huber, K.M. & Warren, S.T. The mGluR theory of fragile X mental retardation. Trends Neurosci. 27, 370–377 (2004).
Michalon, A. et al. Chronic pharmacological mGlu5 inhibition corrects fragile X in adult mice. Neuron 74, 49–56 (2012).
Sharma, A. et al. Dysregulation of mTOR signaling in fragile X syndrome. J. Neurosci. 30, 694–702 (2010).
Hoeffer, C.A. et al. Altered mTOR signaling and enhanced CYFIP2 expression levels in subjects with fragile X syndrome. Genes Brain Behav. 11, 332–341 (2012).
Bhattacharya, A. et al. Genetic removal of p70 S6 kinase 1 corrects molecular, synaptic, and behavioral phenotypes in fragile X syndrome mice. Neuron 76, 325–337 (2012).
Kano, M. et al. Endocannabinoid-mediated control of synaptic transmission. Physiol. Rev. 89, 309–380 (2009).
Varma, N., Carlson, G.C., Ledent, C. & Alger, B.E. Metabotropic glutamate receptors drive the endocannabinoid system in hippocampus. J. Neurosci. 21, RC188 (2001).
Zhang, L. & Alger, B.E. Enhanced endocannabinoid signaling elevates neuronal excitability in fragile X syndrome. J. Neurosci. 30, 5724–5729 (2010).
Maccarrone, M. et al. Abnormal mGlu 5 receptor/endocannabinoid coupling in mice lacking FMRP and BC1 RNA. Neuropsychopharmacology 35, 1500–1509 (2010).
Jung, K.M. et al. Uncoupling of the endocannabinoid signalling complex in a mouse model of fragile X syndrome. Nat. Commun. 3, 1080 (2012).
Penagarikano, O., Mulle, J.G. & Warren, S.T. The pathophysiology of fragile x syndrome. Annu. Rev. Genomics Hum. Genet. 8, 109–129 (2007).
D'Hulst, C. & Kooy, R.F. The GABAA receptor: a novel target for treatment of fragile X? Trends Neurosci. 30, 425–431 (2007).
Henderson, C. et al. Reversal of disease-related pathologies in the fragile X mouse model by selective activation of GABAB receptors with arbaclofen. Sci. Transl. Med. 4, 152ra128 (2012).
Busquets-Garcia, A. et al. Differential role of anandamide and 2-arachidonoylglycerol in memory and anxiety-like responses. Biol. Psychiatry 70, 479–486 (2011).
Puighermanal, E., Busquets-Garcia, A., Maldonado, R. & Ozaita, A. Cellular and intracellular mechanisms involved in the cognitive impairment of cannabinoids. Phil. Trans. R. Soc. Lond. B 367, 3254–3263 (2012).
Hoeffer, C.A. & Klann, E. mTOR signaling: at the crossroads of plasticity, memory and disease. Trends Neurosci. 33, 67–75 (2010).
Puighermanal, E. et al. Cannabinoid modulation of hippocampal long-term memory is mediated by mTOR signaling. Nat. Neurosci. 12, 1152–1158 (2009).
Lafourcade, M. et al. Nutritional omega-3 deficiency abolishes endocannabinoid-mediated neuronal functions. Nat. Neurosci. 14, 345–350 (2011).
Richter, J.D. & Klann, E. Making synaptic plasticity and memory last: mechanisms of translational regulation. Genes Dev. 23, 1–11 (2009).
Troca-Marín, J.A., Alves-Sampaio, A. & Montesinos, M.L. Deregulated mTOR-mediated translation in intellectual disability. Prog. Neurobiol. 96, 268–282 (2012).
Kawamura, Y. et al. The CB1 cannabinoid receptor is the major cannabinoid receptor at excitatory presynaptic sites in the hippocampus and cerebellum. J. Neurosci. 26, 2991–3001 (2006).
Kang-Park, M.H. et al. Differential sensitivity of GABA A receptor–mediated IPSCs to cannabinoids in hippocampal slices from adolescent and adult rats. J. Neurophysiol. 98, 1223–1230 (2007).
Wei, H. et al. The therapeutic effect of memantine through the stimulation of synapse formation and dendritic spine maturation in autism and fragile X syndrome. PLoS ONE 7, e36981 (2012).
Price, T.J. et al. Decreased nociceptive sensitization in mice lacking the fragile X mental retardation protein: role of mGluR1/5 and mTOR. J. Neurosci. 27, 13958–13967 (2007).
Guindon, J. & Hohmann, A.G. The endocannabinoid system and pain. CNS Neurol. Disord. Drug Targets 8, 403–421 (2009).
Chen, L. & Toth, M. Fragile X mice develop sensory hyperreactivity to auditory stimuli. Neuroscience 103, 1043–1050 (2001).
Puighermanal, E. et al. Dissociation of the pharmacological effects of THC by mTOR blockade. Neuropsychopharmacology advance online publication, http://dx.doi.org/10.1038/npp.2013.31 (2013).
Castañé, A., Maldonado, R. & Valverde, O. Role of different brain structures in the behavioural expression of WIN 55,212–2 withdrawal in mice. Br. J. Pharmacol. 142, 1309–1317 (2004).
Guegan, T. et al. Operant behavior to obtain palatable food modifies neuronal plasticity in the brain reward circuit. Eur. Neuropsychopharmacol. 23, 146–159 (2013).
Noble, F. et al. Pain-suppressive effects on various nociceptive stimuli (thermal, chemical, electrical and inflammatory) of the first orally active enkephalin-metabolizing enzyme inhibitor RB 120. Pain 73, 383–391 (1997).
Acknowledgements
We thank C. Fernández-Avilés, D. Real, M. Linares and H. Gómez for expert technical assistance and O.J. Manzoni for helpful comments. Fmr1 knockout mice in the C57BL/6J background were kindly provided by D. Nelson at Baylor College of Medicine. A.B.-G. is the recipient of a predoctoral fellowship (Ministerio de Educación y Cultura). S.M. is the recipient of a Ramón y Cajal contract (Ministerio de Educación y Cultura). This study was supported by grants from Fundació La Marató de TV3 (#090910 to A.O.), Grants from the Ministerio de Ciencia e Innovación (#SAF2009-07309 to A.O., #SAF2011-29864 to R.M. and SAF2010-21547 to C.M.), CureFXS E-Rare EU/FIS PS09102673 to M.D., Instituto de Salud Carlos III (RD06/0001/0001 to R.M.), PLAN E (Plan Español para el Estímulo de la Economía y el Empleo), Generalitat de Catalunya (SGR-2009-00731 to R.M. and SGR-2009-00718 to R.d.l.T.) and ICREA (Institució Catalana de Recerca i Estudis Avançats) Academia to R.M.
Author information
Authors and Affiliations
Contributions
A.B.-G. participated in experimental design, conducted biochemical and behavioral experiments, and wrote the manuscript. M.G.-G. conducted biochemical and behavioral experiments. T.G. performed dendritic spine morphology analysis. C.A.-P. performed stereotaxic surgeries, and M.D. provided Fmr1 knockout and WT mice, and discussed the experiments. A.P. and R.d.l.T. measured endocannabinoid levels. S.M. designed and conducted electrophysiological experiments, and wrote the manuscript. A.P.-S. and S.M. were in charge of electrophysiological equipment. C.M. funded the project. R.M. funded the project and wrote the manuscript. A.O. conceptualized, participated in experimental design, supervised, funded the project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 500 kb)
Rights and permissions
About this article
Cite this article
Busquets-Garcia, A., Gomis-González, M., Guegan, T. et al. Targeting the endocannabinoid system in the treatment of fragile X syndrome. Nat Med 19, 603–607 (2013). https://doi.org/10.1038/nm.3127
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3127
This article is cited by
-
Efficacy of cannabinoids in neurodevelopmental and neuropsychiatric disorders among children and adolescents: a systematic review
European Child & Adolescent Psychiatry (2024)
-
The CB1 receptor interacts with cereblon and drives cereblon deficiency-associated memory shortfalls
EMBO Molecular Medicine (2024)
-
Role of the endocannabinoid system in fragile X syndrome: potential mechanisms for benefit from cannabidiol treatment
Journal of Neurodevelopmental Disorders (2023)
-
Endocannabinoids at the synapse and beyond: implications for neuropsychiatric disease pathophysiology and treatment
Neuropsychopharmacology (2023)
-
Anandamide and 2-arachidonoylglycerol differentially modulate autistic-like traits in a genetic model of autism based on FMR1 deletion in rats
Neuropsychopharmacology (2023)