Abstract
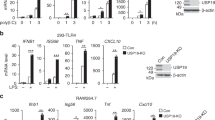
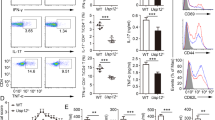
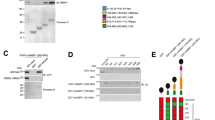
Interleukin 17 (IL-17) is important in infection and autoimmunity; how it signals remains poorly understood. In this study, we identified the ubiquitin-specific protease USP25 as a negative regulator of IL-17-mediated signaling and inflammation. Overexpression of USP25 inhibited IL-17-triggered signaling, whereas USP25 deficiency resulted in more phosphorylation of the inhibitor IκBα and kinase Jnk and higher expression of chemokines and cytokines, as well as a prolonged half-life for chemokine CXCL1–encoding mRNA after treatment with IL-17. Consistent with that, Usp25−/− mice showed greater sensitivity to IL-17-dependent inflammation and autoimmunity in vivo. Mechanistically, stimulation with IL-17 induced the association of USP25 with the adaptors TRAF5 and TRAF6, and USP25 induced removal of Lys63-linked ubiquitination in TRAF5 and TRAF6 mediated by the adaptor Act1. Thus, our results demonstrate that USP25 is a deubiquitinating enzyme (DUB) that negatively regulates IL-17-triggered signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Iwakura, Y., Ishigame, H., Saijo, S. & Nakae, S. Functional specialization of interleukin-17 family members. Immunity 34, 149–162 (2011).
Yang, X.O. et al. Regulation of inflammatory responses by IL-17F. J. Exp. Med. 205, 1063–1075 (2008).
Kagami, S., Rizzo, H.L., Kurtz, S.E., Miller, L.S. & Blauvelt, A. IL-23 and IL-17A, but not IL-12 and IL-22, are required for optimal skin host defense against Candida albicans. J. Immunol. 185, 5453–5462 (2010).
Gaffen, S.L. Recent advances in the IL-17 cytokine family. Curr. Opin. Immunol. 23, 613–619 (2011).
Park, H. et al. A distinct lineage of CD4 T cells regulates tissue inflammation by producing interleukin 17. Nat. Immunol. 6, 1133–1141 (2005).
Dong, C. Diversification of T-helper-cell lineages: finding the family root of IL-17-producing cells. Nat. Rev. Immunol. 6, 329–333 (2006).
Harrington, L.E. et al. Interleukin 17-producing CD4+ effector T cells develop via a lineage distinct from the T helper type 1 and 2 lineages. Nat. Immunol. 6, 1123–1132 (2005).
Cua, D.J. & Tato, C.M. Innate IL-17-producing cells: the sentinels of the immune system. Nat. Rev. Immunol. 10, 479–489 (2010).
Chang, S.H. & Dong, C. Signaling of interleukin-17 family cytokines in immunity and inflammation. Cell. Signal. 23, 1069–1075 (2011).
Hartupee, J., Liu, C., Novotny, M., Li, X. & Hamilton, T. IL-17 enhances chemokine gene expression through mRNA stabilization. J. Immunol. 179, 4135–4141 (2007).
Wright, J.F. et al. The human IL-17F/IL-17A heterodimeric cytokine signals through the IL-17RA/IL-17RC receptor complex. J. Immunol. 181, 2799–2805 (2008).
Hu, Y. et al. IL-17RC is required for IL-17A- and IL-17F-dependent signaling and the pathogenesis of experimental autoimmune encephalomyelitis. J. Immunol. 184, 4307–4316 (2010).
Chang, S.H., Park, H. & Dong, C. Act1 adaptor protein is an immediate and essential signaling component of interleukin-17 receptor. J. Biol. Chem. 281, 35603–35607 (2006).
Qian, Y. et al. The adaptor Act1 is required for interleukin 17-dependent signaling associated with autoimmune and inflammatory disease. Nat. Immunol. 8, 247–256 (2007).
Liu, C. et al. Act1, a U-box E3 ubiquitin ligase for IL-17 signaling. Sci. Signal. 2, ra63 (2009).
Bulek, K. et al. The inducible kinase IKKi is required for IL-17-dependent signaling associated with neutrophilia and pulmonary inflammation. Nat. Immunol. 12, 844–852 (2011).
Reyes-Turcu, F.E., Ventii, K.H. & Wilkinson, K.D. Regulation and cellular roles of ubiquitin-specific deubiquitinating enzymes. Annu. Rev. Biochem. 78, 363–397 (2009).
Lin, S.C. et al. Molecular basis for the unique deubiquitinating activity of the NF-κB inhibitor A20. J. Mol. Biol. 376, 526–540 (2008).
Li, S. et al. Regulation of virus-triggered signaling by OTUB1- and OTUB2-mediated deubiquitination of TRAF3 and TRAF6. J. Biol. Chem. 285, 4291–4297 (2010).
Valero, R. et al. USP25, a novel gene encoding a deubiquitinating enzyme, is located in the gene-poor region 21q11.2. Genomics 62, 395–405 (1999).
Chang, S.H. & Dong, C. IL-17F: regulation, signaling and function in inflammation. Cytokine 46, 7–11 (2009).
Wang, X. et al. Transcription of Il17 and Il17f is controlled by conserved noncoding sequence 2. Immunity 36, 23–31 (2012).
Chang, S.H. et al. Interleukin-17C promotes Th17 cell responses and autoimmune disease via interleukin-17 receptor E. Immunity 35, 611–621 (2011).
Denuc, A., Bosch-Comas, A., Gonzalez-Duarte, R. & Marfany, G. The UBA-UIM domains of the USP25 regulate the enzyme ubiquitination state and modulate substrate recognition. PLoS ONE 4, e5571 (2009).
Sun, D. et al. Treatment with IL-17 prolongs the half-life of chemokine CXCL1 mRNA via the adaptor TRAF5 and the splicing-regulatory factor SF2 (ASF). Nat. Immunol. 12, 853–860 (2011).
May, M.J. IL-17R signaling: new players get in on the Act1. Nat. Immunol. 12, 813–815 (2011).
Lamothe, B. et al. Site-specific Lys-63-linked tumor necrosis factor receptor-associated factor 6 auto-ubiquitination is a critical determinant of IκB kinase activation. J. Biol. Chem. 282, 4102–4112 (2007).
Shi, P. et al. Persistent stimulation with interleukin-17 desensitizes cells through SCFβ-TrCP-mediated degradation of Act1. Sci. Signal. 4, ra73 (2011).
Skaug, B., Jiang, X. & Chen, Z.J. The role of ubiquitin in NF-κB regulatory pathways. Annu. Rev. Biochem. 78, 769–796 (2009).
Boone, D.L. et al. The ubiquitin-modifying enzyme A20 is required for termination of Toll-like receptor responses. Nat. Immunol. 5, 1052–1060 (2004).
Ea, C.K., Sun, L., Inoue, J. & Chen, Z.J. TIFA activates IκB kinase (IKK) by promoting oligomerization and ubiquitination of TRAF6. Proc. Natl. Acad. Sci. USA 101, 15318–15323 (2004).
Mao, A.P. et al. Virus-triggered ubiquitination of TRAF3/6 by cIAP1/2 is essential for induction of interferon-β (IFN-β) and cellular antiviral response. J. Biol. Chem. 285, 9470–9476 (2010).
Deng, L. et al. Activation of the IκB kinase complex by TRAF6 requires a dimeric ubiquitin-conjugating enzyme complex and a unique polyubiquitin chain. Cell 103, 351–361 (2000).
Wang, C. et al. TAK1 is a ubiquitin-dependent kinase of MKK and IKK. Nature 412, 346–351 (2001).
Kanayama, A. et al. TAB2 and TAB3 activate the NF-κB pathway through binding to polyubiquitin chains. Mol. Cell 15, 535–548 (2004).
Xia, Z.P. et al. Direct activation of protein kinases by unanchored polyubiquitin chains. Nature 461, 114–119 (2009).
Fan, Y. et al. Lysine 63-linked polyubiquitination of TAK1 at lysine 158 is required for tumor necrosis factor α- and interleukin-1β-induced IKK/NF-κB and JNK/AP-1 activation. J. Biol. Chem. 285, 5347–5360 (2010).
Xu, M., Skaug, B., Zeng, W. & Chen, Z.J. A ubiquitin replacement strategy in human cells reveals distinct mechanisms of IKK activation by TNFα and IL-1β. Mol. Cell 36, 302–314 (2009).
Zeng, W. et al. Reconstitution of the RIG-I pathway reveals a signaling role of unanchored polyubiquitin chains in innate immunity. Cell 141, 315–330 (2010).
Gack, M.U. et al. TRIM25 RING-finger E3 ubiquitin ligase is essential for RIG-I-mediated antiviral activity. Nature 446, 916–920 (2007).
Zhu, S. et al. Modulation of experimental autoimmune encephalomyelitis through TRAF3-mediated suppression of interleukin 17 receptor signaling. J. Exp. Med. 207, 2647–2662 (2010).
He, J.Q., Oganesyan, G., Saha, S.K., Zarnegar, B. & Cheng, G. TRAF3 and its biological function. Adv. Exp. Med. Biol. 597, 48–59 (2007).
Maezawa, Y. et al. Involvement of TNF receptor-associated factor 6 in IL-25 receptor signaling. J. Immunol. 176, 1013–1018 (2006).
Angkasekwinai, P. et al. Interleukin 25 promotes the initiation of proallergic type 2 responses. J. Exp. Med. 204, 1509–1517 (2007).
Bot, J. et al. Culturing mouse peritoneal mesothelial cells. Pathol. Res. Pract. 199, 341–344 (2003).
Wang, J. et al. Pneumocystis carinii activates the NF-κB signaling pathway in alveolar epithelial cells. Infect. Immun. 73, 2766–2777 (2005).
Bosch-Comas, A., Lindsten, K., Gonzalez-Duarte, R., Masucci, M.G. & Marfany, G. The ubiquitin-specific protease USP25 interacts with three sarcomeric proteins. Cell. Mol. Life Sci. 63, 723–734 (2006).
Zhong, B. et al. The ubiquitin ligase RNF5 regulates antiviral responses by mediating degradation of the adaptor protein MITA. Immunity 30, 397–407 (2009).
Zhong, B. et al. The E3 ubiquitin ligase RNF5 targets virus-induced signaling adaptor for ubiquitination and degradation. J. Immunol. 184, 6249–6255 (2010).
Li, Q. et al. Tripartite motif 8 (TRIM8) modulates TNFα- and IL-1β-triggered NF-κB activation by targeting TAK1 for K63-linked polyubiquitination. Proc. Natl. Acad. Sci. USA 108, 19341–19346 (2011).
Acknowledgements
We thank G. Marfany (University of Barcelona) for rabbit anti-USP25; T. Hamilton (Cleveland Clinic) for Tet-Off HeLa cells and pTRE2-KCΔ4 plasmid; Z. Chen (UT Southwestern Medical Center) for hemagglutinin-tagged ubiquitin (K48 only and K63 only) and suggestions; H.-B. Shu (Wuhan University) for mouse anti-TRAF6, plasmids encoding TRAF1–TRAF6 and suggestions; and members of Dong laboratory for technical help. Supported by the US National Institutes of Health, the University of Texas MD Anderson Cancer Center, the CFP foundation of MD Anderson Cancer Center (B.Z.) and the Leukemia and Lymphoma Society (C.D.).
Author information
Authors and Affiliations
Contributions
B.Z., Xik.L. and C.D. designed the project; Xik.L. did the IL-17-induced peritoneal inflammation experiments; B.Z., X.W., Xin.L. and A.W. did the biochemical experiments and EAE analysis; B.Z. and S.H.C. prepared MEFs and did the IL-17 induced lung-inflammation experiments; B.Z. and J.M.R. prepared lung epithelial cells; and B.Z. and C.D. wrote and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 and Supplementary Table 1 (PDF 3602 kb)
Rights and permissions
About this article
Cite this article
Zhong, B., Liu, X., Wang, X. et al. Negative regulation of IL-17-mediated signaling and inflammation by the ubiquitin-specific protease USP25. Nat Immunol 13, 1110–1117 (2012). https://doi.org/10.1038/ni.2427
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2427
This article is cited by
-
A New Modulator of Neuroinflammation in Diabetic Retinopathy: USP25
Inflammation (2024)
-
Ubiquitin-specific peptidase 25 exacerbated osteoarthritis progression through facilitating TXNIP ubiquitination and NLRP3 inflammasome activation
Journal of Orthopaedic Surgery and Research (2023)
-
Ubiquitin-specific protease 28: the decipherment of its dual roles in cancer development
Experimental Hematology & Oncology (2023)
-
Interleukin-17 as a key player in neuroimmunometabolism
Nature Metabolism (2023)
-
The IL-17 family in diseases: from bench to bedside
Signal Transduction and Targeted Therapy (2023)