Abstract
Purpose
To evaluate the effects of intraoperative triamcinolone injection on the outcome of pterygium surgery.
Methods
This prospective study included 54 eyes with primary nasal pterygia that underwent pterygium surgery with a bare-sclera technique and intraoperative mitomycin C application. Patients were randomized into two groups; the steroid group that received subconjunctival injection of 12 mg triamcinolone acetonide at the end of surgery, and the control group that did not receive such steroid injection. Main outcome measures included presence of conjunctival inflammation at 1 month postoperatively as well as recurrence of pterygium.
Results
Twelve-month follow-up was completed in 48 eyes (23 in the steroid group and 25 in the control group). At 1 month postoperatively, different grades of conjunctival inflammation were present in 11 (47.8%) of the steroid group and in 14 (56%) of the control group (P=0.39). For eyes with moderate or severe postoperative inflammation, subconjunctival triamcinolone was injected; these included 6 (26.1%) and 9 (36%) in the steroid and control groups, respectively (P=0.54). During follow-up, surgical area showed fine episcleral vessels without fibrous tissue in 1 (4.3%) of the steroid group and 3 (12.0%) of the control group (P=0.33), which all regressed after triamcinolone injection. Conjunctival recurrence of pterygium was seen in 2 (8.7%) of the steroid group and in 1 (4.0%) of the control group (P=0.47). No eye developed corneal recurrence in either group.
Conclusions
In pterygium surgery with a bare-sclera technique and mitomycin C application, intraoperative triamcinolone injection did not significantly reduce postoperative conjunctival inflammation or pterygium recurrence.
Similar content being viewed by others
Introduction
Pterygium is characterized by encroachment of a fleshy fibrovascular tissue from the bulbar conjunctiva onto the cornea. Although previously thought to be a solely degenerative disease,1 new evidence has demonstrated the role of cell proliferation and inflammation in the pathogenesis of pterygium. In addition to the data suggesting proliferative features of pterygium,2, 3, 4, 5 the role of inflammation has been proposed by revealing the increased levels of inflammatory cells and markers in pterygium,6, 7, 8, 9 and also by the clinical data that steroids are beneficial in halting progression of impending recurrent pterygium.10, 11
Many techniques have been developed for pterygium surgery over time. The simple method of removing the head and body of pterygium and leaving the sclera uncovered, the so-called bare-sclera technique, has been associated with high recurrence rates of 32–88%.12, 13, 14 To reduce the recurrence rate after pterygium surgery with a bare-sclera technique, various adjunctive modalities have been used such as beta irradiation and chemical agents including mitomycin C (MMC), 5-fluorouracil, and thiotepa.15 Furthermore, when removal of pterygium is accompanied with a graft, such as conjunctival autograft or amniotic membrane transplantation (AMT), lower recurrence rates have been achieved.15, 16, 17 However, it remains unclear why the bare-sclera technique has poorer outcome with higher recurrence rates than other procedures.
One of the factors that may have a role in the outcome of pterygium surgery is postoperative conjunctival inflammation, treatment of which has been demonstrated to improve the final outcome.18 It has been shown that persistent conjunctival inflammation around the surgical site after pterygium surgery is present in 31–84% of cases with AMT,18, 19, 20 and in 15% eyes with conjunctival autograft.20 However, the rate of such conjunctival inflammation after pterygium surgery with a bare-sclera technique is unknown. On the other hand, it has been suggested that higher recurrence rates after pterygium with AMT compared with conjunctival autograft may be due to this higher rates of postoperative conjunctival inflammation.20 Therefore, it may be speculated that higher recurrence rates after pterygium surgery with a bare-sclera technique is partly contributed by higher rates of postoperative conjunctival inflammation.
To address the above-mentioned questions, this study was designed to evaluate the rate of postoperative conjunctival inflammation around the surgical site after pterygium surgery with a bare-sclera technique. In addition, the role of intraoperative steroid injection in this postoperative conjunctival inflammation and the final outcome was also investigated.
Methods
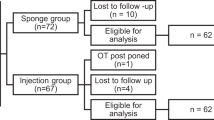
In this prospective randomized study, 54 eyes of 54 patients with primary nasal pterygium underwent surgical excision with a bare-sclera technique and MMC application. The sample size was calculated based on the mean recurrence rates reported by previous studies for bare-sclera technique with intraoperative mitomycin C application for primary pterygia, which were 2.7–38%.21, 22, 23, 24, 25 To detect 10% difference, with α equal to 0.05 and the study power of 80%, at least 21 patients were needed in each group. However, a larger number of patients were included in both groups to compensate for potential loss to follow-up. Using a random-number table, the patients were randomized into two groups; one group received intraoperative injection of 12 mg triamcinolone acetonide (27 eyes, the steroid group), and the other group did not receive such a steroid injection during surgery (27 eyes, the control group). The protocol of the study was approved by Institutional Review Board of Farabi Eye Hospital, Tehran, Iran. Tenets of the Declaration of Helsinki were also followed throughout the study.
Before surgery and at all visits after surgery, each patient had a complete ocular examination including slit lamp photography, and measurement of best-corrected visual acuity and intraocular pressure. Preoperatively, morphology of pterygium was graded according to what was described by Tan et al.21 In this grading, pterygia were graded as grade T1 (atrophic pterygium) in which episcleral vessels were un-obscured by the body of pterygium, grade T3 (fleshy pterygium) in which episcleral vessels were totally obscured, and grade T2 (those between grades T1 and T3) with partially obscured episcleral vessels.
Before surgery, details of the study were fully explained for the patients and all consented to participate in the study. All surgeries were performed by single surgeon under retrobulbar anesthesia. For surgery, the head and body of the pterygium were first removed by a similar technique in all patients, with resection of the body at 2 mm in front of plica semilunaris. This was followed by removal of subconjunctival fibrovascular tissue for 2 mm beyond the conjunctival edges, and polishing of the cornea by a diamond burr. After minimal cauterization of bleeding vessels, MMC was applied for 3 min both on the bare sclera and under the conjunctival edges by using pieces of Weck-Cel surgical sponge soaked in 0.02% MMC solution. After washing the eye surface with 100 ml of balanced salt solution, the conjunctival edges were sutured to the underlying episclera using interrupted 10-0 nylon sutures without conjunctival advancement. Finally, at the end of surgery, 12 mg of triamcinolone acetonide was injected subconjunctivally around the surgical site for patients in the steroid group. Patients in the control group did not receive any steroid injection intraoperatively.
After surgery, all patients received an identical regimen of a topical antibiotic for 2 weeks and tapering topical steroids for 3 months. The latter included 0.1% betamethasone four times a day for 1 month followed by 0.1% fluorometholone × 4/day for 2 weeks, × 3/day for 2 weeks, × 2/day for 2 weeks, and × 1/day for 2 weeks. Postoperative follow-up examinations were performed at 1 day, 1 week, 2 weeks, 1 month, and 3, 6, 9, and 12 months after surgery. Sutures were removed after 1 week postoperatively in both groups.
Presence of postoperative conjunctival inflammation around the surgical site was assessed at 1 month after surgery and graded as 0 (none), I (mild), II (moderate), and III (severe) as described before.18 Eyes with grade 2 and 3 inflammation received subconjunctival injection of 12 mg triamcinolone acetonide. Postoperative outcome of pterygium surgery was reported using a grading system previously described.13 This grading included grade 1 as normal conjunctiva at the surgical area, grade 2 as the presence of fine episcleral vessels without fibrous tissue in the surgical area, grade 3 as the presence fibrovascular tissue in surgical area but without invasion onto the cornea (conjunctival recurrence), and grade 4 as true recurrence in which the fibrovascular tissue invaded onto the cornea (corneal recurrence). For eyes with grade 2 outcome, 12 mg of triamcinolone acetonide was injected subconjunctivally. Eyes with conjunctival recurrence of pterygium (grade 3) received either one single subconjunctival injection of 12 mg triamcinolone acetonide or two weekly intralesional injections of 5 mg 5-fluorouracil.
Statistical analysis was performed using SPSS version 16 (SPSS Inc., Chicago, IL, USA). Chi-square test and Student t test were used to compare qualitative and continuous quantitative variables, respectively, between the steroid group and the control group. P-values ⩽0.05 were considered as statistically significant.
Results
Of 54 eyes included in this study, 12-month follow-up was completed in 48 eyes of 48 patients (36 men and 12 women) with a mean age of 41.8±11.6 years (range, 24–68 years). These included 23 eyes in the steroid group and 25 eyes in the control group. There were no statistically significant differences in age, gender, and grade of preoperative pterygium morphology between the two groups (Table 1). No intraoperative complication was noted in any eye in either group.
At 1 month after surgery, examination revealed conjunctival inflammation around the surgical area, most notably at the caruncular border, in 11 eyes (47.8%) and 14 eyes (56%) in the steroid group and the control group, respectively (P=0.39) (Table 1). In the steroid group, grading of this postoperative inflammation included grade I (mild) in 5 eyes (21.7%), grade II (moderate) in 4 eyes (17.4%), and grade III (severe) in 2 eyes (8.7%). However, in the control group the inflammation was as grade I in 5 eyes (20.0%), grade II in 8 eyes (32%), and grade III in 1 eye (4%). Subconjunctival injection of triamcinolone acetonide was performed in 6 eyes (26.1%) and 9 eyes (36.0%) in the steroid and control groups, respectively (P=0.54). This injection resulted in resolution of the inflammation in all these eyes.
During the follow-up, grade 2 of postoperative outcome was observed in 1 eye (4.3%) and 3 eyes (12.0%) of the steroid group and the control group, respectively (P=0.33, Figure 1). This postoperative appearance was noticed at 3-month visit in all cases. One single subconjunctival injection of 12 mg triamcinolone acetonide resulted in regression of the fine vessels in all these 4 cases (Figure 1). Grade 3 outcome (conjunctival recurrence) was seen in 2 eyes (8.7%) of the steroid group and in 1 eye (4.0%) of the control group, with no statistically significance difference between the two groups (P=0.47, Figure 2). The recurrence was noted at 5 months postoperatively in one patient of the steroid group and at 6-month visit in 2 other cases. The eyes with conjunctival recurrence received either one single subconjunctival injection of 12 mg triamcinolone acetonide (one eye in steroid group) or two weekly intralesional injections of 5 mg 5-fluorouracil (two eyes, one in each group); none developed true corneal recurrence during the follow-up.
Grade 2 outcome after pterygium surgery with a bare-sclera technique and intraoperative MMC application. An eye with grade T2 of pterygium morphology (a) in the steroid group developed fine episcleral vessels at 3-month visit (b). Subconjunctival injection of triamcinolone resulted in regression of the vessels with no recurrence of pterygium during 12 months of follow-up (c). An eye with grade T2 pterygium (d) in the control group showed grade 2 outcome at 3 months postoperatively (e). Regression of the vessels ensued after triamcinolone injection (f).
Grade 3 outcome after pterygium surgery with a bare-sclera technique and intraoperative MMC application. An eye with grade T3 of pterygium morphology (a) in the steroid group had grade I conjunctival inflammation at 1 month postoperatively (b). The patient developed a conjunctival recurrence at 5 months after surgery (c). An eye with grade T3 pterygium (d) in the steroid group showed no conjunctival inflammation at 1 month after surgery (e). However, a conjunctival recurrence was observed at 6 months postoperatively that did not progress to the corneal recurrence after injection of triamcinolone (f). An eye with grade T1 pterygium (g) in the control group had grade II conjunctival inflammation and a large pyogenic granuloma at 1 month postoperatively (h). Despite resolution of inflammation and granuloma with triamcinolone injection, a conjunctival recurrence was noted at 6 months after surgery (i).
Surgical outcomes in various grades of conjunctival inflammation after surgery have been shown in Table 2. In the steroid group, grade 2 or 3 outcomes were observed in 2 eyes (11.8%) of cases with none or mild inflammation, and in 1 eye (16.7%) of patients with moderate or severe inflammation that received steroid injection (P=0.62). In the control group, grade 2 or 3 outcomes were seen in 2 eyes (12.5%) of patients with none or mild inflammation, and in 2 eyes (22.2%) of cases with moderate or severe inflammation that received triamcinolone injection (P=0.46).
Pyogenic granuloma developed in 1 eye (4.3%) of the steroid group at 3 months postoperatively and in 2 eyes (8.0%) of the control group, both at 1 month after surgery (Table 1). All pyogenic granulomata were noted at the nasal border of the surgical area. In the control group, one eye had grade II conjunctival inflammation and a large pyogenic granuloma at 1 month postoperatively (Figure 2h). Despite resolution of inflammation and granuloma with triamcinolone injection, this eye subsequently developed conjunctival recurrence at 6 months postoperatively (Figure 2i). Increased intraocular pressure was noted in one eye in the steroid, which was controlled medically. No MMC-related complication was observed in any eye during 12 months of follow-up.
Discussion
This prospective randomized study showed that conjunctival inflammation was present in approximately half of the patients at 1 month after pterygium surgery with a bare-sclera technique and intraoperative MMC application. In addition to use of MMC, aggressive control of postoperative conjunctival inflammation with steroids resulted in a low recurrence rate of pterygium after surgery. However, intraoperative injection of triamcinolone acetonide did not significantly reduce the postoperative conjunctival inflammation or pterygium recurrence.
In our study, at 1 month postoperatively 11 eyes (47.8%) in the steroid group and 14 eyes (56%) in the control group had conjunctival inflammation around the surgical site, most notably at the caruncular border. Such inflammation has previously been reported after pterygium surgery with other techniques including conjunctival autograft and AMT.18, 19, 20 Without intraoperative steroid injection, previous studies have showed rates of 15% or 31–84% for such postoperative conjunctival inflammation after pterygium surgery with conjunctival autograft or AMT, respectively.18, 19, 20 The cause for such persistent inflammation and its varying rates after different surgical techniques are unknown. Although longer duration of suture stay had been speculated to be partly responsible for the higher rate in the AMT group,20 this postoperative inflammation has also been observed with using fibrin glue instead of sutures for AMT, even though with lower incidence (21.4% vs 61.5%).18
Intraoperative injection of triamcinolone acetonide in our study was associated with a lower rate of conjunctival inflammation at 1 month after surgery (47.8% vs 56%); however, the difference was not statistically significant (P=0.39). A previous retrospective non-randomized study also showed lack of the correlation between intraoperative triamcinolone injection and the conjunctival inflammation at 1 month after pterygium surgery with AMT.18 Triamcinolone acetonide is an intermediate-acting steroid that has a duration of effect for 15–21 days in the conjunctiva.11 As the inflammation was evaluated at 1 month postoperatively, it may be speculated that by that time the effects of the steroid might have already been worn off. This may explain the non-significant difference in incidence of the conjunctival inflammation between the two groups in our study. Usage of steroids with longer duration of effect may be more effective in suppressing such inflammation, even though they may be associated with higher rates of steroid-related complications.
In our study during 12 months of follow-up after pterygium surgery using a bare-sclera technique with MMC application, conjunctival recurrences (grade 3 outcome) were noted in 2 eyes (8.7%) and 1 eye (4.0%) in the steroid group and the control group, respectively (Figure 2). These cases received subconjunctival injection of either 5-fluorouracil or triamcinolone, as described before,10, 11 resulting in no corneal recurrence in either group during the follow-up. Surgery for primary pterygium using a bare-sclera technique with intraoperative MMC application and without intraoperative steroid injection has previously been reported to have recurrence rates of 2.7–38%.21, 22, 23, 24, 25, 26 However, these studies have most commonly included only cases with corneal recurrence and not the conjunctival recurrence. The better outcome of our study may be attributed to the removal of subconjunctival fibrovascular tissue,19, 27 and aggressive control of postoperative inflammation with steroid injection.
In the present study, intraoperative triamcinolone injection did not significantly reduce the recurrence rate compared with the control group (P=0.47). In one previous study on 51 patients with primary pterygium, surgery using a bare-sclera technique with intraoperative MMC application and subconjunctival injection of 20 mg depot steroid resulted in no recurrence during 4–14 months of follow-up.28 However, no control group was included. On the other hand, it has also been demonstrated that treatment of 1-month postoperative conjunctival inflammation by steroid injection improved the surgical outcome of pterygium.18, 20 Not only inadequate postoperative topical steroids have been associated with higher recurrence rates,29 but one study also showed that in eyes with risk factors for recurrence (including conjunctival inflammation and granuloma formation) postoperative subconjunctival injection of steroid leads to an improved surgical outcome.11 Therefore, it seems postoperative steroid therapy tailored to the conjunctival inflammation has a more important role than intraoperative steroids do.
In the present study, grade 2 outcome developed in 4 eyes (8.3%), including 1 eye (4.3%) in the steroid group and 3 eyes (12%) in the control group (Figure 1). Although the difference between the two groups seems to be clinically significant, it was not statistically significant (P=0.33), which may be due to the small sample size. The incidence and significance of grade 2 outcome after pterygium surgery have not been completely described in the literature. In addition, it is not clear whether such fine vessels will invariably progress to the conjunctival recurrence if left untreated. As a cautionary measure, all eyes with grade 2 outcome in our study received subconjunctival triamcinolone injection, which resulted in regression of the fine episcleral vessels in all cases (Figure 1). Interestingly, in all these cases grade 2 outcome was noticed at the 3-month visit. None of the cases with grade 3 outcome, which were detected at 5–6 months postoperatively, had a prior grade 2 outcome. This may be due to the fact that in these cases grade 2 outcome developed at some point between the 3-month visit and the diagnosis of the conjunctival recurrence. Therefore, more frequent patient visits during this time period to detect and treat grade 2 outcomes might be a reasonable measure to prevent possible progression to the conjunctival recurrence. Further studies are required to determine the significance of grade 2 outcomes after pterygium surgery and its optimal management.
Correlation of the surgical outcome with the grade of conjunctival inflammation at 1 month after surgery revealed that the recurrence rates were comparable between those with grade 0 or 1 inflammation and cases with more severe conjunctival inflammation that has been treated with steroid injection (Table 2). This was the case in both the steroid group and the control group. As the role of postoperative conjunctival inflammation in pterygium recurrence has well been documented before,18 the data showed the beneficial role of treatment of postoperative inflammation in the final surgical outcome. Interestingly, one eye with moderate inflammation and large pyogenic granuloma at 1 month postoperatively developed conjunctival recurrence despite triamcinolone injection (Figure 2, Bottom row). Cases with such exuberant postoperative reaction may need more aggressive anti-inflammatory and/or anti-VEGF treatment.
This study was performed in a prospective randomized fashion and patients in the two groups were similar in terms of age, gender, and more importantly, grade of pterygium morphology preoperatively. On the other hand, patients were followed for 1 year after surgery, which is long enough to detect the vast majority of pterygium recurrences. However, it is not sufficient to observe the possible complications of MMC, such as necrotizing scleritis or scleral melting and calcification, which may develop years later.30 With these limitations in mind, our study showed that pterygium surgery with a bare-sclera technique using intraoperative MMC application and aggressive control of conjunctival inflammation resulted in a good surgical outcome with a low recurrence rate. There was no significant additional benefit of using intraoperative triamcinolone injection.

References
Austin P, Jakobiec FA, Iwamoto T . Elastodysplasia and elastodystrophy as the pathologic bases of ocular pterygia and pinguecula. Ophthalmology 1983; 90: 96–109.
Bradley JC, Yang W, Bradley RH, Reid TW, Schwab IR . The science of pterygia. Br J Ophthalmol 2010; 94: 815–820.
Bai H, Teng Y, Wong L, Pang CP, Yam GH . Proliferative and migratory aptitude in pterygium. Histochem Cell Biol 2010; 134: 527–535.
Mortada A, Hamdi EE, Shiwi TE, Einein GA . Histopathology of recurrent true pterygium. Bull Ophthalmol Soc Egypt 1968; 61: 117–122.
Hill JC, Maske R . Pathogenesis of pterygium. Eye (Lond) 1989; 3 (Pt 2): 218–226.
Awdeh RM, DeStafeno JJ, Blackmon DM, Cummings TJ, Kim T . The presence of T-lymphocyte subpopulations (CD4 and CD8) in pterygia: evaluation of the inflammatory response. Adv Ther 2008; 25: 479–487.
Tekelioglu Y, Turk A, Avunduk AM, Yulug E . Flow cytometrical analysis of adhesion molecules, T-lymphocyte subpopulations and inflammatory markers in pterygium. Ophthalmologica 2006; 220: 372–378.
Wen Z, Liu Z . The abnormal expression of interleukine-1 family in pterygium. Yan Ke Xue Bao 2003; 19: 133–136.
Di Girolamo N, McCluskey P, Lloyd A, Coroneo MT, Wakefield D . Expression of MMPs and TIMPs in human pterygia and cultured pterygium epithelial cells. Invest Ophthalmol Vis Sci 2000; 41: 671–679.
Prabhasawat P, Tesavibul N, Leelapatranura K, Phonjan T . Efficacy of subconjunctival 5-fluorouracil and triamcinolone injection in impending recurrent pterygium. Ophthalmology 2006; 113: 1102–1109.
Paris Fdos S, de Farias CC, Melo GB, Dos Santos MS, Bastista JL, Gomes JA . Postoperative subconjunctival corticosteroid injection to prevent pterygium recurrence. Cornea 2008; 27: 406–410.
Chen PP, Ariyasu RG, Kaza V, LaBree LD, McDonnell PJ . A randomized trial comparing mitomycin C and conjunctival autograft after excision of primary pterygium. Am J Ophthalmol 1995; 120: 151–160.
Panda A, Das GK, Tuli SW, Kumar A . Randomized trial of intraoperative mitomycin C in surgery for pterygium. Am J Ophthalmol 1998; 125: 59–63.
Frucht-Pery J, Ilsar M, Hemo I . Single dosage of mitomycin C for prevention of recurrent pterygium: preliminary report. Cornea 1994; 13: 411–413.
Hirst LW . The treatment of pterygium. Surv Ophthalmol 2003; 48: 145–180.
Ang LP, Chua JL, Tan DT . Current concepts and techniques in pterygium treatment. Curr Opin Ophthalmol 2007; 18: 308–313.
Marcovich AL, Bahar I, Srinivasan S, Slomovic AR . Surgical management of pterygium. Int Ophthalmol Clin 2010; 50: 47–61.
Kheirkhah A, Casas V, Sheha H, Raju VK . Tseng SCG. Role of conjunctival inflammation in surgical outcome after amniotic membrane transplantation with or without fibrin glue for pterygium. Cornea 2008; 27: 56–63.
Solomon A, Pires RTF, Tseng SCG . Amniotic membrane transplantation after extensive removal of primary and recurrent pterygia. Ophthalmology 2001; 108: 449–460.
Kheirkhah A, Nazari R, Nikdel M, Ghassemi H, Hashemi H, Behrouz MJ . Postoperative conjunctival inflammation after pterygium surgery with amniotic membrane transplantation versus conjunctival autograft. Am J Ophthalmol 2011; 152: 733–738.
Tan DT, Chee SP, Dear KB, Lim AS . Effect of pterygium morphology on pterygium recurrence in a controlled trial comparing conjunctival autografting with bare sclera excision. Arch Ophthalmol 1997; 115: 1235–1240.
Prabhasawat P, Barton K, Burkett G, Tseng SC . Comparison of conjunctival autografts, amniotic membrane grafts, and primary closure for pterygium excision. Ophthalmology 1997; 104: 974–985.
Sánchez-Thorin JC, Rocha G, Yelin JB . Meta-analysis on the recurrence rates after bare sclera resection with and without mitomycin C use and conjunctival autograft placement in surgery for primary pterygium. Br J Ophthalmol 1998; 82: 661–665.
Koranyi G, Artzén D, Seregard S, Kopp ED . Intraoperative mitomycin C versus autologous conjunctival autograft in surgery of primary pterygium with four-year follow-up. Acta Ophthalmol 2012; 90: 266–270.
Avisar R, Gaton DD, Loya N, Appel I, Weinberger D . Intraoperative mitomycin C 0.02% for pterygium: effect of duration of application on recurrence rate. Cornea 2003; 22: 102–104.
Frucht-Pery J, Siganos CS, Ilsar M . Intraoperative application of topical mitomycin C for pterygium surgery. Ophthalmology 1996; 103: 674–677.
Levy RL, Naidu S, Jacobson L . Safety and efficacy of the technique of complete tenon's membrane excision and mitomycin C in pterygium surgery. Eye Contact Lens 2005; 31: 105–108.
Mpyet C, Oko H . Results of intra-operative 0.5mg/ml mitomycin C with 20mg depo steroid in the treatment of primary pterygium. Cent Afr J Med 2000; 46: 330–332.
Yaisawang S, Piyapattanakorn P . Role of post-operative topical corticosteroids in recurrence rate after pterygium excision with conjunctival autograft. J Med Assoc Thai 2003; 86 (suppl 2): S215–S223.
Hardten DR, Samuelson TW . Ocular toxicity of mitomycin-C. Int Ophthalmol Clin 1999; 39: 79–90.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kheirkhah, A., Nazari, R., Safi, H. et al. Effects of intraoperative steroid injection on the outcome of pterygium surgery. Eye 27, 906–914 (2013). https://doi.org/10.1038/eye.2013.142
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/eye.2013.142