« Prev Next »
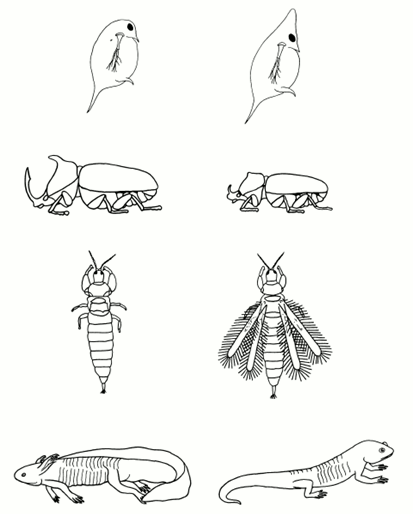
It is frequently, but erroneously, assumed that a trait that appears phenotypically in one or more discrete states is genetically determined by one or a few genes. Examples of traits that appear in this way include diseases such as pyloric stenosis and club foot, twinning in lambs, multiple digits in guinea pigs, dimorphic variation in zooplankton body form, horn size in beetles, paedomorphosis in salamanders, and wing dimorphism in many insect species (Figure 1). The explanation of how such traits can arise while having a polygenic basis can be traced back to Sir Francis Galton (a cousin of Charles Darwin) in the nineteenth century, although it is Sewall Wright and Douglas Falconer who are most properly credited with the Mendelian explanation.
The Liability Trait
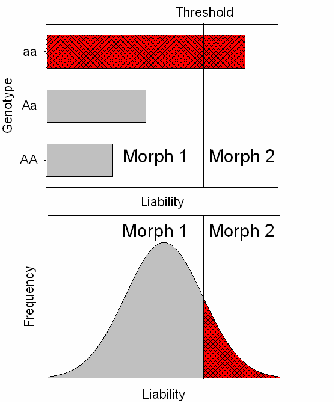
One likely candidate for the liability trait is the level of one or more hormones at a particular stage of development. For example, paedomorphosis in salamanders is strongly influenced by the growth hormone thyroxin, while horn and wing development in insects is mediated in part by juvenile hormone. In some wing-dimorphic insects, genetic control of the wing trait has been shown to be due to a single locus in which short-wingedness (brachytery or microptery)—and, in its extreme, winglessness (aptery)—is dominant. The mechanism of determination can be accommodated under the threshold concept by postulating that a single gene controls the level of the liability and that the heterozygote lies to one side of the threshold (Figure 2). The threshold model can be enlarged to include the occurrence of multiple discrete morphs, as found in the occurrence of extra digits in guinea pigs, by supposing that there are multiple thresholds. Thus, for example, individuals with liabilities lying between the first and second threshold might develop one extra digit, whereas individuals with liabilities lying beyond the second threshold might develop two extra digits.
Types of Dimorphism
The presence of dimorphic or polymorphic variation suggests that there are costs and benefits to each morph such that selection maintains both forms in the population. Numerous studies have supported this hypothesis (Roff, 1996). The different types of dimorphism studied in this context can be divided into several categories: protective dimorphism, mating dimorphism, life-cycle dimorphism, and trophic dimorphism. For the purpose of illustration, let's briefly consider the first three types.
Protective Dimorphism
Many species of zooplankton and protozoa show two discrete morphs (Figure 1). Experiments have indicated that one of these morphs is much better protected against predators than the other; however, the cost of developing this protection is a reduced fecundity or growth rate. Thus, in the absence of predators, the unprotected morph is at a reproductive advantage and is most fit, whereas in the presence of predators, the protected morph is at a survival advantage and is most fit. If predation pressure is fluctuating, then both morphs can at least temporarily be maintained in the population. However, theory shows that such a state is not evolutionarily stable and that we would expect that the development of the morphs would be determined, in part, by the presence of an environmental cue that signaled the presence of a predator. Indeed, this has been shown to be the case, as subjecting the population to a cue, such as water in which predators have been maintained, induces formation of the protected morph. Induction is due to chemicals, known as kairomones, released by the predator. The chemical identity of most kairomones is not known, but they have been shown to be made primarily of proteins. As yet, the kairomone recognition mechanism in zooplankton has not been identified. The propensity for the protected morph to be induced is genetically variable, and it is predicted that in populations in which predation is rare, the strength of the cue required to induce production of the protected morph is greater than in populations in which the predator is common.
Mating Dimorphism
Mating dimorphism is perhaps best exemplified by large and small male Atlantic salmon (Hutchings & Myers, 1988). The large male is the morph that people typically imagine when thinking of Atlantic salmon; this fish migrates to sea after one year in freshwater, spends several years growing to roughly one meter in length, and then returns to its natal stream to establish a territory and attract a female. On the other hand, the small male, called a jack, never goes to sea, matures after one year, and is approximately 30 centimeters in length. It cannot compete with the large, territorial males, and it thus attempts to "sneak" fertilization by staying close to territorial males and dashing in when female salmon release their eggs. The large males have greater mating success than the small males, but, because they take longer to mature, the larger fish suffer a higher mortality rate. Here, selection is frequency dependent in the sense that jacks cannot exist in the absence of territorial males. The two forms of male salmon are maintained in the population by the interaction of costs and benefits to the jacks and the frequency dependent selection that it creates.
As with protective dimorphism, the jack morph can be induced by certain environmental conditions. Interestingly, theory predicts that a high growth rate will favor the induction of jacks. Experience in aquaculture has shown this to be correct and a bit of a nuisance. Aquaculturalists prefer a fast-growing fish that can be marketed early; however, when salmon are fed at a high rate, this leads not only to fast growth but also to the production of a high frequency of jacks, which have little market value.
Life-Cycle Dimorphism
Life-cycle dimorphism includes a heterogeneous collection of dimorphisms, but one excellent example is wing dimorphism in insects (Roff & Fairbairn, 2007). The flight-capable insect morph has the advantage of being able to disperse to other habitats. Thus, in a heterogenous environment, new habitat patches are colonized by this morph. However, because the flightless morph does not have to invest in the machinery of flight (particularly flight fuels, which are metabolically expensive), it has a greater fecundity than the flight-capable morph. Female insects that colonize new habitat patches are flight capable but produce a mixture of offspring; then, because of its greater fecundity and the fact that it does not leave the habitat patch, the flightless morph increases in frequency over time. Indeed, the frequency of each morph in the metapopulation is a function of the persistence time of patches: as persistence time increases, so will the frequency of the flightless morph.
With insect life-cycle dimorphism, induction of a morph is typically under both genetic and environmental control, with sensitivity to environmental cues being genetically variable. Environmental cues include photoperiod and temperature, as well as food quality, quantity, and density. The proximate mechanism appears to be variation in juvenile hormone (JH) in the stage immediately prior to emergence of the adult form, although the variation in JH may be determined by events occurring earlier in an insect's life history (e.g., sensitivity to temperature may occur in early life).
The Importance of Dimorphism and Threshold Traits
Regardless of the species or type of dimorphism in question, it is clear that numerous traits are influenced by multiple genes. Many characteristics of a species that seem simple at first glance are really threshold traits, and these traits ultimately influence the success of a population.
References and Recommended Reading
Hutchings, J. A., & Myers, R. A. Mating success of alternative maturation phenotypes in male Atlantic salmon, Salmo salar. Oecologia 75, 169–174 (1988)
Roff, D. A. The evolution of threshold traits in animals. Quarterly Review of Biology 71, 3–35 (1996)
Roff, D. A., & Fairbairn, D. J. The evolution and genetics of migration in insects. BioScience 57, 155–164 (2007)






























