Abstract
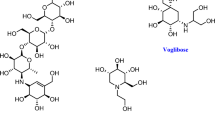
The complex plant flavonol glycoside montbretin A is a potent (Ki = 8 nM) and specific inhibitor of human pancreatic α-amylase with potential as a therapeutic for diabetes and obesity. Controlled degradation studies on montbretin A, coupled with inhibition analyses, identified an essential high-affinity core structure comprising the myricetin and caffeic acid moieties linked via a disaccharide. X-ray structural analyses of the montbretin A–human α-amylase complex confirmed the importance of this core structure and revealed a novel mode of glycosidase inhibition wherein internal π-stacking interactions between the myricetin and caffeic acid organize their ring hydroxyls for optimal hydrogen bonding to the α-amylase catalytic residues D197 and E233. This novel inhibitory motif can be reproduced in a greatly simplified analog, offering potential for new strategies for glycosidase inhibition and therapeutic development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
World Health Organization. WHO Fact Sheet 312: Diabetes (WHO, 2013) http://www.who.int/mediacentre/factsheets/fs312/en/.
Chiasson, J.L. et al. The efficacy of acarbose in the treatment of patients with non-insulin-dependent diabetes mellitus—a multicenter controlled clinical trial. Ann. Intern. Med. 121, 928–935 (1994).
Mooradian, A.D. & Thurman, J.E. Drug therapy of postprandial hyperglycaemia. Drugs 57, 19–29 (1999).
Scott, L.J. & Spencer, C.M. Miglitol—A review of its therapeutic potential in type 2 diabetes mellitus. Drugs 59, 521–549 (2000).
Brayer, G.D., Luo, Y.G. & Withers, S.G. The structure of human pancreatic alpha-amylase at 1.8 Angstrom resolution and comparisons with related enzymes. Protein Sci. 4, 1730–1742 (1995).
Li, C.M. et al. Acarbose rearrangement mechanism implied by the kinetic and structural analysis of human pancreatic alpha-amylase in complex with analogues and their elongated counterparts. Biochemistry 44, 3347–3357 (2005).
Tarling, C.A. et al. The search for novel human pancreatic alpha-amylase inhibitors: high-throughput screening of terrestrial and marine natural product extracts. ChemBioChem 9, 433–438 (2008).
Asada, Y., Hirayama, Y. & Furuya, T. Acylated flavonols from Crocosmia crocosmiiflora. Phytochemistry 27, 1497–1501 (1988).
Lo Piparo, E. et al. Flavonoids for controlling starch digestion: structural requirements for inhibiting human alpha-amylase. J. Med. Chem. 51, 3555–3561 (2008).
Ong, K.C. & Khoo, H.E. Effects of myricetin on glycemia and glycogen metabolism in diabetic rats. Life Sci. 67, 1695–1705 (2000).
Yoshida, K., Hishida, A., Iida, O., Hosokawa, K. & Kawabata, J. Flavonol caffeoylglycosides as alpha-glucosidase inhibitors from Spiraea cantoniensis flower. J. Agric. Food Chem. 56, 4367–4371 (2008).
Manaharan, T., Appleton, D., Cheng, H.M. & Palanisamy, U.D. Flavonoids isolated from Syzygium aqueum leaf extract as potential antihyperglycaemic agents. Food Chem. 132, 1802–1807 (2012).
Williams, L.K., Li, C.M., Withers, S.G. & Brayer, G.D. Order and disorder: differential structural impacts of myricetin and ethyl caffeate on human amylase, an antidiabetic target. J. Med. Chem. 55, 10177–10186 (2012).
Brayer, G.D. et al. Subsite mapping of the human pancreatic alpha-amylase active site through structural, kinetic, and mutagenesis techniques. Biochemistry 39, 4778–4791 (2000).
Sim, L., Quezada-Calvillo, R., Sterchi, E.E., Nichols, B.L. & Rose, D.R. Human intestinal maltase-glucoamylase: crystal structure of the N-terminal catalytic subunit and basis of inhibition and substrate specificity. J. Mol. Biol. 375, 782–792 (2008).
Sim, L. et al. Structural basis for substrate selectivity in human maltase-glucoamylase and sucrase-isomaltase N-terminal domains. J. Biol. Chem. 285, 17763–17770 (2010).
McCarter, J.D. & Withers, S.G. Mechanisms of enzymatic glycoside hydrolysis. Curr. Opin. Struct. Biol. 4, 885–892 (1994).
Mhlongo, N.N., Skelton, A.A., Kruger, G., Soliman, M.E.S. & Williams, I.H. A critical survey of average distances between catalytic carboxyl groups in glycoside hydrolases. Proteins 82, 1747–1755 (2014).
Wang, Q.P., Graham, R.W., Trimbur, D., Warren, R.A.J. & Withers, S.G. Changing enzymatic reaction mechanisms by mutagenesis- conversion of a retaining glucosidase to an inverting enzyme. J. Am. Chem. Soc. 116, 11594–11595 (1994).
Rydberg, E.H. et al. Mechanistic analyses of catalysis in human pancreatic alpha-amylase: detailed kinetic and structural studies of mutants of three conserved carboxylic acids. Biochemistry 41, 4492–4502 (2002).
The PyMOL Molecular Graphics System, version 1.3r1 (Schrodinger LLC, San Diego, California, USA, 2010).
Rydberg, E.H. et al. Cloning, mutagenesis, and structural analysis of human pancreatic alpha-amylase expressed in Pichia pastoris. Protein Sci. 8, 635–643 (1999).
Sambrook, J., Fritsch, E.F. & Maniatis, T. Molecular Cloning: A Laboratory Manual edn. 2 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York, USA, 1989).
Numao, S. et al. Probing the role of the chloride ion in the mechanism of human pancreatic alpha-amylase. Biochemistry 41, 215–225 (2002).
Woods, K.B. Bioactive natural products. PhD Thesis, University of British Columbia (2010).
Kabsch, W. XDS. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Karplus, P.A. & Diederichs, K. Linking crystallographic model and data quality. Science 336, 1030–1033 (2012).
McCoy, A.J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Adams, P.D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004).
Schüttelkopf, A.W. & van Aalten, D.M.F. PRODRG: a tool for high-throughput crystallography of protein-ligand complexes. Acta Crystallogr. D Biol. Crystallogr. 60, 1355–1363 (2004).
Moriarty, N.W., Grosse-Kunstleve, R.W. & Adams, P.D. electronic Ligand Builder and Optimization Workbench (eLBOW): a tool for ligand coordinate and restraint generation. Acta Crystallogr. D Biol. Crystallogr. 65, 1074–1080 (2009).
Acknowledgements
We thank R. Maurus for helpful discussions regarding structural analyses and E. Goddard-Borger for advice on synthesis. We thank E. Kwan for preparation of HPA, T. Hill for isolation of human gut bacterial amylases, C. Hii, P. Lincez and K. Liu for additional technical assistance, H. Flint for providing clones of the human gut bacterial amylases and D.R. Rose (University of Waterloo) for supplies of ntMGAM, ctMGAM and ctSI. This work was supported by Operating and Proof of Principle grants from the Canadian Institutes of Health Research (CIHR; ref. nos. (FRN) 111082, 200704PPP (to G.D.B. and S.G.W.)), by an operating grant from the Natural Sciences and Engineering Research Council (NSERC) of Canada to R.J.A. and through the CDRD-Pfizer Innovation Fund. L.K.W. was supported by an Alexander Graham Bell Canada Graduate Scholarship (NSERC). S.G.W. is supported by a Tier 1 Canada Research Chair. Portions of this research were carried out at the Stanford Synchrotron Radiation Lightsource (SSRL), SLAC National Accelerator Laboratory, which is supported by the US Department of Energy (DOE), Office of Science, Office of Basic Energy Sciences under contract no. DE-AC02-76SF00515. The SSRL Structural Molecular Biology Program is supported by the DOE Office of Biological and Environmental Research and by the National Institutes of Health (NIH), National Institute of General Medical Sciences (NIGMS) (including P41GM103393). The contents of this publication are solely the responsibility of the authors and do not necessarily represent the official views of NIGMS or NIH. The graphics program PyMOL (ref. 21) was used in part to generate figures.
Author information
Authors and Affiliations
Contributions
N.T.N. crystallized the MbA–HPA complex, and L.K.W. and S.C. solved and analyzed its structure; X.Z. performed the degradation studies of MbA and kinetic studies thereon; C.T. synthesized inhibitor 10 and performed kinetic studies thereon; J.W. performed kinetic studies on MbA variants; D.E.W. performed the ROESY NMR studies on MbA; J.C., V.Y., J.M., R.J.A., S.G.W. and G.D.B. designed and supervised the project. Writing was primarily done by V.Y., G.D.B., L.K.W. and S.G.W. with contributions from all.
Corresponding author
Ethics declarations
Competing interests
The University of British Columbia holds a patent on the use of MbA as a blood glucose controlling agent with R.J.A., G.D.B. and S.G.W. as inventors.
Supplementary information
Supplementary Text and Figures
Supplementary Results, Supplementary Tables 1–3, Supplementary Figures 1–5 and Supplementary Notes 1 and 2. (PDF 1922 kb)
Rights and permissions
About this article
Cite this article
Williams, L., Zhang, X., Caner, S. et al. The amylase inhibitor montbretin A reveals a new glycosidase inhibition motif. Nat Chem Biol 11, 691–696 (2015). https://doi.org/10.1038/nchembio.1865
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.1865
This article is cited by
-
Flavonoids as dual-target inhibitors against α-glucosidase and α-amylase: a systematic review of in vitro studies
Natural Products and Bioprospecting (2024)
-
Design, synthesis, molecular docking study and molecular dynamics simulation of new coumarin-pyrimidine hybrid compounds having anticancer and antidiabetic activity
Medicinal Chemistry Research (2023)
-
Characterizations of Zinc Acetate micro-crystals (ZAμ) and Co-60 irradiated micro-crystals (GZAμ) for photonic and electro-optic relevances
Journal of Materials Science: Materials in Electronics (2023)
-
Synthesis, Characterizations of Macro, Micro, Irradiated Crystals of KDP, the Standard Non-linear Optical Reference Material for Mechano, Photonic, Electronic Uses
Chemistry Africa (2023)
-
Molecular cloning and characterization of an alpha-amylase inhibitor (TkAAI) gene from Trichosanthes kirilowii Maxim.
Biotechnology Letters (2022)