Abstract
Objective:
Reduction of melanocortin signaling in the brain results in obesity. However, where in the brain reduced melanocortin signaling mediates this effect is poorly understood.
Design:
We determined the effects of long-term inhibition of melanocortin receptor activity in specific brain regions of the rat brain. Melanocortin signaling was inhibited by injection of a recombinant adeno-associated viral (rAAV) vector that overexpressed Agouti-related peptide (AgRP) into the paraventricular nucleus (PVN), the ventromedial hypothalamus (VMH), the lateral hypothalamus (LH) or the accumbens shell (Acc).
Results:
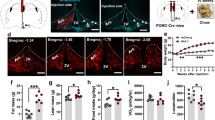
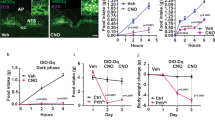
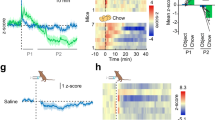
Overexpression of AgRP in the rat PVN, VMH or LH increased bodyweight, the percentage of white adipose tissue, plasma leptin and insulin concentrations and food intake. Food intake was mainly increased because of an increase in meal size in the light and dark phases, after overexpression of AgRP in the PVN, LH or VMH. Overexpression of AgRP in the PVN or VMH reduced average body core temperature in the dark on day 40 post injection, whereas AgRP overexpression in the LH did not affect temperature. In addition, overexpression of AgRP in the PVN, LH or VMH did not significantly alter mRNA expression of AgRP, neuropeptide Y (NPY), pro-opiomelanocortin (POMC) or suppressor of cytokine signaling 3 (SOCS3) in the arcuate. Overexpression of AgRP in the Acc did not have any effect on the measured parameters.
Conclusions:
Reduction of melanocortin signaling in several hypothalamic regions increased meal size. However, there were brain area-specific effects on other parameters such as core temperature and plasma leptin concentrations. In a previous study, where NPY was overexpressed with an rAAV vector in the PVN and LH, meal frequency and meal size were increased respectively, whereas locomotor activity was reduced by NPY overexpression at both nuclei. Taken together, AgRP and NPY have complementary roles in energy balance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Nijenhuis WA, Oosterom J, Adan RA . AgRP(83–132) acts as an inverse agonist on the human-melanocortin-4 receptor. Mol Endocrinol 2001; 15: 164–171.
Haskell-Luevano C, Monck EK . Agouti-related protein functions as an inverse agonist at a constitutively active brain melanocortin-4 receptor. Regul Pept 2001; 99: 1–7.
Graham M, Shutter JR, Sarmiento U, Sarosi I, Stark KL . Overexpression of Agrt leads to obesity in transgenic mice. Nat Genet 1997; 17: 273–274.
Small CJ, Liu YL, Stanley SA, Connoley IP, Kennedy A, Stock MJ et al. Chronic CNS administration of Agouti-related protein (Agrp) reduces energy expenditure. Int J Obes Relat Metab Disord 2003; 27: 530–533.
Semjonous NM, Smith KL, Parkinson JR, Gunner DJ, Liu YL, Murphy KG et al. Coordinated changes in energy intake and expenditure following hypothalamic administration of neuropeptides involved in energy balance. Int J Obes (Lond) 2009; 33: 775–785.
Ollmann MM, Wilson BD, Yang YK, Kerns JA, Chen Y, Gantz I et al. Antagonism of central melanocortin receptors in vitro and in vivo by agouti-related protein. Science 1997; 278: 135–138.
Klebig ML, Wilkinson JE, Geisler JG, Woychik RP . Ectopic expression of the agouti gene in transgenic mice causes obesity, features of type II diabetes, and yellow fur. Proc Natl Acad Sci USA 1995; 92: 4728–4732.
Kas MJ, Tiesjema B, van Dijk G, Garner KM, Barsh GS, ter Brake O et al. Induction of brain-region-specific forms of obesity by agouti. J Neurosci 2004; 24: 10176–10181.
Huszar D, Lynch CA, Fairchild-Huntress V, Dunmore JH, Fang Q, Berkemeier LR et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 1997; 88: 131–141.
Chen AS, Metzger JM, Trumbauer ME, Guan XM, Yu H, Frazier EG et al. Role of the melanocortin-4 receptor in metabolic rate and food intake in mice. Transgenic Res 2000; 9: 145–154.
Ste ML, Miura GI, Marsh DJ, Yagaloff K, Palmiter RD . A metabolic defect promotes obesity in mice lacking melanocortin-4 receptors. Proc Natl Acad Sci USA 2000; 97: 12339–12344.
Butler AA, Marks DL, Fan W, Kuhn CM, Bartolome M, Cone RD . Melanocortin-4 receptor is required for acute homeostatic responses to increased dietary fat. Nat Neurosci 2001; 4: 605–611.
Chen AS, Marsh DJ, Trumbauer ME, Frazier EG, Guan XM, Yu H et al. Inactivation of the mouse melanocortin-3 receptor results in increased fat mass and reduced lean body mass. Nat Genet 2000; 26: 97–102.
Butler AA, Kesterson RA, Khong K, Cullen MJ, Pelleymounter MA, Dekoning J et al. A unique metabolic syndrome causes obesity in the melanocortin-3 receptor-deficient mouse. Endocrinology 2000; 141: 3518–3521.
Kishi T, Aschkenasi CJ, Lee CE, Mountjoy KG, Saper CB, Elmquist JK . Expression of melanocortin 4 receptor mRNA in the central nervous system of the rat. J Comp Neurol 2003; 457: 213–235.
Liu H, Kishi T, Roseberry AG, Cai X, Lee CE, Montez JM et al. Transgenic mice expressing green fluorescent protein under the control of the melanocortin-4 receptor promoter. J Neurosci 2003; 23: 7143–7154.
Roselli-Rehfuss L, Mountjoy KG, Robbins LS, Mortrud MT, Low MJ, Tatro JB et al. Identification of a receptor for gamma melanotropin and other proopiomelanocortin peptides in the hypothalamus and limbic system. Proc Natl Acad Sci USA 1993; 90: 8856–8860.
Kim MS, Rossi M, Abusnana S, Sunter D, Morgan DG, Small CJ et al. Hypothalamic localization of the feeding effect of agouti-related peptide and alpha-melanocyte-stimulating hormone. Diabetes 2000; 49: 177–182.
Shrestha YB, Wickwire K, Giraudo SQ . Role of AgRP on Ghrelin-induced feeding in the hypothalamic paraventricular nucleus. Regul Pept 2006; 133: 68–73.
Wirth MM, Giraudo SQ . Agouti-related protein in the hypothalamic paraventricular nucleus: effect on feeding. Peptides 2000; 21: 1369–1375.
Berthoud HR . Multiple neural systems controlling food intake and body weight. Neurosci Biobehav Rev 2002; 26: 393–428.
Tiesjema B, Adan RA, Luijendijk MC, Kalsbeek A, la Fleur SE . Differential effects of recombinant adeno-associated virus-mediated neuropeptide Y overexpression in the hypothalamic paraventricular nucleus and lateral hypothalamus on feeding behavior. J Neurosci 2007; 27: 14139–14146.
Tiesjema B, la Fleur SE, Luijendijk MC, Brans MA, Lin EJ, During MJ et al. Viral mediated neuropeptide Y expression in the rat paraventricular nucleus results in obesity. Obesity (Silver Spring) 2007; 15: 2424–2435.
Broekman MLD, Comer LA, Hyman BT, Siena-Esteves M . Adeno-associated virus vectors serotyped with AAV8 capsid are more efficient than AAV-1 or-2 serotypes for widespread gene delivery to the neonatal mouse brain. Neuroscience 2006; 138: 501–510.
Grimm D, Kay MA, Kleinschmidt JA . Helper virus-free, optically controllable, and two-plasmid-based production of adeno-associated virus vectors of serotypes 1 to 6. Mol Ther 2003; 7: 839–850.
de Backer M, Brans M, Luijendijk M, Garner K, Adan R . Optimization of adeno-associated viral vector mediated gene delivery to the hypothalamus. Hum Gene Ther 2010; 21: 673–682.
Schaeren-Wiemers N, Gerfin-Moser A . A single protocol to detect transcripts of various types and expression levels in neural tissue and cultured cells: in situ hybridization using digoxigenin-labelled cRNA probes. Histochemistry 1993; 100: 431–440.
Kas MJH, van Dijk G, Scheurink AJW, Adan RAH . Agouti-related protein prevents self-starvation. Mol Psychiatry 2003; 8: 235–240.
la Fleur SE, van Rozen AJ, Luijendijk MC, Groeneweg F, Adan RA . A free-choice high-fat high-sugar diet induces changes in arcuate neuropeptide expression that support hyperphagia. Int J Obes (Lond) 2009; 34: 537–546.
Fisher KJ, Gao GP, Weitzman MD, DeMatteo R, Burda JF, Wilson JM . Transduction with recombinant adeno-associated virus for gene therapy is limited by leading-strand synthesis. J Virol 1996; 70: 520–532.
Ferrari FK, Samulski T, Shenk T, Samulski RJ . Second-strand synthesis is a rate-limiting step for efficient transduction by recombinant adeno-associated virus vectors. J Virol 1996; 70: 3227–3234.
Reimsnider S, Manfredsson FP, Muzyczka N, Mandel RJ . Time course of transgene expression after intrastriatal pseudotyped rAAV2/1, rAAV2/2, rAAV2/5, and rAAV2/8 transduction in the rat. Mol Ther 2007; 15: 1504–1511.
Balthasar N, Dalgaard LT, Lee CE, Yu J, Funahashi H, Williams T et al. Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell 2005; 123: 493–505.
Kask A, Schioth HB . Tonic inhibition of food intake during inactive phase is reversed by the injection of the melanocortin receptor antagonist into the paraventricular nucleus of the hypothalamus and central amygdala of the rat. Brain Res 2000; 887: 460–464.
Woods SC . The control of food intake: behavioral versus molecular perspectives. Cell Metab 2009; 9: 489–498.
Erlanson-Albertsson C . Appetite regulation and energy balance. Acta Paediatr 2005; 94: 40–41.
Tang-Christensen M, Vrang N, Ortmann S, Bidlingmaier M, Horvath TL, Tschop M . Central administration of ghrelin and agouti-related protein (83–132) increases food intake and decreases spontaneous locomotor activity in rats. Endocrinology 2004; 145: 4645–4652.
Santollo J, Eckel LA . Estradiol decreases the orexigenic effect of neuropeptide Y, but not agouti-related protein, in ovariectomized rats. Behav Brain Res 2008; 191: 173–177.
Zheng H, Patterson LM, Phifer CB, Berthoud HR . Brain stem melanocortinergic modulation of meal size and identification of hypothalamic POMC projections. Am J Physiol Regul Integr Comp Physiol 2005; 289: R247–R258.
Azzara AV, Sokolnicki JP, Schwartz GJ . Central melanocortin receptor agonist reduces spontaneous and scheduled meal size but does not augment duodenal preload-induced feeding inhibition. Physiol Behav 2002; 77: 411–416.
Williams DL, Grill HJ, Weiss SM, Baird JP, Kaplan JM . Behavioral processes underlying the intake suppressive effects of melanocortin 3/4 receptor activation in the rat. Psychopharmacology (Berl) 2002; 161: 47–53.
Farooqi IS, Keogh JM, Yeo GS, Lank EJ, Cheetham T, O’Rahilly S . Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. N Engl J Med 2003; 348: 1085–1095.
Creemers JW, Pritchard LE, Gyte A, Le Rouzic P, Meulemans S, Wardlaw SL et al. Agouti-related protein is posttranslationally cleaved by proprotein convertase 1 to generate agouti-related protein (AGRP)83-132: interaction between AGRP83-132 and melanocortin receptors cannot be influenced by syndecan-3. Endocrinology 2006; 147: 1621–1631.
Skibicka KP, Grill HJ . Hypothalamic and hindbrain melanocortin receptors contribute to the feeding, thermogenic, and cardiovascular action of melanocortins. Endocrinology 2009; 150: 5351–5361.
Wortley KE, Anderson KD, Yasenchak J, Murphy A, Valenzuela D, Diano S et al. Agouti-related protein-deficient mice display an age-related lean phenotype. Cell Metab 2005; 2: 421–427.
Kreier F, Kap YS, Mettenleiter TC, van Heijningen C, van der Vliet J, Kalsbeek A et al. Tracing from fat tissue, liver, and pancreas: a neuroanatomical framework for the role of the brain in type 2 diabetes. Endocrinology 2006; 147: 1140–1147.
Song CK, Jackson RM, Harris RB, Richard D, Bartness TJ . Melanocortin-4 receptor mRNA is expressed in sympathetic nervous system outflow neurons to white adipose tissue. Am J Physiol Regul Integr Comp Physiol 2005; 289: R1467–R1476.
Song CK, Vaughan CH, Keen-Rhinehart E, Harris RB, Richard D, Bartness TJ . Melanocortin-4 receptor mRNA expressed in sympathetic outflow neurons to brown adipose tissue: neuroanatomical and functional evidence. Am J Physiol Regul Integr Comp Physiol 2008; 295: R417–R428.
Tallam LS, Stec DE, Willis MA, da Silva AA, Hall JE . Melanocortin-4 receptor-deficient mice are not hypertensive or salt-sensitive despite obesity, hyperinsulinemia, and hyperleptinemia. Hypertension 2005; 46: 326–332.
Greenfield JR, Miller JW, Keogh JM, Henning E, Satterwhite JH, Cameron GS et al. Modulation of blood pressure by central melanocortinergic pathways. N Engl J Med 2009; 360: 44–52.
Nogueiras R, Wiedmer P, Perez-Tilve D, Veyrat-Durebex C, Keogh JM, Sutton GM et al. The central melanocortin system directly controls peripheral lipid metabolism. J Clin Invest 2007; 117: 3475–3488.
Arase K, Sakaguchi T, Bray GA . Lateral hypothalamic lesions and activity of the sympathetic nervous system. Life Sci 1987; 41: 657–662.
Yoshida T, Kemnitz JW, Bray GA . Lateral hypothalamic lesions and norepinephrine turnover in rats. J Clin Invest 1983; 72: 919–927.
Sakaguchi T, Arase K, Bray GA . Sympathetic activity and food intake of rats with ventromedial hypothalamic lesions. Int J Obes 1988; 12: 285–291.
Takahashi A, Shimazu T . Hypothalamic regulation of lipid metabolism in the rat: effect of hypothalamic stimulation on lipolysis. J Auton Nerv Syst 1981; 4: 195–205.
Zhong MK, Duan YC, Chen AD, Xu B, Gao XY, De W et al. Paraventricular nucleus is involved in the central pathway of cardiac sympathetic afferent reflex in rats. Exp Physiol 2008; 93: 746–753.
Gettys TW, Harkness PJ, Watson PM . The beta 3-adrenergic receptor inhibits insulin-stimulated leptin secretion from isolated rat adipocytes. Endocrinology 1996; 137: 4054–4057.
Cong L, Chen K, Li J, Gao P, Li Q, Mi S et al. Regulation of adiponectin and leptin secretion and expression by insulin through a PI3K-PDE3B dependent mechanism in rat primary adipocytes. Biochem J 2007; 403: 519–525.
Kishi T, Aschkenasi CJ, Choi BJ, Lopez ME, Lee CE, Liu H et al. Neuropeptide Y Y1 receptor mRNA in rodent brain: distribution and colocalization with melanocortin-4 receptor. J Comp Neurol 2005; 482: 217–243.
Tiesjema B, la Fleur SE, Luijendijk MC, Adan RA . Sustained NPY overexpression in the PVN results in obesity via temporarily increasing food intake. Obesity (Silver Spring) 2009; 17: 1448–1450.
Acknowledgements
This work was performed in Utrecht, the Netherlands, and was supported by the Netherlands Organization for Scientific Research (NWO Grant No. 90339175).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
de Backer, M., la Fleur, S., Brans, M. et al. Melanocortin receptor-mediated effects on obesity are distributed over specific hypothalamic regions. Int J Obes 35, 629–641 (2011). https://doi.org/10.1038/ijo.2010.169
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2010.169
Keywords
This article is cited by
-
The Role of Ventromedial Hypothalamus Receptors in the Central Regulation of Food Intake
International Journal of Peptide Research and Therapeutics (2021)
-
A novel form of ciliopathy underlies hyperphagia and obesity in Ankrd26 knockout mice
Brain Structure and Function (2015)
-
Modulation of feeding by chronic rAAV expression of a relaxin-3 peptide agonist in rat hypothalamus
Gene Therapy (2013)
-
The Genetics of Anorexia Nervosa
Clinical Pharmacology & Therapeutics (2012)
-
Central nervous system control of metabolism
Nature (2012)