Abstract
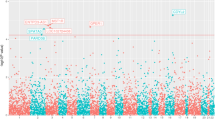
Recent advances demonstrate a relationship between chronic/recurrent inflammation and prostate cancer (PCA). Among inflammatory regulators, toll-like receptors (TLRs) have a critical role in innate immune responses. However, it remains unclear whether variant TLR genes influence PCA risk among men of African descent. Therefore, we evaluated the impact of 32 TLR-associated single-nucleotide polymorphisms (SNPs) on PCA risk among African Americans and Jamaicans. SNP profiles of 814 subjects were evaluated using Illumina’s Veracode genotyping platform. Single and combined effects of SNPs in relation to PCA risk were assessed using age-adjusted logistic regression and entropy-based multifactor dimensionality reduction (MDR) models. Seven sequence variants detected in TLR6, TOLLIP (Toll-interacting protein), IRAK4 (interleukin-1 receptor-associated kinase 4) and IRF3 (interferon regulatory factor 3) were marginally related to PCA. However, none of these effects remained significant after adjusting for multiple hypothesis testing. Nevertheless, MDR modeling revealed a complex interaction between IRAK4 rs4251545 and TLR2 rs1898830 as a significant predictor of PCA risk among US men (permutation testing P-value=0.001). However, these findings require further assessment and validation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 digital issues and online access to articles
$119.00 per year
only $19.83 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
American Cancer Society. Cancer Facts and Figures 2012. American Cancer Society: Atlanta, GA, USA, 2012.
Hsing AW, Tsao L, Devesa SS . International trends and patterns of prostate cancer incidence and mortality. Int J Cancer 2000; 85: 60–67.
American Cancer Society. Cancer Facts and Figures for African Americans 2011–2012. American Cancer Society: Atlanta, GA, USA, 2011.
Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM . Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer 2010; 127: 2893–2917.
Nickel CJ . Prostatitis syndromes: an update for urologic practice. Can J Urol 2000; 7: 1091–1098.
Palapattu GS, Sutcliffe S, Bastian PJ, Platz EA, De Marzo AM, Isaacs WB et al. Prostate carcinogenesis and inflammation: emerging insights. Carcinogenesis 2005; 26: 1170–1181.
Sfanos KS, De Marzo AM . Prostate cancer and inflammation: the evidence. Histopathology 2012; 60: 199–215.
Latz E, Visintin A, Lien E, Fitzgerald KA, Monks BG, Kurt-Jones EA et al. Lipopolysaccharide rapidly traffics to and from the Golgi apparatus with the toll-like receptor 4-MD-2-CD14 complex in a process that is distinct from the initiation of signal transduction. J Biol Chem 2002; 277: 47834–47843.
Frantz S, Ertl G, Bauersachs J . Mechanisms of disease: Toll-like receptors in cardiovascular disease. Nat Clin Pract Cardiovasc Med 2007; 4: 444–454.
Tsujimoto H, Ono S, Efron PA, Scumpia PO, Moldawer LL, Mochizuki H . Role of Toll-like receptors in the development of sepsis. Shock 2008; 29: 315–321.
Blasius AL, Beutler B . Intracellular toll-like receptors. Immunity 2010; 32: 305–315.
Akira S, Yamamoto M, Takeda K . Role of adapters in Toll-like receptor signalling. Biochem Soc Trans 2003; 31 (Part 3): 637–642.
Beutler B . The Toll-like receptors: analysis by forward genetic methods. Immunogenetics 2005; 57: 385–392.
Beutler B . Innate immune responses to microbial poisons: discovery and function of the Toll-like receptors. Annu Rev Pharmacol Toxicol 2003; 43: 609–628.
Gay NJ, Keith FJ . Drosophila Toll and IL-1 receptor. Nature 1991; 351: 355–356.
Gay NJ, Packman LC, Weldon MA, Barna JC . A leucine-rich repeat peptide derived from the Drosophila Toll receptor forms extended filaments with a beta-sheet structure. FEBS Lett 1991; 291: 87–91.
Nakata T, Yasuda M, Fujita M, Kataoka H, Kiura K, Sano H et al. CD14 directly binds to triacylated lipopeptides and facilitates recognition of the lipopeptides by the receptor complex of Toll-like receptors 2 and 1 without binding to the complex. Cell Microbiol 2006; 8: 1899–1909.
Muzio M, Polentarutti N, Bosisio D, Prahladan MK, Mantovani A . Toll-like receptors: a growing family of immune receptors that are differentially expressed and regulated by different leukocytes. J Leukoc Biol 2000; 67: 450–456.
Jin MS, Kim SE, Heo JY, Lee ME, Kim HM, Paik SG et al. Crystal structure of the TLR1–TLR2 heterodimer induced by binding of a tri-acylated lipopeptide. Cell 2007; 130: 1071–1082.
Jin X, Qin Q, Tu L, Zhou X, Lin Y, Qu J . Toll-like receptors (TLRs) expression and function in response to inactivate hyphae of Fusarium solani in immortalized human corneal epithelial cells. Mol Vis 2007; 13: 1953–1961.
Farhat K, Riekenberg S, Heine H, Debarry J, Lang R, Mages J et al. Heterodimerization of TLR2 with TLR1 or TLR6 expands the ligand spectrum but does not lead to differential signaling. J Leukoc Biol 2008; 83: 692–701.
Beutler B, Hoebe K, Georgel P, Tabeta K, Du X . Genetic analysis of innate immunity: identification and function of the TIR adapter proteins. Adv Exp Med Biol 2005; 560: 29–39.
Salaun B, Romero P, Lebecque S . Toll-like receptors’ two-edged sword: when immunity meets apoptosis. Eur J Immunol 2007; 37: 3311–3318.
Rakoff-Nahoum S, Medzhitov R . Toll-like receptors and cancer. Nat Rev Cancer 2009; 9: 57–63.
Creighton CJ, Benham AL, Zhu H, Khan MF, Reid JG, Nagaraja AK et al. Discovery of novel microRNAs in female reproductive tract using next generation sequencing. PLoS One 2010; 5: e9637.
Heinz LX, Rebsamen M, Rossi DC, Staehli F, Schroder K, Quadroni M et al. The death domain-containing protein Unc5CL is a novel MyD88-independent activator of the pro-inflammatory IRAK signaling cascade. Cell Death Differen 2012; 19: 722–731.
Romoser AA, Chen PL, Berg JM, Seabury C, Ivanov I, Criscitiello MF et al. Quantum dots trigger immunomodulation of the NFkappaB pathway in human skin cells. Mol Immunol 2011; 48: 1349–1359.
De Marzo AM, Platz EA, Sutcliffe S, Xu J, Gronberg H, Drake CG et al. Inflammation in prostate carcinogenesis. Nat Rev Cancer 2007; 7: 256–269.
Huang B, Zhao J, Unkeless JC, Feng ZH, Xiong H . TLR signaling by tumor and immune cells: a double-edged sword. Oncogene 2008; 27: 218–224.
Uno K, Kato K, Atsumi T, Suzuki T, Yoshitake J, Morita H et al. Toll-like receptor (TLR) 2 induced through TLR4 signaling initiated by Helicobacter pylori cooperatively amplifies iNOS induction in gastric epithelial cells. Am J Physiol Gastrointest Liver Physiol 2007; 293: G1004–G1012.
Song Z, Yin J, Yao C, Sun Z, Shao M, Zhang Y et al. Variants in the Toll-interacting protein gene are associated with susceptibility to sepsis in the Chinese Han population. Crit Care 2011; 15: R12.
Qian FH, Zhang Q, Zhou LF, Jin GF, Bai JL, Yin KS . Polymorphisms in the toll-like receptor 2 subfamily and risk of asthma: a case–control analysis in a Chinese population. J Invest Allerg Clin Immunol 2010; 20: 340–346.
Slattery ML, Herrick JS, Bondurant KL, Wolff RK . Toll-like receptor genes and their association with colon and rectal cancer development and prognosis. Int J Cancer 2011; 130: 2974–2980.
Pandey S, Mittal RD, Srivastava M, Srivastava K, Singh S, Srivastava S et al. Impact of Toll-like receptors [TLR] 2 (−196 to −174 del) and TLR 4 (Asp299Gly, Thr399Ile) in cervical cancer susceptibility in North Indian women. Gynecol Oncol 2009; 114: 501–505.
Hold GL, Rabkin CS, Chow WH, Smith MG, Gammon MD, Risch HA et al. A functional polymorphism of toll-like receptor 4 gene increases risk of gastric carcinoma and its precursors. Gastroenterology 2007; 132: 905–912.
Rajaraman P, Brenner AV, Neta G, Pfeiffer R, Wang SS, Yeager M et al. Risk of meningioma and common variation in genes related to innate immunity. Cancer Epidemiol Biomarkers Prev 2010; 19: 1356–1361.
Srivastava K, Srivastava A, Kumar A, Mittal B . Gallbladder cancer predisposition: a multigenic approach to DNA-repair, apoptotic and inflammatory pathway genes. PLoS One 2011; 6: e16449.
Song J, Kim DY, Kim CS, Kim HJ, Lee DH, Lee HM et al. The association between Toll-like receptor 4 (TLR4) polymorphisms and the risk of prostate cancer in Korean men. Cancer Genet Cytogenet 2009; 190: 88–92.
Sun J, Wiklund F, Hsu FC, Balter K, Zheng SL, Johansson JE et al. Interactions of sequence variants in interleukin-1 receptor-associated kinase4 and the toll-like receptor 6-1-10 gene cluster increase prostate cancer risk2. Cancer Epidemiol Biomarkers Prev 2006; 15: 480–485.
Cheng I, Plummer SJ, Casey G, Witte JS . Toll-like receptor 4 genetic variation and advanced prostate cancer risk1. Cancer Epidemiol Biomarkers Prev 2007; 16: 352–355.
Kim HJ, Bae JS, Chang IH, Kim KD, Lee J, Shin HD et al. Sequence variants of toll-like receptor 4 (TLR4) and the risk of prostate cancer in Korean men. World J Urol 2011; 30: 225–232.
Zheng SL, Augustsson-Balter K, Chang B, Hedelin M, Li L, Adami HO et al. Sequence variants of toll-like receptor 4 are associated with prostate cancer risk: results from the CAncer Prostate in Sweden Study3. Cancer Res 2004; 64: 2918–2922.
Chen YC, Giovannucci E, Lazarus R, Kraft P, Ketkar S, Hunter DJ . Sequence variants of Toll-like receptor 4 and susceptibility to prostate cancer. Cancer Res 2005; 65: 11771–11778.
Stevens VL, Hsing AW, Talbot JT, Zheng SL, Sun J, Chen J et al. Genetic variation in the toll-like receptor gene cluster (TLR10–TLR1–TLR6) and prostate cancer risk. Int J Cancer 2008; 123: 2644–2650.
Lindstrom S, Hunter DJ, Gronberg H, Stattin P, Wiklund F, Xu J et al. Sequence variants in the TLR4 and TLR6-1-10 genes and prostate cancer risk. Results based on pooled analysis from three independent studies. Cancer Epidemiol Biomarkers Prev 2010; 19: 873–876.
Chen YC, Giovannucci E, Kraft P, Lazarus R, Hunter DJ . Association between Toll-like receptor gene cluster (TLR6, TLR1, and TLR10) and prostate cancer. Cancer Epidemiol Biomarkers Prev 2007; 16: 1982–1989.
Giri VN, Egleston B, Ruth K, Uzzo RG, Chen DY, Buyyounouski M et al. Race, genetic West African ancestry, and prostate cancer prediction by prostate-specific antigen in prospectively screened high-risk men. Cancer Prev Res (Phila, PA) 2009; 2: 244–250.
Tian C, Hinds DA, Shigeta R, Kittles R, Ballinger DG, Seldin MF . A genomewide single-nucleotide-polymorphism panel with high ancestry information for African American admixture mapping. Am J Hum Genet 2006; 79: 640–649.
Falush D, Stephens M, Pritchard JK . Inference of population structure using multilocus genotype data: linked loci and correlated allele frequencies. Genetics 2003; 164: 1567–1587.
Huang QX, Cheng XY, Mao ZC, Wang YS, Zhao LL, Yan X et al. MicroRNA discovery and analysis of pinewood nematode Bursaphelenchus xylophilus by deep sequencing. PLoS One 2010; 5: e13271.
Sun J, Wiklund F, Zheng SL, Chang B, Balter K, Li L et al. Sequence variants in Toll-like receptor gene cluster (TLR6–TLR1–TLR10) and prostate cancer risk4. J Natl Cancer Inst 2005; 97: 525–532.
Greene CS, Penrod NM, Williams SM, Moore JH . Failure to replicate a genetic association may provide important clues about genetic architecture. PLoS One 2009; 4: e5639.
Kidd LC, Vancleave TT, Doll MA, Srivastava DS, Thacker B, Komolafe O et al. No association between variant N-acetyltransferase genes, cigarette smoking and prostate cancer susceptibility among men of African descent. Biomark Cancer 2011; 2011: 1–13.
Jackson MD, Walker SP, Simpson-Smith CM, Lindsay CM, Smith G, McFarlane-Anderson N et al. Associations of whole-blood fatty acids and dietary intakes with prostate cancer in Jamaica. Cancer Causes Control 2012; 23: 23–33.
Menashe I, Rosenberg PS, Chen BE . PGA: power calculator for case–control genetic association analyses. BMC Genet 2008; 9: 36.
Gui J, Andrew AS, Andrews P, Nelson HM, Kelsey KT, Karagas MR et al. A simple and computationally efficient sampling approach to covariate adjustment for multifactor dimensionality reduction analysis of epistasis. Hum Hered 2010; 70: 219–225.
Greene CS, Penrod NM, Kiralis J, Moore JH . Spatially uniform relieff (SURF) for computationally-efficient filtering of gene-gene interactions. BioData Min 2009; 2: 5.
Moore JH, Asselbergs FW, Williams SM . Bioinformatics challenges for genome-wide association studies. Bioinformatics 2010; 26: 445–455.
Moore JH, Gilbert JC, Tsai CT, Chiang FT, Holden T, Barney N et al. A flexible computational framework for detecting, characterizing, and interpreting statistical patterns of epistasis in genetic studies of human disease susceptibility. J Theor Biol 2006; 241: 252–261.
Jakulin A, Bratko I, Smrike D, Demsar J, Zupan B . Attribute Interactions in Medical Data Analysis. Protarus, Cyprus, pp 229–238 2003.
Jakulin A, Bratko I . Analyzing attribute interations. Lect Notes Artif Intell 2003; 2838: 229.
McGill WL . Multivariate information transmission. Psychometrika 1954; 19: 97–116.
Mramor M, Leban G, Demsar J, Zupan B . Visualization-based cancer microarray data classification analysis. Bioinformatics 2007; 23: 2147–2154.
Acknowledgements
We thank Tiva T VanCleave and Dr Nicole A Lavender for preparing DNA samples used in this study. We also thank Dr Rick A Kittles for providing US African-American DNA samples. We appreciate the contract services of Expression Analysis Inc. (http://expressionanalysis.com/) for the generation of genotype data. This work was supported by the following grants: Clinical Translational Science Pilot Grant (to LRK); the JGBCC Bucks for Brains ‘Our Highest Potential’ in Cancer Research Endowment (to LRK); and the P20-MD000175 NIH NCMHD (to KSK).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Genes and Immunity website
Rights and permissions
About this article
Cite this article
Rogers, E., Jones, D., Kidd, N. et al. Toll-like receptor-associated sequence variants and prostate cancer risk among men of African descent. Genes Immun 14, 347–355 (2013). https://doi.org/10.1038/gene.2013.22
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gene.2013.22
Keywords
This article is cited by
-
Tollip interaction with STAT3: a novel mechanism to regulate human airway epithelial responses to type 2 cytokines
Respiratory Research (2022)
-
High-density neutrophils in MGUS and multiple myeloma are dysfunctional and immune-suppressive due to increased STAT3 downstream signaling
Scientific Reports (2020)
-
Beyond the Black Box: A Systematic Review of Breast, Prostate, Colorectal, and Cervical Screening Among Native and Immigrant African-Descent Caribbean Populations
Journal of Immigrant and Minority Health (2015)
-
Association between Toll-like receptor 3 polymorphisms and cancer risk: a meta-analysis
Tumor Biology (2014)