« Prev Next »
The Cell's War Against Viruses
As part of our war against viruses, scientists have tried to understand all they can about these tiny, but complex, enemies. We have learned much in the past decades. For example, with the aid of powerful microscopes, genetic tools, and biochemical methods, scientists have discovered that viruses only carry the essential elements necessary for recognizing their host cells and replicating their genome inside the host. However, this economy has a cost. Given their simplicity, viruses are completely dependent on the biosynthetic machinery of the host cell. Therefore, they have developed exquisite tricks to harness the host cell's functions for their own benefit. So to speak, viruses are analogous to an enemy army that will not stop until it conquers the host cell's territory and uses up the host cell's resources.
For example, cells have evolved the endocytic machinery, a complex array of vesicles and proteins, to internalize molecules. In response, some viruses have developed strategies to hijack the cell's endocytic machinery to enter the cell cytoplasm or the nucleus, depending on the replication strategy of the particular virus. The location in which the viral replication cycle takes place is dictated by the type of nucleic acid that makes up the viral genome (either DNA or RNA). The irony is that by studying the mechanisms of viral entry, scientists have gained a greater understanding of the cell's endocytic machinery.
A Stroll Near the Sea, a Starfish Larva, and Some Inquisitive Minds
In the mid-1950s, several researchers observed that small molecules such as ions, amino acids, and sugars can traverse the plasma membrane through channels or pumps made of integral membrane proteins. In contrast, they learned that large molecules and particles are internalized in vesicles derived from the invagination and pinching-off of segments of the plasma membrane in a process they named endocytosis. Endocytosis also serves as a mechanism to control the protein and lipid (a type of fat) composition of the plasma membrane. Therefore, endocytosis is involved in the regulation of different cellular processes, including mitosis, antigen presentation, cell migration, and many intracellular signaling cascades.
Understanding Endocytosis
Our modern understanding of endocytosis comes from two important observations made in the 1970s. The first was by Ralph Steinman and his colleagues who used quantitative biochemical and electron microscopy approaches to demonstrate that mammalian cells in culture were able to internalize enormous amounts of the plasma membrane. These scientists made the important observation that most of the membrane that was internalized was later recycled back to the plasma membrane (Steinman, Brodie & Cohn 1976).
The second key discovery was by Michael Brown, Joseph Goldstein, and their colleagues, who were studying the regulation of cholesterol metabolism. They intended to find treatments for diseases caused by high levels of cholesterol in the blood. While studying the uptake of low-density lipoproteins (LDLs) by skin fibroblasts, they observed that LDL internalization was mediated by a specific receptor present in the surface of the cells. They coined the term "receptor-mediated endocytosis" to describe the entry of LDL into the cells (Goldstein & Brown 2009). In another series of experiments, Brown and Goldstein used electron microscopy to examine the endocytosis of LDL (and several other ligands). They observed that the vesicles responsible for the endocytosis of these molecules were "coated vesicles" surrounded by a lattice-like coat. Brown and Goldstein were awarded the Nobel Prize for their outstanding contributions to the study of cholesterol metabolism, including their discovery of receptor-mediated endocytosis.
In 1975, Barbara Pearse identified the protein that coated the endocytic vesicles. She named the protein clathrin for its ability to form lattice-like structures (Pearse 1976). By sequencing clathrin protein fragments (peptides), Pearse discovered that the protein was highly conserved in different types of cells and even in different animal species.
Viruses Enter the Scene
During the 1970s, Ari Helenius began working with the Semliki Forest virus (SFV), a virus first isolated from mosquitoes in Uganda that can cause diseases in humans and animals. Helenius and his colleagues learned that SFV initially attaches to the cell surface. Following attachment, most of the virus is rapidly engulfed by coated vesicles and sequestered in intracellular vacuoles and lysosomes. Using microscopy and biochemical studies, they concluded that SFV was entering cells by using the clathrin-mediated endocytosis pathway. Helenius's work linked the story of viruses to the story of endocytosis. How were these viruses using the cell's endocytic pathway?
The first step for a virus to invade a cell is to cross the cell's plasma membrane, which is a lipid barrier. In general, a virus consists of one or more layers of protein that enclose its viral genome. In some cases, the virus also contains enzymes, such as polymerases and proteases, which are necessary for viral replication inside the cell. Some viruses also have a lipid envelope embedded with proteins. Both enveloped and nonenveloped viruses use the proteins present on their surfaces to bind to and enter the host cell. Helenius's research showed that viruses have evolved the ability to efficiently hijack and use the cell's endocytosis mechanism to invade their host cells. The endocytic vesicles transport the incoming viral particles from the plasma membrane to the perinuclear area of the host cell, where the conditions for viral replication are optimal.
At that time, scientists knew that endosomal vesicles mature during their trip into the cell and that the pH of the vesicle's lumen gradually decreases. In their studies of SFV, Helenius and his colleagues made a second discovery. They observed that the decrease in the vesicle's pH induced conformational changes in the SFV viral particles (virions), allowing them to escape from the vesicle and enter the cell's cytoplasm, where they could begin their replication cycle. The researchers were quite excited. If their observations were correct, then they might be able to inhibit SFV entry into the cells by preventing the acidification of the endosomes. In a seminal experiment, they used compounds that prevent the endosomal acidification to see if they could block SFV cell invasion (Helenius et al. 1980). To their delight, their hypothesis was correct!
Not One, but Several Pathways to Enter Cells
Some years after the discovery of receptor-mediated endocytosis, it became clear that not all cargos were internalized via clathrin-dependent endocytosis. For example, when scientists inhibited clathrin-mediated endocytosis with a treatment that removed clathrin lattices from the plasma membrane, the cells could still efficiently internalize the toxin ricin (a protein from castor beans) (Larkin et al. 1983). How could ricin be internalized without clathrin? The most obvious explanation was that clathrin-independent modes of endocytosis existed. Scientists set out to find them.
Thanks to their work, we now know that there are two broad classes of endocytotic pathways in mammalian cells, depending on whether the uptake involves large particles (phagocytosis) or mainly solutes and water (pinocytosis). These pathways differ in the cargo they internalize, the signals needed for activation, the protein machinery involved in the endocytic process, and the fate of the internalized material (Figure 2).

Phagocytosis
Since Metchnikoff's times, scientists have come to understand phagocytosis relatively well. Phagocytosis is an active and highly regulated process that involves specific cell-surface receptors and signaling cascades. In mammals, it takes place primarily in specialized cells, such as macrophages, monocytes, and neutrophils, which function to clear away large pathogens such as bacteria and parasites and large cell debris. Recently, scientists learned that viruses such as herpes simplex 1 (which causes herpes in humans) and mimivirus (a peculiar virus originally found in a type of free-living amoeba) can use phagocytosis to enter cells (Clement et al. 2006; Ghigo et al. 2008).
Pinocytosis
In both phagocytosis and macropinocytosis, the actin cytoskeleton seems to play a very important and active role. Scientists know it does because these processes can be inhibited by drugs that block or inhibit the polymerization of actin (Mercer & Helenius 2009).
Besides macropinocytosis, there is a group of pinocytic pathways that use different proteins to coat the endocytic vesicles or pinch off vesicles from the plasma membrane. In addition, some pinocytic pathways involve different membrane lipids or lipid-modifying enzymes. There are three types of pathways:
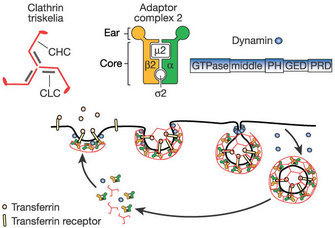
1. Clathrin-mediated endocytosis:
Clathrin-mediated endocytosis is a pinocytic pathway. Studies of clathrin structure showed that it forms triskelions comprised of three clathrin heavy chains and three light chains. The triskelions assemble into a polygonal lattice that helps deform the plasma membrane into a coated pit (Figure 3). By using live-cell imaging, dominant negative mutants (that produce nonfunctional proteins), and interference RNA (to block specific messengers), scientists showed that viruses such as the hepatitis C virus, dengue virus, and mammalian reovirus (which affects the gastrointestinal and respiratory tracts) use this pathway (Ehrilch et al. 2004; Meertens, Bertaux & Dragic 2006; van der Schaar et al. 2008). In fact, most viruses use this type of endocytosis to enter their host cells.
2. Caveolae-mediated endocytosis:
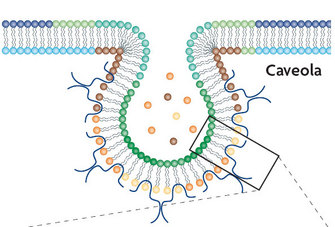
Both the SV40 virus (a virus found in monkeys and humans) and the papillomavirus (which may cause genital warts and is associated with cervical cancer) use caveolae-mediated endocytosis to infect cells. How do we know? In one case, researchers were able to block SV40 entry into monkey and human cells by preventing the formation of caveolae using a drug called nystatin. As a control, they inhibited the formation of clathrin-coated vesicles, and as they expected, the virus remained fully infective to the cells (Anderson et al. 1996). Using the same strategy together with dominant negative caveolin mutants, Jessica Smith and her group showed that papillomavirus also uses the caveolae-mediated entry pathway (Smith, Campos & Ozbun 2007).
3. Alternate endocytic pathways:
Besides the previously described, well-established endocytosis processes, there are a number of endocytic pathways that do not involve clathrin and caveolin. In these alternate endocytic pathways, the specific coat protein or pinching-off systems have not yet been identified. Scientists have learned that some viruses — including the rotavirus (the most common cause of diarrhea in infants), lymphocytic choriomeningitis virus (a rodent virus that may infect humans), and, in some cell types, the influenza (flu) virus — enter cells through these alternate pathways (Rojek, Perez & Kunz 2008; Sanchez-San Martin et al. (2004).
The Final Entry Step
Once a patch of membrane is coated, the endocytic vesicle must be split from the plasma membrane. In 1993, Sandra Schmid and her colleagues observed that the fission of the endocytic vesicles was impaired in eukaryotic cells with mutations in a large GTPase named dynamin (van der Bliek et al. 1993). Their results suggested that dynamin played a role in endocytosis. Later on, scientists confirmed that dynamin is required for phagocytosis and clathrin and caveolae-mediated endocytosis as well as for some clathrin and caveolae-independent endocytic pathways. During the late stages of vesicle formation, dynamin self-assembles at the neck of the deeply invaginated coated pits and forms a collar that strangles and releases the vesicle from the plasma membrane.
Paradoxically, viruses are often used as model cargos to study different endocytic pathways because successful viral infection can be efficiently monitored by detecting the amplified expression of viral proteins in infected cells. Also, viral particles can be easily visualized using electron microscopy, and fluorescently tagged virions can be individually tracked in living cells.
Summary
It is now clear that in addition to the well-characterized clathrin-mediated endocytic pathway, there is a multitude of endocytic pathways for the cell to internalize cargo molecules. We are only beginning to understand endocytosis. How many more endocytic pathways are there, waiting to be found?
References and Recommended Reading
Amstutz, B. et al. Subversion of CtBP1-controlled macropinocytosis by human adenovirus serotype 3. Embo Journal 27, 956–969 (2008).
Anderson H. A. et al. Bound simian virus 40 translocates to caveolin-enriched membrane domains, and its entry is inhibited by drugs that selectively disrupt caveolae. Molecular Biology of the Cell 7, 1825–1834 (1996).
Clement, C. et al. A novel role for phagocytosis-like uptake in herpes simplex virus entry. Journal of Cell Biology 174, 1009–1021 (2006).
Ehrlich, M. et al. Endocytosis by random initiation and stabilization of clathrin-coated pits. Cell 118, 591–605 (2004).
Ghigo, E. et al. Ameobal pathogen mimivirus infects macrophages through phagocytosis. PLoS Pathogens 4, e1000087 (2008).
Goldstein, J. L. & Brown, M. S. History of discovery: The LDL receptor. Arteriosclerosis, Thrombosis, and Vascular Biology 29, 431–438 (2009).
Helenius, A. et al. On the entry of Semliki Forest virus into BHK-21 cells. Journal of Cell Biology 84, 404–420 (1980).
Huang, C. Y. et al. A novel cellular protein, VPEF, facilitates vaccinia virus penetration into HeLa cells through fluid phase endocytosis. Journal of Virology 82, 7988–7999 (2008).
Larkin, J. M. et al. Depletion of intracellular potassium arrests coated pit formation and receptor-mediated endocytosis in fibroblasts. Cell 33, 273–285 (1983).
Liberali, P. et al. The closure of Pak1-dependent macropinosomes requires the phosphorylation of CtBP1/BARS. Embo Journal 27, 970–981 (2008).
Marechal, V. et al. Human immunodeficiency virus type 1 entry into macrophages mediated by macropinocytosis. Journal of Virology 75, 11166–11177 (2001).
Meertens, L., Bertaux, C. & Dragic, T. Hepatitis C virus entry requires a critical postinternalization step and delivery to early endosomes via clathrin-coated vesicles. Journal of Virology 80, 11571–11578 (2006).
Mercer, J. & Helenius, A. Virus entry by macropinocytosis. Nature Cell Biology 11, 510–520 (2009) doi:10.1038/ncb0509-510.
Pearse, B. M. F. Clathrin: A unique protein associated with intracellular transfer of membrane by coated vesicles. Proceedings of the National Academy of Sciences 73, 1255–1259 (1976).
Rojek, J. M., Perez, M. & Kunz, S. Cellular entry of lymphocytic choriomeningitis virus. Journal of Virology 82, 1505–1517 (2008).
Sanchez-San Martin, C. et al. Characterization of rotavirus cell entry. Journal of Virology 78, 2310–2318 (2004).
Smith, J. L., Campos, S. K. & Ozbun, M. A. Human papillomavirus type 31 uses a caveolin 1- and dynamin 2-mediated entry pathway for infection of human keratinocytes. Journal of Virology 81, 9922–9931 (2007).
Steinman, R. M., Brodie, S. E. & Cohn, Z. A. Membrane flow during pinocytosis: A stereologic analysis. Journal of Cell Science 68, 665–687 (1976).
Tauber, A. I. Metchnikoff and the phagocytosis theory. Nature Reviews Molecular Cell Biology 4, 897–901 (2003) doi:10.1038/nrm1244.
van der Bliek, A. M. et al. Mutations in human dynamin block an intermediate stage in coated vesicle formation. Journal of Cell Biology 122, 553–563 (1993).
van der Schaar, H. M. et al. Dissecting the cell entry pathway of dengue virus by single-particle tracking in living cells. PLoS Pathogens 4, e1000244 (2008).



 Figure 1: The phagocytosis theory
Figure 1: The phagocytosis theory


























