Abstract
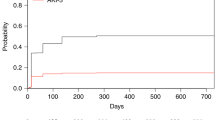
To analyze the incidence, characteristics and risk factors of hyperbilirubinemia after allogeneic hematopoietic cell transplantation with reduced-intensity conditioning (allo-RIC), we conducted a retrospective study in three Spanish centers. We analyzed 452 consecutive patients receiving allo-RIC. Of these, 92 patients (20%) developed marked hyperbilirubinemia (>4 mg/day or >68.4 μM) after allo-RIC. The main causes of marked hyperbilirubinemia after transplant were cholestasis due to GVHD or sepsis (n=57, 62%) and drug-induced cholestasis (n=13, 14%). A total of 22 patients with marked hyperbilirubinemia (24%) underwent liver biopsy. The most frequent histological finding was iron overload alone (n=6) or in combination with other features (n=6). In multivariate analysis, the risk factors for marked hyperbilirubinemia after allo-RIC were non-HLA-identical sibling donors (hazard ratio (HR) 2.2 (95% confidence interval (CI) 1.4–3.6) P=0.001), female donors to male recipients (HR 2.1 (95% CI 1.3–3.3) P=0.003) and high levels of bilirubin and γ-glutamyl transpeptidase before transplant (HR 4.5 (95% CI 2.5–8.4) P<0.001 and HR 4.6 (95% CI 2.6–8.1) P<0.001, respectively). Patients with marked hyperbilirubinemia showed higher 4-year nonrelapse mortality (HR 1.3 (95% CI 1–1.7), P=0.02) and lower 4-year OS (HR 1.4 (95%CI 1.3–1.7), P<0.001) than patients without. In conclusion, we confirm that marked hyperbilirubinemia is frequent and diverse after allo-RIC. Development of marked hyperbilirubinemia after allo-RIC is associated with worse outcome of the procedure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Hogan WJ, Maris M, Storer B, Sandmaier BM, Maloney DG, Schoch HG et al. Hepatic injury after nonmyeloablative conditioning followed by allogeneic hematopoietic cell transplantation: a study of 193 patients. Blood 2004; 103: 78–84.
Kusumi E, Kami M, Kanda Y, Murashige N, Seki K, Fujiwara M et al. Hepatic injury following reduced intensity unrelated cord blood transplantation for adult patients with hematological diseases. Biol Blood Marrow Transplant 2006; 12: 1302–1309.
Ozdoǧan O, Ratip S, Ahdab YA, Dane F, Ahdab HA, Imeryüz N et al. Causes and risk factors for liver injury following bone marrow transplantation. J Clin Gastroenterol 2003; 36: 421–426.
Kami M, Mori S, Tanikawa S, Akiyama H, Onozawa Y, Tanaka T et al. Risk factors for hepatic veno-occlusive disease after bone marrow transplantation: retrospective analysis of 137 cases at a single institution. Bone Marrow Transplant 1997; 20: 397–402.
Sucak GT, Yegin ZA, Ozkurt ZN, Aki SZ, Karakan T, Akyol G . The role of liver biopsy in the workup of liver dysfunction late after SCT: is the role of iron overload underestimated? Bone Marrow Transplant 2008; 42: 461–467.
Duarte RF, Delgado J, Shaw BE, Wrench DJ, Ethell M, Patch D et al. Histologic features of the liver biopsy predict the clinical outcome for patients with graft-versus-host disease of the liver. Biol Blood Marrow Transplant 2005; 11: 805–813.
Barba P, Piñana JL, Valcárcel D, Querol L, Martino R, Sureda A et al. Early and late neurological complications after reduced-intensity conditioning allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2009; 15: 1439–1446.
Piñana JL, Valcárcel D, Martino R, Barba P, Moreno E, Sureda A et al. Study of kidney function impairment after reduced-intensity conditioning allogeneic hematopoietic stem cell transplantation. A single-center experience. Biol Blood Marrow Transplant 2009; 15: 21–29.
Peres E, Levine JE, Khaled YA, Ibrahim RB, Braun TM, Krijanovski OI et al. Cardiac complications in patients undergoing a reduced-intensity conditioning hematopoietic stem cell transplantation. Bone Marrow Transplant 2010; 45: 149–152.
Martino R, Iacobelli S, Brand R, Jansen T, van Biezen A, Finke J et al. Retrospective comparison of reduced-intensity conditioning and conventional high-dose conditioning for allogeneic hematopoietic stem cell transplantation using HLA-identical sibling donors in myelodysplastic syndromes. Blood 2006; 108: 836–846.
Valcárcel D, Martino R, Caballero D, Martin J, Ferra C, Nieto JB et al. Sustained remissions of high-risk acute myeloid leukemia and myelodysplastic syndrome after reduced-intensity conditioning allogeneic hematopoietic transplantation: chronic graft-versus-host disease is the strongest factor improving survival. J Clin Oncol 2008; 26: 577–584.
Martino R, Pérez-Simón JA, Moreno E, Queraltó JM, Caballero D, Mateos M et al. Reduced-intensity conditioning allogeneic blood stem cell transplantation with fludarabine and oral busulfan with or without pharmacokinetically targeted busulfan dosing in patients with myeloid leukemia ineligible for conventional conditioning. Biol Blood Marrow Transplant 2005; 11: 437–447.
Barba P, Piñana JL, Fernández-Avilés F, Perez-Simón JA, Martino R, López-Guerrero E et al. Pretransplant liver function impacts on the outcome of allogeneic hematopoietic stem cell transplantation: a study of 455 patients. Biol Blood Marrow Transplant 2011; 17: 1653–1661.
Piñana JL, Martino R, Barba P, Margall N, Roig MC, Valcarcel D et al. Cytomegalovirus infection and disease after reduced intensity conditioning allogeneic stem cell transplantation: single-centre experience. Bone Marrow Transplant 2009; 45: 534–542.
Martino R, Piñana JL, Parody R, Valcárcel D, Sureda A, Brunet S et al. Lower respiratory tract respiratory virus infections increase the risk of invasive aspergillosis after a reduced-intensity allogeneic hematopoietic SCT. Bone Marrow Transplant 2009; 44: 749–756.
McDonald GB, Sharma P, Matthews DE, Shulman HM, Thomas ED . Venocclusive disease of the liver after bone marrow transplantation: diagnosis, incidence, and predisposing factors. Hepatology 198; 4: 116–122.
Klein JP, Rizzo JD, Zhang MJ, Keiding N . Statistical methods for the analysis and presentation of the results of bone marrow transplants. Part I: unadjusted analysis. Bone Marrow Transplant 2001; 28: 909–915.
Klein JP, Rizzo JD, Zhang MJ, Keiding N . Statistical methods for the analysis and presentation of the results of bone marrow transplants. Part 2: regression modeling. Bone Marrow Transplant 2001; 28: 1001–1011.
Kaplan EL, Meier P . Non parametric estimation from incomplete observations. J Am Statistical Assoc 1958; 53: 457–481.
Sakai M, Strasser SI, Shulman HM, McDonald SJ, Schoch HG, McDonald GB . Severe hepatocellular injury after hematopoietic cell transplant: incidence, etiology and outcome. Bone Marrow Transplant 2009; 44: 441–447.
Subbarao G, Haut PR, Johnson CS, Gowan D, Molleston JP . Incidence, etiology, and risk factors for liver dysfunction in children following hematopoietic stem cell transplantation. Pediatr Transplant 2006; 10: 682–689.
Ho GT, Parker A, MacKenzie JF, Morris AJ, Stanley AJ . Abnormal liver function tests following bone marrow transplantation: aetiology and role of liver biopsy. Eur J Gastroenterol Hepatol 2004; 16: 157–162.
National Cancer Institute. Common Toxicity Criteria for Adverse Events. Version 3.0. National Cancer Institute: Bethesda, MD, 2003.
Guilfoyle R, Demers A, Bredeson C, Richardson E, Rubinger M, Szwajcer D et al. Performance status, but not the hematopoietic cell transplantation comorbidity index (HCT-CI), predicts mortality at a Canadian transplant center. Bone Marrow Transplant 2009; 43: 133–139.
Xhaard A, Porcher R, Chien JW, de Latour RP, Robin M, Ribaud P et al. Impact of comorbidity indexes on non-relapse mortality. Leukemia 2008; 22: 2062–2069.
Ramasamy K, Lim ZY, Pagliuca A, Grundy R, Devereux S, Ho AY et al. Incidence and management of hepatic venoocclusive disease in 237 patients undergoing reduced-intensity conditioning (RIC) haematopoietic stem cell transplantation (HSCT). Bone Marrow Transplant 2006; 38: 823–824.
Russell JA, Tran HT, Quinlan D, Chaudhry A, Duggan P, Brown C et al. Once-daily intravenous busulfan given with fludarabine as conditioning for allogeneic stem cell transplantation: study of pharmacokinetics and early clinical outcomes. Biol Blood Marrow Transplant 2002; 8: 468–476.
Carreras E, Bertz H, Arcese W, Vernant JP, Tomas JF, Hagglund H et al. Incidence and outcome of hepatic veno-occlusive disease after blood or marrow transplantation: a prospective cohort study of the European Group for Blood and Marrow Transplantation. European Group for Blood and Marrow Transplantation Chronic Leukemia Working Party. Blood 1998; 92: 3599–3604.
Cutler C, Stevenson K, Kim HT, Richardson P, Ho VT, Linden E et al. Sirolimus is associated with veno-occlusive disease of the liver after myeloablative allogeneic stem cell transplantation. Blood 2008; 112: 4425–4431.
Coppell JA, Richardson PG, Soiffer R, Martin PL, Kernan NA, Chen A et al. Hepatic veno-occlusive disease following stem cell transplantation: incidence, clinical course, and outcome. Biol Blood Marrow Transplant 2010; 16: 157–168.
Lim ZY, Fiaccadori V, Gandhi S, Hayden J, Kenyon M, Ireland R et al. Impact of pre-transplant serum ferritin on outcomes of patients with myelodysplastic syndromes or secondary acute myeloid leukaemia receiving reduced intensity conditioning allogeneic haematopoietic stem cell transplantation. Leuk Res 2010; 34: 723–727.
Gooley TA, Rajvanshi P, Schoch HG, McDonald GB . Serum bilirubin levels and mortality after myeloablative allogeneic hematopoietic cell transplantation. Hepatology 2005; 41: 345–352.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005; 106: 2912–2919.
Flowers ME, Inamoto Y, Carpenter PA, Lee SJ, Petersdorf EW, Pereira SE et al. Comparative analysis of risk factors for acute and for chronic graft-versus-host-disease according to National Institute of Health consensus criteria. Blood 2011; 117: 3214–3219.
Parody R, Martino R, Rovira M, Vazquez L, Vázquez MJ, de la Cámara R et al. Severe infections after unrelated donor allogeneic hematopoietic stem cell transplantation in adults: comparison of cord blood transplantation with peripheral blood and bone marrow transplantation. Biol Blood Marrow Transplant 2006; 12: 734–748.
Qureshi A, Marshall L, Lancaster D . Defibrotide in the prevention and treatment of veno-occlusive disease in autologous and allogeneic stem cell transplantation in children. Pediatr Blood Cancer 2008; 50: 831–832.
Gaziev J, Nguyen L, Puozzo C, Mozzi AF, Casella M, Perrone Donnorso M et al. Novel pharmacokinetic behavior of intravenous busulfan in children with thalassemia undergoing hematopoietic stem cell transplantation: a prospective evaluation of pharmacokinetic and pharmacodynamic profile with therapeutic drug monitoring. Blood 2010; 115: 4597–4604.
Hassan Z, Hellström-Lindberg E, Alsadi S, Edgren M, Hägglund H, Hassan M . The effect of modulation of glutathione cellular content on busulphan-induced cytotoxicity on hematopoietic cells in vitro and in vivo. Bone Marrow Transplant 2002; 30: 141–147.
Cantoni N, Gerull S, Heim D, Halter J, Bucher C, Buser A et al. Order of application and liver toxicity in patients given BU and CY containing conditioning regimens for allogeneic hematopoietic SCT. Bone Marrow Transplant 2011; 46: 344–349.
Barkholt L, Remberger M, Hassan Z, Fransson K, Omazic B, Svahn BM et al. A prospective randomized study using N-acetyl-L-cysteine for early liver toxicity after allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant 2008; 41: 785–790.
Sakaguchi H, Watanabe N, Muramatsu H, Doisaki S, Yoshida N, Matsumoto K et al. Danaparoid as the prophylaxis for hepatic veno-occlusive disease after allogeneic hematopoietic stem cell transplantation in childhood hematological malignancy. Pediatr Blood Cancer 2010; 55: 1118–1125.
Chalandon Y, Roosnek E, Mermillod B, Newton A, Ozsahin H, Wacker P et al. Prevention of veno-occlusive disease with defibrotide after allogeneic stem cell transplantation. Biol Blood Marrow Transplant 2004; 10: 347–354.
Ruutu T, Eriksson B, Remes K, Juvonen E, Volin L, Remberger M et al. Ursodeoxycholic acid for the prevention of hepatic complications in allogeneic stem cell transplantation. Blood 2002; 100: 1977–1983.
Park SH, Lee MH, Lee H, Kim HS, Kim K, Kim WS et al. A randomized trial of heparin plus ursodiol vs heparin alone to prevent hepatic veno-occlusive disease after hematopoietic stem cell transplantation. Bone Marrow Transplant 2002; 29: 137–143.
Tay J, Tinmouth A, Fergusson D, Huebsch L, Allan DS . Systematic review of controlled clinical trials on the use of ursodeoxycholic acid for the prevention of hepatic veno-occlusive disease in hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2007; 13: 206–217.
Acknowledgements
JL Piñana is supported by grant from the Instituto de Salud Carlos III (CM06/00139, Ministerio de Sanidad, Spain). This study was supported in part by Grant RD06/0020/0101 from the Instituto de Salud Carlos III, Ministerio de Sanidad, Spain, and from the Fundació d’Investigació Sant Pau (Ferrer-Salat Award).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Barba, P., Martino, R., Perez-Simón, J. et al. Incidence, characteristics and risk factors of marked hyperbilirubinemia after allogeneic hematopoietic cell transplantation with reduced-intensity conditioning. Bone Marrow Transplant 47, 1343–1349 (2012). https://doi.org/10.1038/bmt.2012.25
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2012.25
Keywords
This article is cited by
-
HEV infection in stem cell transplant recipients—retrospective study of EBMT Infectious Diseases Working Party
Bone Marrow Transplantation (2022)
-
Early bilirubinemia after allogeneic stem cell transplantation—an endothelial complication
Bone Marrow Transplantation (2021)
-
Diagnostic Yield of Transjugular Liver Biopsy Samples to Evaluate for Infectious Etiology of Liver Dysfunction in Bone Marrow Transplant Recipients
CardioVascular and Interventional Radiology (2014)