Abstract
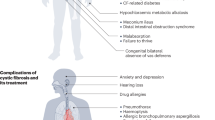
Cystic fibrosis is caused by mutations in the cystic fibrosis transmembrane conductance regulator (encoded by Cftr) that impair its role as an apical chloride channel that supports bicarbonate transport1. Individuals with cystic fibrosis show retained, thickened mucus that plugs airways and obstructs luminal organs2 as well as numerous other abnormalities that include inflammation of affected organs1, alterations in lipid metabolism3 and insulin resistance4. Here we show that colonic epithelial cells and whole lung tissue from Cftr-deficient mice show a defect in peroxisome proliferator–activated receptor-γ (PPAR-γ, encoded by Pparg) function that contributes to a pathological program of gene expression. Lipidomic analysis of colonic epithelial cells suggests that this defect results in part from reduced amounts of the endogenous PPAR-γ ligand 15-keto-prostaglandin E2 (15-keto-PGE2). Treatment of Cftr-deficient mice with the synthetic PPAR-γ ligand rosiglitazone partially normalizes the altered gene expression pattern associated with Cftr deficiency and reduces disease severity. Rosiglitazone has no effect on chloride secretion in the colon, but it increases expression of the genes encoding carbonic anhydrases 4 and 2 (Car4 and Car2), increases bicarbonate secretion and reduces mucus retention. These studies reveal a reversible defect in PPAR-γ signaling in Cftr-deficient cells that can be pharmacologically corrected to ameliorate the severity of the cystic fibrosis phenotype in mice.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
O'Sullivan, B.P. & Freedman, S.D. Cystic fibrosis. Lancet 373, 1891–1904 (2009).
Zuelzer, W.W. & Newton, W.A. Jr. The pathogenesis of fibrocystic disease of the pancreas; a study of 36 cases with special reference to the pulmonary lesions. Pediatrics 4, 53–69 (1949).
Freedman, S.D. et al. Association of cystic fibrosis with abnormalities in fatty acid metabolism. N. Engl. J. Med. 350, 560–569 (2004).
Hardin, D.S., LeBlanc, A., Lukenbough, S. & Seilheimer, D.K. Insulin resistance is associated with decreased clinical status in cystic fibrosis. J. Pediatr. 130, 948–956 (1997).
Snouwaert, J.N. et al. An animal model for cystic fibrosis made by gene targeting. Science 257, 1083–1088 (1992).
Eckman, E.A., Cotton, C.U., Kube, D.M. & Davis, P.B. Dietary changes improve survival of CFTR S489X homozygous mutant mouse. Am. J. Physiol. 269, L625–L630 (1995).
Clarke, L.L., Gawenis, L.R., Franklin, C.L. & Harline, M.C. Increased survival of CFTR knockout mice with an oral osmotic laxative. Lab. Anim. Sci. 46, 612–618 (1996).
Gene Ontology Consortium. Creating the gene ontology resource: design and implementation. Genome Res. 11, 1425–1433 (2001).
Ollero, M. et al. Decreased expression of peroxisome proliferator activated receptor γ in Cftr−/− mice. J. Cell. Physiol. 200, 235–244 (2004).
Yu, S. et al. Human peroxisome proliferator–activated receptor α (PPARα) supports the induction of peroxisome proliferation in PPARα–deficient mouse liver. J. Biol. Chem. 276, 42485–42491 (2001).
Tachibana, K. et al. Gene expression profiling of potential peroxisome proliferator–activated receptor (PPAR) target genes in human hepatoblastoma cell lines inducibly expressing different PPAR isoforms. Nucl. Recept. 3, 3 (2005).
Lytle, C. et al. The peroxisome proliferator–activated receptor γ ligand rosiglitazone delays the onset of inflammatory bowel disease in mice with interleukin 10 deficiency. Inflamm. Bowel Dis. 11, 231–243 (2005).
Madison, B.B. et al. Cis elements of the villin gene control expression in restricted domains of the vertical (crypt) and horizontal (duodenum, cecum) axes of the intestine. J. Biol. Chem. 277, 33275–33283 (2002).
Adachi, M. et al. Peroxisome proliferator activated receptor γ in colonic epithelial cells protects against experimental inflammatory bowel disease. Gut 55, 1104–1113 (2006).
Hamosh, A., Rosenstein, B.J. & Cutting, G.R. CFTR nonsense mutations G542X and W1282X associated with severe reduction of CFTR mRNA in nasal epithelial cells. Hum. Mol. Genet. 1, 542–544 (1992).
Zeitlin, P.L. et al. A cystic fibrosis bronchial epithelial cell line: immortalization by adeno-12–SV40 infection. Am. J. Respir. Cell Mol. Biol. 4, 313–319 (1991).
Leppilampi, M. et al. Carbonic anhydrase isozyme-II–deficient mice lack the duodenal bicarbonate secretory response to prostaglandin E2 . Proc. Natl. Acad. Sci. USA 102, 15247–15252 (2005).
McMurtrie, H.L. et al. The bicarbonate transport metabolon. J. Enzyme Inhib. Med. Chem. 19, 231–236 (2004).
Sellers, Z.M. et al. Heat-stable enterotoxin of Escherichia coli stimulates a non–CFTR-mediated duodenal bicarbonate secretory pathway. Am. J. Physiol. Gastrointest. Liver Physiol. 288, G654–G663 (2005).
Rodríguez, J.C., Gil-Gomez, G., Hegardt, F.G. & Haro, D. Peroxisome proliferator–activated receptor mediates induction of the mitochondrial 3-hydroxy-3-methylglutaryl-CoA synthase gene by fatty acids. J. Biol. Chem. 269, 18767–18772 (1994).
Mandard, S. et al. The direct peroxisome proliferator–activated receptor target fasting-induced adipose factor (FIAF/PGAR/ANGPTL4) is present in blood plasma as a truncated protein that is increased by fenofibrate treatment. J. Biol. Chem. 279, 34411–34420 (2004).
Ge, K. et al. Transcription coactivator TRAP220 is required for PPARγ2–stimulated adipogenesis. Nature 417, 563–567 (2002).
Yuan, C.X., Ito, M., Fondell, J.D., Fu, Z.Y. & Roeder, R.G. The TRAP220 component of a thyroid hormone receptor- associated protein (TRAP) coactivator complex interacts directly with nuclear receptors in a ligand-dependent fashion. Proc. Natl. Acad. Sci. USA 95, 7939–7944 (1998).
Wigren, J. et al. Differential recruitment of the coactivator proteins CREB-binding protein and steroid receptor coactivator-1 to peroxisome proliferator–activated receptor γ/9-cis-retinoic acid receptor heterodimers by ligands present in oxidized low-density lipoprotein. J. Endocrinol. 177, 207–214 (2003).
Chou, W.L. et al. Identification of a novel prostaglandin reductase reveals the involvement of prostaglandin E2 catabolism in regulation of peroxisome proliferator-activated receptor γ activation. J. Biol. Chem. 282, 18162–18172 (2007).
Perez, A. et al. Peroxisome proliferator-activated receptor-γ in cystic fibrosis lung epithelium. Am. J. Physiol. Lung Cell. Mol. Physiol. 295, L303–L313 (2008).
Lucidi, V., Ciabattoni, G., Bella, S., Barnes, P.J. & Montuschi, P. Exhaled 8-isoprostane and prostaglandin E2 in patients with stable and unstable cystic fibrosis. Free Radic. Biol. Med. 45, 913–919 (2008).
Su, C.G. et al. A novel therapy for colitis utilizing PPAR-γ ligands to inhibit the epithelial inflammatory response. J. Clin. Invest. 104, 383–389 (1999).
Lewis, J.D. et al. An open-label trial of the PPAR-γ ligand rosiglitazone for active ulcerative colitis. Am. J. Gastroenterol. 96, 3323–3328 (2001).
Matsui, H. et al. Evidence for periciliary liquid layer depletion, not abnormal ion composition, in the pathogenesis of cystic fibrosis airways disease. Cell 95, 1005–1015 (1998).
Garcia, M.A., Yang, N. & Quinton, P.M. Normal mouse intestinal mucus release requires cystic fibrosis transmembrane regulator–dependent bicarbonate secretion. J. Clin. Invest. 119, 2613–2622 (2009).
Quinton, P.M. Cystic fibrosis: impaired bicarbonate secretion and mucoviscidosis. Lancet 372, 415–417 (2008).
Nichols, D.P., Konstan, M.W. & Chmiel, J.F. Anti-inflammatory therapies for cystic fibrosis–related lung disease. Clin. Rev. Allergy Immunol. 35, 135–153 (2008).
Konstan, M.W. et al. Effect of ibuprofen on neutrophil migration in vivo in cystic fibrosis and healthy subjects. J. Pharmacol. Exp. Ther. 306, 1086–1091 (2003).
Akiyama, T.E. et al. Conditional disruption of the peroxisome proliferator-activated receptor γ gene in mice results in lowered expression of ABCA1, ABCG1 and apoE in macrophages and reduced cholesterol efflux. Mol. Cell. Biol. 22, 2607–2619 (2002).
Rogler, G. et al. Establishment of long-term primary cultures of human small and large intestinal epithelial cells. Lab. Invest. 78, 889–890 (1998).
Ogawa, S. et al. A nuclear receptor corepressor transcriptional checkpoint controlling activator protein 1–dependent gene networks required for macrophage activation. Proc. Natl. Acad. Sci. USA 101, 14461–14466 (2004).
Blaho, V.A., Buczynski, M.W., Brown, C.R. & Dennis, E.A. Lipidomic analysis of dynamic eicosanoid responses during the induction and resolution of Lyme arthritis. J. Biol. Chem. 284, 21599–21612 (2009).
Zarini, S., Gijon, M.A., Folco, G. & Murphy, R.C. Effect of arachidonic acid reacylation on leukotriene biosynthesis in human neutrophils stimulated with granulocyte-macrophage colony–stimulating factor and formyl-methionyl-leucyl-phenylalanine. J. Biol. Chem. 281, 10134–10142 (2006).
Quehenberger, O., Armando, A., Dumlao, D., Stephens, D.L. & Dennis, E.A. Lipidomics analysis of essential fatty acids in macrophages. Prostaglandins Leukot. Essent. Fatty Acids 79, 123–129 (2008).
Acknowledgements
We thank P. Quinton for advice and critical reading of the manuscript. We thank the late J. Isenberg (University of California–San Diego) for providing Cftrtm1Unc mice, R. Sasik for assistance with microarray data analysis and D. McCole for assistance with chloride transport studies. Microarray analysis was performed at the Biogem Core Facility of the University of California–San Diego, and histopathology was performed by the University of California–San Diego Histopathology Core Facility. These studies were supported by US National Institutes of Health grants P01DK074868, GM 069338-03 and DK063491 to C.K.G. and E.A.D.; US National Institutes of Health grant DK007202 and Fellowship to Faculty Transition Award from the Foundation for Digestive Health and Nutrition to G.S.H.
Author information
Authors and Affiliations
Contributions
G.S.H. wrote the manuscript and conducted the breeding, survival, histology, chloride ion transport, gene expression and ChIP experiments. D.S.D. performed the lipidomic analysis by mass spectrometry. D.T.N. performed western blot and luciferase assays. H.D. conducted the bicarbonate ion transport experiments. K.E.B. and E.A.D. contributed to experimental design and data analysis and edited the manuscript. C.K.G. supervised the project, analyzed data and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Tables 1 and 2 and Supplementary Methods (PDF 2061 kb)
Rights and permissions
About this article
Cite this article
Harmon, G., Dumlao, D., Ng, D. et al. Pharmacological correction of a defect in PPAR-γ signaling ameliorates disease severity in Cftr-deficient mice. Nat Med 16, 313–318 (2010). https://doi.org/10.1038/nm.2101
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2101
This article is cited by
-
The role of essential fatty acids in cystic fibrosis and normalizing effect of fenretinide
Cellular and Molecular Life Sciences (2020)
-
Fenretinide differentially modulates the levels of long- and very long-chain ceramides by downregulating Cers5 enzyme: evidence from bench to bedside
Journal of Molecular Medicine (2017)
-
Intestinal PPARγ signalling is required for sympathetic nervous system activation in response to caloric restriction
Scientific Reports (2016)
-
Expression-based GWAS identifies variants, gene interactions and key regulators affecting intramuscular fatty acid content and composition in porcine meat
Scientific Reports (2016)
-
Eicosanoid storm in infection and inflammation
Nature Reviews Immunology (2015)